UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
For the fiscal year ended
or
For the transition period from ________ to ________
Commission File Number:
(Exact name of registrant as specified in its charter)
State of |
| |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
(Address of principal executive offices; Zip Code)
Registrant’s telephone number, including area code: (
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
| Trading Symbol(s) |
| Name of each exchange on which registered |
The | ||||
Ordinary Shares, no par value per share* | N/A |
* | Not for trading, but only in connection with the registration of the American Depositary Shares pursuant to requirements of the Securities and Exchange Commission. |
Securities registered or to be registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. Yes ☐
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer | ☐ |
| Accelerated filer | ☐ | |
☒ | Smaller reporting company | ||||
Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the Registrant included in the filing reflect the correction of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the Registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes
The aggregate market value of the registrant’s voting equity held by non-affiliates, computed by reference to the closing price at which the American Depositary Shares (“ADS”) were last sold on The Nasdaq Stock Market LLC as of June 30, 2024, the last business day of the registrant’s most recently completed second fiscal quarter, was approximately $
As of March 10, 2025, the registrant had
GENERAL INFORMATION
Unless otherwise indicated or the context otherwise requires, all references in this Annual Report on Form 10-K (the “Annual Report”) to the terms “Quoin,” “Quoin Ltd.,” the “Company,” “us,” “we”, “our” and the “Registrant” refer to Quoin Pharmaceuticals Ltd., an Israeli company, and its consolidated subsidiaries. In this Annual Report, the U.S. Securities and Exchange Commission is referred to as the “SEC”, the Securities Act of 1933, as amended, is referred to as the “Securities Act” and the Securities Exchange Act of 1934, as amended, is referred to as the “Exchange Act.”
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
AND SUMMARY OF RISK FACTORS
Certain information included in this Annual Report may be deemed to be “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 and other securities laws. Forward-looking statements are often characterized by the use of forward-looking terminology such as “may,” “will,” “expect,” “anticipate,” “estimate,” “continue,” “believe,” “should,” “intend,” “project” or other similar words, but are not the only way these statements are identified.
These forward-looking statements may include, but are not limited to, statements relating to our objectives, plans and strategies, statements that contain projections of results of operations or of financial condition, expected capital needs and expenses, statements relating to the research, development, completion and use of our products, and all statements (other than statements of historical facts) that address activities, events or developments that we intend, expect, project, believe or anticipate will or may occur in the future.
Forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties. We have based these forward-looking statements on assumptions and assessments made by our management in light of their experience and their perception of historical trends, current conditions, expected future developments and other factors they believe to be appropriate.
Important factors that could cause actual results, developments and business decisions to differ materially from those anticipated in these forward-looking statements include, among other things:
| ● | our limited operating history and the difficulties encountered by a small developing company; |
| ● | our history of losses and need for additional capital to fund our operations and our inability to obtain additional capital on acceptable terms, or at all; |
| ● | our lack of revenue and potential inability to be profitable; |
| ● | uncertainties of cash flows and inability to meet working capital needs; |
| ● | our ability to obtain regulatory approvals; |
| ● | our ability to generate favorable pre-clinical and clinical trial results; |
| ● | our ability to identify and develop potential product candidates; |
| ● | additional costs or delays associated with unsuccessful clinical trials; |
| ● | the inability to predict the timing of revenue from sales of a future product; |
| ● | the extensive regulatory requirements and future developmental and regulatory challenges we will still face even if we obtain approval for a product candidate; |
| ● | our ability to obtain or maintain orphan drug designation or data exclusivity for our product candidates; |
| ● | our ability to obtain Orphan Disease and Rare Pediatric Disease designations for our product candidates; |
1
| ● | the potential oversight of programs or product candidates that may be more profitable or more successful; |
| ● | our manufacturing processes may not be validated and our methodology may not be accepted by the scientific community; |
| ● | the ability to conduct clinical trials, because of difficulties enrolling patients or other reasons; |
| ● | the requirements of being a publicly traded company may strain our resources; |
| ● | potential adverse effects resulting from failure to maintain effective internal controls; |
| ● | our ability to comply with the applicable continued listing requirements of Nasdaq; |
| ● | the potential negative impact on our securities price and trading volume if securities or industry analysts do not publish reports about us or if they adversely change their recommendations about our business; |
| ● | the potential volatility of the market price for our ADSs; |
| ● | the potential dilution of our shareholders’ potential ownership due to future issuances of share capital; |
| ● | the requirement for holders of ADSs to act through the depositary to exercise their rights; |
| ● | the potential limitations on ADS holders with respect to the transfer of their ADSs; |
| ● | the risks of securities class action litigation; and |
| ● | other risks and uncertainties, including those listed under Part I, Item 1A of this Annual Report titled “Risk Factors.” |
You are urged to carefully review and consider the various disclosures made throughout this Annual Report which are designed to advise interested parties of the risks and factors that may affect our business, financial condition, results of operations and prospects.
You should not put undue reliance on any forward-looking statements. Although the forward-looking statements in this Annual Report are based on our beliefs, assumptions and expectations, taking into account all information currently available to us, we cannot guarantee future transactions, results, performance, achievements or outcomes. No assurance can be made that the expectations reflected in our forward-looking statements will be attained, or that deviations from them will not be material and adverse. We undertake no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
In addition, certain sections of this Annual Report contain information obtained from independent industry sources and other sources that we have not independently verified.
2
SUMMARY OF RISK FACTORS
An investment in our Company is subject to a number of risks. Set forth below is a high-level summary of some, but not all, of these risks. You should review and consider carefully the risks and uncertainties described in more detail in “Part I, Item 1A. Risk Factors” of this Annual Report, which includes a more complete discussion of the risks summarized below as well as a discussion of other risks related to our business and an investment in our securities.
Risks Related to Our Financial Position and Capital Requirements
| ● | We have a limited operating history that you can use to evaluate us, and the likelihood of our success must be considered in light of the problems, expenses, difficulties, complications and delays frequently encountered by a small developing company. |
| ● | We have incurred significant losses since our inception and have limited cash available for our operations. |
| ● | We have never generated any revenue from product sales or any other sources since inception, and may never be profitable. |
| ● | We expect that we will need to raise additional capital, which may not be available on acceptable terms, or at all. |
Risks Related to the Discovery and Development of Product Candidates
| ● | Preclinical and clinical studies of our product candidates may not be successful. If we are unable to generate successful results from preclinical and clinical studies of our product candidates, or experience significant delays in doing so, our business may be materially harmed. |
| ● | We may not be successful in our efforts to identify or develop potential product candidates. |
| ● | If clinical trials of our product candidates fail to demonstrate safety and efficacy to the satisfaction of regulatory authorities or do not otherwise produce positive results, we may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our product candidates. |
| ● | Any of our product candidates may cause undesirable side effects or have other properties impacting safety that could delay or prevent their regulatory approval or limit the scope of any approved label or market acceptance. |
| ● | Even if we complete the necessary preclinical studies and clinical trials, we cannot predict whether or when we will obtain regulatory approval to commercialize a product candidate and we cannot, therefore, predict the timing of any revenue from a future product. |
| ● | Even if we obtain regulatory approval for a product candidate, we will still face extensive regulatory requirements and our products may face future development and regulatory challenges. |
| ● | We may not be able to obtain or maintain orphan drug designation or exclusivity for our product candidates. |
| ● | We may pursue Rare Pediatric Disease designation for QRX003 for the treatment of NS or other of our product candidates. There is no assurance that we will obtain such designation. Moreover, a Rare Pediatric Disease designation by the FDA does not guarantee that the NDA for the product will qualify for a priority review voucher upon approval, and it does not lead to a faster development or regulatory review process, or increase the likelihood that any of our product candidates will receive marketing approval. |
| ● | We may use our financial and human resources to pursue a particular research program or product candidate and fail to capitalize on programs or product candidates that may be more profitable or for which there is a greater likelihood of success. |
| ● | We expect competition in the marketplace for our product candidates, should any of them receive regulatory approval. |
Risks Related to Our Reliance on Third Parties
| ● | We rely on third parties to conduct some aspects of our compound formulation, research and preclinical studies, and those third parties may not perform satisfactorily, including failing to meet deadlines for the completion of such formulation, research or testing. |
| ● | We rely, or will rely, on third-party manufacturers to produce the supply of our preclinical product, clinical product candidates and commercial supplies of any approved product candidates. |
3
Risks Related to Our Intellectual Property
| ● | If we are unable to obtain or protect intellectual property rights related to our future products and product candidates, we may not be able to compete effectively in our markets. |
| ● | Third-party claims of intellectual property infringement may prevent or delay our development and commercialization efforts. |
| ● | If we fail to obtain licenses or comply with our obligations in these agreements under which we license intellectual property rights from third parties or otherwise experience disruptions to our business relationships with our licensors, we could lose license rights that are important to our business. |
Other Risks Related to Our Business Operations and Industry
| ● | Our future success depends on our ability to attract and retain key executives and to attract, retain and motivate qualified personnel. |
| ● | We may need to expand our organization and may experience difficulties in managing our growth, which could disrupt our operations. |
| ● | Our operations may be impacted from changes to current regulations and future legislation. |
Risks Related to Us Being an Israeli Company
| ● | Shareholders may have difficulties enforcing a U.S. judgment, including judgments based upon the civil liability provisions of the U.S. federal securities laws, against us or our executive officers and directors, or asserting U.S. securities laws claims in Israel. |
| ● | The rights and responsibilities of our shareholders are governed by Israeli law, which may differ in some respects from the rights and responsibilities of shareholders of U.S. corporations. |
| ● | Provisions of Israeli law may delay, prevent or otherwise impede a merger with, or an acquisition of, our company, which could prevent a change of control, even when the terms of such a transaction are favorable to us and our shareholders. |
Risks Related to Ownership of Our ADSs and Ordinary Shares
| ● | We do not know whether a market for our securities will be sustained and as a result it may be difficult for shareholders to sell our securities. |
| ● | The requirements of being a publicly traded company may strain our resources and divert management’s attention. |
| ● | Failure to achieve and maintain effective internal controls in accordance with Section 404 of the Sarbanes-Oxley Act could have a material adverse effect on our business, results of operation or financial condition. In addition, current and potential shareholders could lose confidence in our financial reporting, which could have a material adverse effect on the price of the ADSs. |
| ● | Our failure to meet the continued listing requirements of the Nasdaq Capital Market could result in a delisting of our ADSs. |
| ● | The market price for our ADSs may be volatile. |
| ● | Substantial future sales or perceived potential sales of our ADSs in the public market could cause the price of our ADSs decline. |
| ● | Our shareholders may experience substantial dilution as a result of future issuances of our equity securities. |
| ● | We have not paid, and do not intend to pay, dividends on our ordinary shares and, therefore, unless our traded securities appreciate in value, our investors may not benefit from holding our securities. |
| ● | Holders of ADSs must act through the depositary to exercise their rights. |
| ● | Holders of ADSs may be subject to limitations on the transfer of their ADSs. |
4
Table of Contents
5
PART I
Item 1.Business
Company Overview
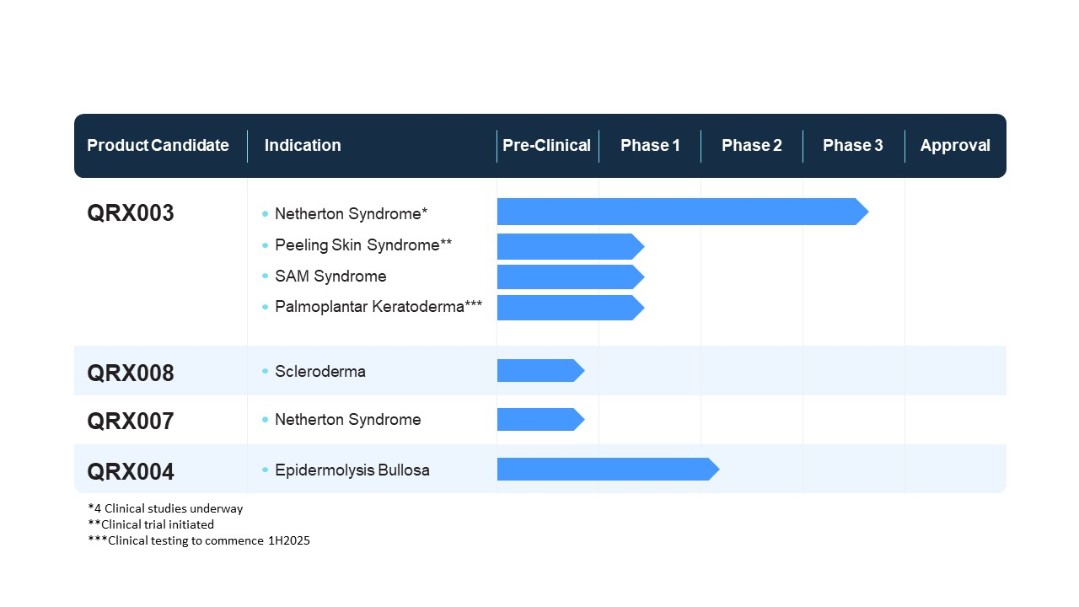
We are a late-stage clinical specialty pharmaceutical company focused on the development and commercialization of therapeutic products that treat rare and orphan diseases for which there are currently very limited or no approved treatments or cures. Our initial focus is on the development of products, using our proprietary owned and in-licensed drug delivery technologies, that could help address rare genetic diseases. Our lead product, QRX003, is under clinical development as a potential treatment for Netherton Syndrome (“NS”), a rare hereditary genetic disease. QRX003 is currently being tested in three regulatory clinical studies under an open Investigational New Drug (“IND”) application with the Food and Drug Administration (“FDA”). We have opened five clinical sites in the United States (“US”) and intend to open a sixth clinical site at Northwestern University. We are expanding our trials internationally into the Middle East, the United Kingdom and additional countries in Europe, including Spain and Germany. QRX003 is currently being tested in a pediatric NS patient at the Children’s Hospital in Dublin, Ireland and we intend to expand this study to include additional children with NS in Spain, the United Kingdom and potentially other countries. QRX003 is also being developed as a potential treatment for Peeling Skin Syndrome with the first subject being treated in New Zealand. In addition, we entered into two separate Research Agreements with the Queensland University of Technology (“QUT”), under which we have obtained an option for global licenses to QRX007 for the potential treatment of NS and QRX008 for the potential treatment of scleroderma, as well as a Research Agreement with the University College Cork (“UCC”) for the development of novel topical formulations of rapamycin (sirolimus) as potential treatments for a number of rare and orphan diseases. We are also initiating the development of novel topical formulations of rapamycin using our in-licensed technology as potential treatments for microcystic lymphatic malformations, venous malformations and angifibromas. Other development products in our pipeline include QRX004 as a potential treatment for Recessive Dystrophic Epidermolysis Bullosa (“RDEB”). To date, no products have been commercialized and no revenue has been generated.
Our Product Candidates
QRX003
QRX003 is a topical lotion being developed for the treatment of NS. The active ingredient in QRX003 is a broad-spectrum serine protease inhibitor whose mechanism of action is to target the kallikreins responsible for the process of skin shedding. Due to the genetic mutation of the SPINK5 gene, which results in the absence of the kallikrein regulating LEKTI protein, these kallikreins go unregulated and become hyperactive resulting in the uncontrolled desquamation that leads to the highly defective skin barrier in NS patients. When applied to the skin, QRX003 is designed to perform the function of the missing LEKTI protein and down regulate, but not completely stop, the activity of kallikreins, leading to a more normalized skin shedding process and the formation of a stronger and more effective skin barrier.
While several other companies are pursuing the development of products to treat NS, we believe, to date we are the only company that is actively dosing subjects in multiple NS clinical studies under an open IND with the FDA. QRX003 was developed using Invisicare® polymer delivery technology licensed from Skinvisible Pharmaceuticals, Inc. (“Skinvisible”). See “—Intellectual Property—License Agreement with Skinvisible.” The Invisicare® polymer delivery technology is an optimized topical delivery system that moisturizes the skin whilst simultaneously providing a protective barrier against allergens, toxins and other environmental agents.
QRX004
QRX004 contains two active ingredients as a potential treatment for RDEB. One active ingredient induces a read-through of nonsense mutations and leads to creation of robust and sustained type VII collagen, which is designed to improve wound closure, reduce blistering and stronger skin. This product is being developed using Invisicare® delivery technology in-licensed from Skinvisible. See “—Intellectual Property—License Agreement with Skinvisible.”
QRX007 and QRX008
In November 2021, we entered into the Research Agreement with QUT, pursuant to which we have an option for up to six months after the project completion to in-license the QRX007 product. QRX007 is a bi-functional protein designed to be highly selective and potent inhibitor of the KLK5 and KLK7 kallikreins as a potential treatment for NS. QRX007 is in pre-clinical testing for NS. In May
6
2022, we entered into another Research Agreement with QUT, pursuant to which we have an option for up to six months after the project completion to in-license a small molecule VLA - 4 inhibitor, the QRX008 product. QRX008 is a potential treatment for scleroderma, a rare autoimmune disease for which there is currently no approved treatment, and it is under early-stage development by QUT. Quoin is planning to schedule a meeting with QUT to discuss the future direction of both research programs.
Research Agreement with University College Cork
On June 10, 2024 we signed a research agreement with The School of Pharmacy at University College Cork (UCC). The scope of the agreement encompasses the development of novel topical formulations of rapamycin (sirolimus) as potential treatments for a number of rare and orphan diseases for which there are currently very limited or no approved therapies or cures. UCC will apply its proprietary dissolvable microneedle delivery technology along with other formulation approaches to optimize the local delivery of rapamycin and potentially enhance its therapeutic effectiveness as a potential treatment for several pre-identified clinical targets.
Under the terms of the agreement, we will fund a research program at UCC over an anticipated 2-1/2 year period to investigate the development of a number of topical rapamycin formulations for future development as potential treatments for several rare and orphan diseases, where it is believed that the drug’s mechanism of action may provide for clinical efficacy in these settings. Following completion of the research program, we will have the option to advance the clinical development of rapamycin formulations developed by UCC. The terms of the agreement do not require us to pay any upfront license or milestone fees or any royalties based on future product sales.
Our Current Product Pipeline
Netherton Syndrome
NS is a rare autosomal recessive genetic disease affecting approximately 6,000 – 8,000 patients combined in the U.S. and Europe which is caused by a mutation in the SPINK5 gene and has an incidence of approximately 1/200,000 births. The SPINK5 gene encodes a protein, called lympho-epithelial kazal type related inhibitor (“LEKTI”) that serves as a brake system on the activity of certain proteases (enzymes that digest proteins) in the skin called Kallikreins. The absence of the LEKTI protein, as a result of the genetic defect that
7
causes NS, leads to unregulated protease activity in the skin by the Kallikreins, resulting in too few layers of the outer skin (stratum corneum), thereby leading to a highly defective and compromised skin barrier. As a result, patients with NS suffer from a variety of medical issues including regular, severe infections, skin cancer, chronic pruritis, asthma, and allergies among others.
Newborns with NS have reddened skin (erythroderma) and sometimes a thick parchment-like covering of skin (collodion membrane). The skin is red and scaly all over. Hair shafts are fragile and break easily due to trichorrhexis or “bamboo hair,” resulting in short sparse hair. In older children and adults, the scaling may have a distinctive circular pattern (ichthyosis linearis circumflexa). Babies with NS may be born prematurely. Trouble gaining weight in infancy and childhood is common and can be severe. Infants may also have recurrent skin infections and septicemia. They may develop hypernatremia (elevated sodium levels in the blood) due to excessive loss of fluid from the skin surface. Because hairs may not be affected at birth, and then may be sparse in all babies in the first months of life, the characteristic hair defect that is diagnostic of NS may not be detected initially. Infants with NS may be misdiagnosed as having congenital ichthyosiform erythroderma, atopic dermatitis or psoriasis. Atopic dermatitis (red, itchy patches of skin) may be present, and a cradle cap-like scale and redness may appear on the face, scalp and eyebrows.
There are currently no approved therapies to treat NS. In the absence of an approved therapeutic product, patients can only obtain minor symptomatic relief, generally by the regular use of emollients and moisturizing creams and lotions. Other topical agents must be used with caution because the highly compromised skin in NS patients may allow ingredients from some topically applied medications to be excessively absorbed into the bloodstream, which may pose a danger to the patient. Use of topical keratolytic agents, such as urea or lactic acid derivatives, may be limited by skin irritation and is generally reserved for older children or adults. Base line treatment may also include oral antihistamines, which can help to control the itchy, eczematous component, and topical or systemic antibiotics as needed. Oral and topical steroids and systemic biologics may be beneficial in reducing inflammation and the eczematous component of the disease. However, the well-documented side effects of long-term steroid use need to be carefully considered. There is a critical need for a new and effective treatment for NS.
Regulatory Status of QRX003 for the Treatment of NS
Our lead asset, QRX003, is currently in late-stage clinical development in the U.S. under an open IND application with the FDA. We submitted an IND in March 2022 to the FDA to initiate a clinical study of QRX003 in adult NS patients. We received a ‘Study May Proceed’ notification from the FDA on June 13, 2022, which cleared us to initiate clinical testing of QRX003 in NS patients. This study is fully up and running and five clinical sites in the U.S. have been opened and are actively recruiting and dosing patients. This study originally was designed as a randomized, double blinded assessment of two different doses of QRX003 (4% and 2%) versus a placebo vehicle in 18 adult NS patients. Initially, the test materials were applied once daily, over a twelve-week period, to pre-selected areas of the patient’s body, primarily the arms and lower legs. Based on discussions with the FDA, a number of different clinical endpoints are being assessed in the study, including but not limited to, an Investigators Global Assessment (IGA), Patient’s Global Assessment (PaGA), Modified Ichthyosis Area Severity Index (M-IASI) and Pruritus.
In March 2022, we submitted a briefing document to the European Medicines Agency (“EMA”) seeking guidance regarding the clinical and regulatory development of QRX003 for the European Union (“EU”), to which we received comprehensive and constructive feedback. We also intend to apply for Orphan Drug status in the U.S. and Europe as well as Rare Pediatric Disease designation in the U.S. for QRX003.
In November 2022, we submitted a protocol for our second clinical study in NS patients to the FDA under our open IND (the “Open Label Study”). This study was cleared by the FDA to initiate clinical testing in December 2022. This study originally was designed to be conducted in ten adult NS patients who are currently receiving, and will continue to do so throughout the study, off-label systemic therapy, primarily systemic biologic therapy. This is an open-label study with no placebo control in which all enrolled subjects receive QRX003. Both of these NS clinical studies are running concurrently and utilize the same clinical trial sites and investigators in the US.
On October 24, 2023, we released positive initial clinical results obtained from the first six evaluable subjects in the Open Label Study. Five of the six subjects reported absence of or negligible pruritiu, with one subject reporting no change; three subjects demonstrated improvement with respect to skin appearance on completion of the study and three showing such improvements during the study though not necessarily on completion of the study. In addition, all six subjects reported a favorable impression of QRX003 across a number of key metrics, including: ease of use, time to start working, overall satisfaction, lack of side effects.
As a result of this positive initial data and the absence of any adverse events from both studies, on November 8, 2023 we submitted a number of protocol amendments to the FDA, under our open IND, with a view to optimizing both studies and potentially leading to
8
even better clinical outcomes and a more rapid regulatory approval. These protocol amendments included eliminating the lower 2% dose from the double-blinded study, modifying the dosing frequency from once-daily to twice-daily and increasing the number of subjects from 18 to 30. For the Open Label Study, the number of subjects was increased from 10 to 20 and dosing was modified from once-daily to twice-daily. On December 13, 2023, we announced that we were cleared by the FDA to implement these protocol amendments. In February 2024 we submitted a further protocol amendment to the FDA requesting permission to lower the age of eligibility for participation in both studies to 14 years and older from 18 years and older. On March 4, 2024 we announced that we were cleared to implement this protocol amendment. All protocol amendments have now been implemented and going forward participants in both regulatory studies will be dosed twice-daily with those enrolled in the blinded study receiving either a 4% dose of QRX003 or a placebo, while subjects in the Open Label Study will receive a 4% dose of QRX003 only.
On June 27, 2024 we announced that we will expand our ongoing Netherton Syndrome clinical studies to include international sites. The first international site will be opened at a research hospital in Saudi Arabia. This hospital is currently treating a number of NS patients who will now become eligible for recruitment into our studies. An experienced Clinical Research Organization has been engaged to manage the study locally.
On August 6, 2024, we announced the planned initiation of an investigator-led clinical study in New Zealand for QRX003 in pediatric patients with Peeling Skin Syndrome. This rare genetic condition currently has no approved treatments or cures. The first clinical site has been opened and dosing of the patient has commenced Quoin is actively evaluating additional clinical sites in other countries.
On October 22, 2024 we announced the further expansion of our ongoing Netherton Syndrome clinical studies to include two additional international sites in the United Kingdom (UK). Both of these sites, Great Ormond Street Hospital and St. Thomas’ Hospital, which are located in London, are highly qualified centers of excellence for treating Netherton Syndrome patients in the UK. Both sites have available cohorts of patients potentially eligible to participate in Quoin’s studies. A globally recognized expert in the treatment of NS patients has been appointed as Principal Investigator for the UK studies and a Clinical Research Organization has been engaged.
The UK and Saudi Arabia sites will operate under the auspices of Quoin’s open IND application with the FDA. Quoin is also in advanced stage of preparation for the opening of additional sites in several other Western European countries, including Spain and Germany, and is concluding a feasibility study in multiple Eastern European countries with both territories having available cohorts of patients with Netherton Syndrome.
On November 5, 2024, we announced that QRX003 is being tested in a pediatric NS patient at the Children’s Hospital in Dublin, Ireland and that we intend to expand this study to include up to three additional pediatric patients with NS in Spain and up to six additional pediatric patients in the UK.
On December 19, 2024, we announced FDA clearance to initiate a new additional Netherton Syndrome (NS) clinical study for QRX003. The study will be conducted by Dr. Amy Paller, of Northwestern University. It is planned that up to eight subjects will be enrolled into the study and will have QRX003 applied twice daily to greater than 80% of their entire body surface area (BSA) over a 12-week period. By comparison, in Quoin’s ongoing open-label and double-blinded clinical studies, QRX003 is applied to approximately 20% of the subject’s BSA, typically the arms and lower leg. This new study, designed to mimic how NS patients will use QRX003 if approved, represents the most extensive use of QRX003 in a clinical setting to date. It is anticipated that the data generated from this study will be used to supplement the data package to support the potential regulatory approval of QRX003 as a treatment for NS.
In December 2024 and January 2025, we provided data from the first subject dosed twice daily in our ongoing open label study. On December 18, 2024, we announced positive data after 6-weeks of dosing with QRX003, marking the midpoint of testing. On January 6, 2025 we shared clinical data for that subject upon completion of testing which showed clear improvements from baseline through 12 weeks of twice-daily dosing with QRX003 across all measured clinical endpoints. In addition, the patient satisfaction scores across multiple assessed metrics which were highly positive after 6 weeks of testing demonstrated even further improvement after 12 weeks. No adverse events were reported for the subject during this testing period. On January 23, 2025, we issued data on that same subject four weeks post-discontinuation of treatment with QRX003 which should showed that all of the positive clinical benefits observed after 12 weeks of testing with QRX003 were completely reversed by 4 weeks after discontinuation of treatment resulting in the subject’s disease state reverting to the baseline status observed prior to QRX003 treatment.
9
The following table sets forth the first patient data from the open label study dosed twice daily with QRX003.
End Point | Baseline | 6 weeks | 12 weeks | 4 weeks post |
M-IASI* | 18 | 4 | 3 | 18 |
WINRS** | 7 | 4 | 2 | 8 |
IGA*** | Moderate | Mild | Almost Clear | Moderate |
*M-IASI: Modified Ichthyosis Area of Severity Index, a score used to assess the severity and extent of skin symptoms associated with ichthyosis. Lower scores indicate improvement.
**WINRS: Worst Itch Numeric Rating Scale, which measures the severity of itch on an 11-point scale (0 = no itch, 10 = worst imaginable itch).
***IGA: Investigator’s Global Assessment, which uses descriptive categories (e.g., clear, mild, moderate, severe) to evaluate the overall severity of Netherton Syndrome symptoms.
In December 2024 and January 2025 we also provided data on our ongoing pediatric Netherton Syndrome study. On December 18, 2024, we announced positive data from the initial 12 days of dosing in our ongoing 12-week Investigator Pediatric Study, namely that a significant improvement was observed in the skin area treated with QRX003 versus the non-treated area. Specifically, at baseline prior to dosing with QRX003, the IGA assessment of the subject’s skin was classified as “severe.” After 12 days of treatment with QRX003, this was improved to “mild-moderate,” representing a very rapid improvement in skin appearance. On January 14, 2025, we announced that after six weeks of dosing, the IGA assessment of the subject’s skin was classified as “mild.” As a result of these positive results, we further announced on January 14, 2024, that the subject was being transitioned to having QRX003 applied to their whole body surface area (BSA) as opposed to the approximately 20% of their BSA that was tested for the initial six weeks. In addition, there were no adverse events reported for the subject during this initial six week testing period.
On February 27, 2025, we announced positive clinical data from our ongoing Investigator Pediatric Netherton Syndrome (NS) study. Both key clinical endpoints, Investigator’s Global Assessment (IGA) and Pruritus or itch, demonstrated highly significant clinical improvements from baseline after two weeks of treatment with QRX003 on patient’s whole body as set forth in the table below. No adverse events have been reported to date.
Table 1: Results for First Pediatric Patient Receiving QRX003 ‘Whole-Body’ Application
Endpoint* | Baseline | Two Week |
Investigator Global Assessment | 7 | 1–2 |
Pruritus | 5 | 1 |
*Both IGA and Pruritus scores based on a 0-10 scale.
Commercial Strategy
QRX003 has the potential to become the first approved treatment for NS to reach the market both in the U.S. and Europe and may therefore likely be used in a large proportion of patients. We currently anticipate that QRX003, if approved, would be applied once or twice daily over the patient’s entire body. Because NS is a chronic disease and does not spontaneously resolve, we believe there is an opportunity for the product, should it be approved, for long-term chronic use.
We intend to self-commercialize QRX003, and other rare disease products the company may develop, if approved, in both the U.S. and Europe. Because of the very low number of patients and the fact that diagnosis and treatment are generally provided by a relatively small number of board-certified dermatologists in major urban areas, this concentration of care will enable us to market QRX003 with a small, dedicated salesforce to target patients and caregivers in the U.S. Outside of the U.S., we have currently established nine separate marketing partnerships for QRX003 that cover 61 different countries including Australia, New Zealand, the Middle East, Central and Eastern Europe, Turkey, Canada, China, Taiwan, Hong Kong, Singapore and the major countries in Latin America.
Once the commercial infrastructure has been established for QRX003 for NS, the subsequent approval and addition of new rare disease indications or products will not result in a significant increase in the size of that infrastructure. In particular, we believe it is highly likely that physicians who treat patients with NS would also treat patients with Peeling Skin Syndrome, SAM Syndrome,
10
Palmoplantar Keratoderma and Epidermolysis Bullosa, enabling our sales personnel to discuss several products, once approved, with each treating physician.
A key element of our commercial strategy will be to add new products to our portfolio beyond those which we develop ourselves. This will be achieved through in-licensing, acquisition or the establishment of research partnerships with universities or other institutions. While it is intended that these products will treat rare and orphan diseases, we may widen our scope of interest beyond rare skin diseases as we believe this will not add significant incremental burden to an already established commercial infrastructure.
Pricing
We have not conducted a formal pricing analysis of QRX003 in NS. We anticipate that pricing at launch may be influenced by the product label negotiated with the FDA, by pharmacoeconomic data developed to support pricing and the potential for greater sales under negotiated government contracts.
Competition
The clinical biotechnology industry is a competitive industry characterized by technological innovation and growth. Our competitors include other biotechnology and pharmaceutical companies, academic institutions, and public and private research institutions. These entities engage in efforts to research, discover and develop new medicines and treatments for substance use. These entities also seek patent protection and licensing revenues for their research results and may compete with us in recruiting skilled talent. Some of these entities are larger and better funded than us. Our management can make no assurances that we can effectively compete with these competitors. We also may be unable to keep pace with technological developments and other market factors. Currently, there are no approved products to treat NS. However, to our knowledge, there are a number of therapeutic products at various stages of development for the treatment of NS, including candidates from LifeMax Laboratories, Inc., Krystal Biotech, Inc., Sixera Pharmaceuticals, ResVita Bio, BioCryst and Azitra Inc. As of now, to the best of our knowledge, only Azitra is actively dosing subjects in clinical studies on NS patients under an open IND.
Manufacturing
Our manufacturing strategy is to contract with third parties to manufacture our clinical and commercial active pharmaceutical ingredient (API) and drug product supplies. The formulation and processes used to manufacture our products are proprietary, and we have agreements with various third-party manufacturers and suppliers, such as Ferndale Contract Manufacturing and TopChem Pharmaceuticals Limited, that are intended to restrict these manufacturers from using or revealing any unpublished proprietary information.
Intellectual Property
Patents and Trademarks
Our success depends in part on our ability to obtain and maintain patents and other forms of intellectual property rights, including in-licenses of intellectual property rights of others, for our product candidates, methods used to develop and manufacture our product candidates and methods for treating patients using our product candidates, as well as our ability to preserve our trade secrets, to prevent third parties from infringing upon our proprietary rights and to operate without infringing upon the proprietary rights of others. The strength of patents in the biotechnology and pharmaceutical field involves complex legal and scientific questions and can be uncertain.
11
The following table lists patents and trademarks that we use in our business.
Patents | Trademarks |
U.S. Patent No. 8,318,818 (exp. July 10, 2025) directed to Invisicare® technology licensed from Skinvisible. | U.S. Trademark Registration No. 6918421 for word mark “RARE DISEASES ARE ONLY RARE IF YOU DON’T LIVE WITH ONE” filed by Quoin Pharmaceuticals, Inc. |
U.S. and PCT patent applications directed to adjunctive therapy for NS with QRX003 filed by Quoin Pharmaceuticals Inc. | U.S. Trademark Registration No. 7071539 for design and words “Quoin Pharmaceuticals” filed by Quoin Pharmaceuticals, Inc. |
U.S. Trademark Application No. 98/184,357 for word mark “QELTIQ” filed by Quoin Pharmaceuticals, Inc. | |
U.S. provisional patent application directed to rapamycin formulation for treatment of particular skin disorders | U.S. Trademark Application No. 98/850,670 for word mark “NETHERTON NOW: BECAUSE EVERYONE DESERVES TO FEEL COMFORTABLE IN THEIR OWN SKIN” filed by Quoin Pharmaceuticals, Inc. |
U.S. provisional patent application directed to use of QRX003 according to a new dosage and for particular skin disorders. | U.S. Trademark Application No. 98/850,671 for word mark “NETHERTON NOW” filed by Quoin Pharmaceuticals, Inc. |
License Agreement with Skinvisible
In October 2019, we entered into the Exclusive Licensing Agreement (as amended from time to time, the “License Agreement”) with Skinvisible Pharmaceuticals, Inc. (“Skinvisible”), under which Skinvisible granted us an exclusive royalty-bearing license relating to the production and manufacture of prescription drug products related to certain patents held by Skinvisible, including those related to QRX003 and QRX004. We made Skinvisible a one-time non-refundable, non-creditable license fee of $1 million (the “License Fee”). In addition, we agreed to pay Skinvisible a single digit royalty percentage of our net sales revenues for any licensed product covered by the patent rights licensed to us under the License Agreement. We also agreed to pay Skinvisible 25% of any revenues we receive as royalties in the event that we sublicense any licensed products to a third party. The License Agreement also requires that we make a $5 million payment to Skinvisible upon receiving approval in the U.S. for the first drug product developed using intellectual property licensed thereunder.
Trade Secrets
In addition to the protection afforded by patents, we rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable, including processes for which patents are difficult to enforce and any other elements of our drug discovery and development processes that involve proprietary know-how, information or technology that is not covered by patents. Although each of our employees agrees to assign their inventions to us through an employee inventions agreement, and all of our employees, consultants, advisors and any third parties who have access to our proprietary know-how, information or technology are required to enter into confidentiality agreements, we cannot provide any assurances that all such agreements have been duly executed, that our trade secrets and other confidential proprietary information will not be disclosed or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques.
Regulatory
General
Government authorities in the United States and other countries extensively regulate, among other things, the pre-clinical and clinical testing, manufacturing, labeling, storage, record-keeping, advertising, promotion, export, marketing and distribution of pharmaceutical products. In the United States, pharmaceutical products are subject to rigorous review under the Federal Food, Drug, and Cosmetic Act, and other federal statutes and regulations.
FDA Approval Process
To obtain approval of our product candidates from the FDA, we must, among other requirements, demonstrate in preclinical studies and well-controlled clinical trials that the product is safe and effective for its intended use and that the manufacturing facilities, processes and controls are adequate to preserve the drug’s identity, strength, quality and purity. The drug approval process generally includes:
| ● | preclinical laboratory tests, in vitro and in vivo preclinical studies and formulation and stability studies; |
12
| ● | the submission to the FDA of an application for human clinical testing, which is known as an IND application; |
| ● | adequate and well-controlled human clinical trials to demonstrate the safety and effectiveness of the drug; |
| ● | the submission to the FDA of a new drug application (“NDA”) for a drug; and |
| ● | satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the drug is produced to assess compliance with current GMP (“cGMP”) requirements to assure that the facilities, methods and controls are adequate to preserve the drug’s identity, strength, quality and purity; |
| ● | the approval by the FDA of an NDA. |
Preclinical tests include laboratory evaluations of product chemistry, toxicity and formulation, as well as animal studies. Preclinical trials must also be conducted in accordance with FDA and comparable foreign authorities’ legal requirements, regulations or guidelines, including Good Laboratory Practice. Violations of these regulations can, in some cases, lead to invalidation of the studies, requiring them to be replicated. Before human clinical testing can begin, a sponsor must submit the results of the preclinical tests, together with manufacturing information and analytical data, to the FDA as part of the IND, a request for authorization from the FDA to administer an investigational new drug product to humans.
A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA has neither commented on nor questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin. Clinical trials involve the administration of the investigational drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations; (ii) in compliance with good clinical practices (“GCP”), an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators, and monitors; as well as (iii) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety, and the effectiveness criteria to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as part of the IND. Clinical trials must be conducted under the supervision of one or more qualified investigators pursuant to protocols detailing, among other things, the objectives of the trial, dosing procedures, subject selection and exclusion criteria and the safety and effectiveness criteria to be evaluated. For each institution where a clinical trial will be conducted, an institutional review board (“IRB”) must review and approve the clinical trial protocol and informed consent form required to be provided to each trial subject or his or her legal representative prior to a clinical trial commencing, and conduct on-going monitoring of the study until completed or termination to assure that appropriate steps are taken to protect the human subjects participating in the research.
The FDA may order the temporary or permanent discontinuation of a clinical trial at any time, or impose other sanctions, if it believes that the clinical trial either is not being conducted in accordance with FDA regulations or presents an unacceptable risk to the clinical trial patients. Imposition of a clinical hold may be full or partial. The IRB will also monitor the clinical trial until completed. An IRB may also require the clinical trial at the site to be halted, either temporarily or permanently, for failure to comply with the IRB’s requirements, or may impose other conditions. Additionally, some clinical trials are overseen by an independent group of qualified experts organized by the clinical trial sponsor, known as a data safety monitoring board or committee. This group provides authorization for whether a trial may move forward at designated checkpoints based on access to certain data from the trial.
Human clinical trials are typically conducted in three sequential phases that may overlap or be combined:
Phase 1: In Phase 1 studies, the product candidate is initially introduced into healthy human volunteers and tested for safety, dosage and tolerability, absorption, distribution, metabolism and excretion and, effect on the body.
Phase 2: Phase 2 studies are conducted in a limited patient population. These studies continue to evaluate safety while gathering preliminary data on effectiveness in patients with the targeted disease or condition.
Phase 3: Phase 3 trials further evaluate efficacy and safety in an expanded patient population, generally at geographically dispersed clinical study sites. These clinical trials are intended to establish the overall risk-benefit ratio of the product candidate and provide, if appropriate, an adequate basis for product labeling. In most cases, the FDA requires two adequate and well-controlled Phase 3 clinical trials to demonstrate the safety and efficacy of the drug. In rare instances, a single Phase 3 trial may be sufficient when either (1) the trial is a large, multicenter trial demonstrating internal consistency and a statistically very persuasive finding of a clinically meaningful effect on mortality, irreversible morbidity or prevention of a disease with a
13
potentially serious outcome and confirmation of the result in a second trial would be practically or ethically impossible or (2) the single trial is supported by other confirmatory evidence.
Post-approval studies, sometimes referred to as Phase 4 studies, may be conducted after initial marketing approval. These studies are used to gather additional information about a product’s safety and/or efficacy in patients affected by the therapeutic indication.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing and distribution of the product may begin in the United States. The NDA must include the results of all preclinical, clinical, and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture, and controls. The submission of most NDAs is subject to the payment of a substantial application user fee. Under an approved NDA, the applicant is also subject to an annual program fee. These fees typically increase annually. An NDA for a drug that has been designated as an orphan drug is not subject to an application fee, unless the NDA includes an indication for other than a rare disease or condition.
Pursuant to the current Prescription Drug User Fee Act (“PDUFA”) goals, FDA’s goal for acting on the submission of an NDA for a new molecular entity is ten months from the date the FDA files the NDA. The FDA conducts a preliminary review of an NDA within 60 days after submission to determine whether it is sufficiently complete to permit substantive review, before determining whether to file the NDA. This two-month preliminary review effectively extends the typical NDA review period to twelve months. In rare cases, the FDA may request additional information rather than file an NDA. In this event, the NDA must be resubmitted with the additional information. The resubmitted application also is subject to review before the FDA accepts it for filing.
The FDA may also refer applications for novel pharmaceutical products, as well as pharmaceutical products that present difficult questions of safety or efficacy, to be reviewed by an advisory committee, typically a panel that includes clinicians, statisticians and other experts, for review, evaluation, and a recommendation as to whether the NDA should be approved. The FDA is not bound by the recommendation of an advisory committee, but generally follows such recommendations. Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the pharmaceutical product is manufactured. The FDA will not approve the product unless compliance with cGMP is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the respective claimed indication.
Following the FDA’s evaluation of an NDA, it will issue an approval letter or a complete response letter (“CRL”). An approval letter authorizes the sponsor to begin commercial marketing of the drug for specific indications. A CRL means that the review cycle of the application is complete and the application will not be approved in its present form. A CRL describes the specific deficiencies in the NDA identified by the FDA and may recommend actions that the applicant might take, including providing additional clinical data, such as an additional Phase 3 trial or other significant and time-consuming requirements related to clinical trials, nonclinical studies or manufacturing, to resolve the deficiencies. If a CRL is issued, the sponsor must resubmit the NDA addressing all of the deficiencies identified in the letter, or withdraw the application. Even if the sponsor submits the recommended data and information, the FDA may decide that the NDA does not satisfy the criteria for approval.
As condition to a product’s regulatory approval, the FDA may require a sponsor to conduct Phase 4 studies designed to further assess the drug’s safety and effectiveness after NDA approval, or may require other testing and surveillance programs to monitor the safety of the approved product. The FDA may also place other conditions on approval including the requirement for a risk evaluation and mitigation strategy (“REMS”) to assure the safe use of the drug. A REMS could include medication guides, communication plans to healthcare professionals or other elements to assure safe use, such as provider certification or training, restricted distribution methods, and patient registries.
There are a variety of regulations governing clinical trials and requirements for obtaining marketing approval for pharmaceutical products outside the United States. Whether or not FDA approval has been obtained, approval of a product by the comparable regulatory authorities of foreign countries and regions must be obtained prior to the commencement of marketing the product in those countries. The approval process varies from one regulatory authority to another and the time may be longer or shorter than that required for FDA approval. In the EU, Canada and Australia, regulatory requirements and approval processes are similar, in principle, to those in the United States.
14
Disclosure of Clinical Trial Information
Sponsors of clinical trials of FDA-regulated products, including drugs and biologic products, are required to register and disclose certain clinical trial information on the website www.clinicaltrials.gov. Information related to the product, patient population, phase of investigation, trial sites and investigators, and other aspects of a clinical trial are then made public as part of the registration. Sponsors are also obligated to disclose the results of their clinical trials after completion. Disclosure of the results of clinical trials can be delayed in certain circumstances for up to two years after the date of completion of the trial. Competitors may use this publicly available information to gain knowledge regarding the progress of clinical development programs as well as clinical trial design.
Pediatric Information
Under the Pediatric Research Equity Act (“PREA”), NDAs or supplements to NDAs must contain data to assess the safety and effectiveness of the drug product for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may grant full or partial waivers, or deferrals, for submission of data. Unless otherwise required by regulation, PREA does not apply to any product with orphan product designation except a product with a new active ingredient that is a molecularly targeted cancer product intended for the treatment of an adult cancer and directed at a molecular target determined by the FDA to be substantially relevant to the growth or progression of a pediatric cancer.
The Best Pharmaceuticals for Children Act (“BPCA”) provides a six-month extension of any patent or non-patent exclusivity for a drug if certain conditions are met. Conditions for exclusivity include the FDA’s determination that information relating to the use of a new drug in the pediatric population may produce health benefits in that population, the FDA making a written request for pediatric studies, and the applicant agreeing to perform, and reporting on, the requested studies within the statutory timeframe. Applications under the BPCA are treated as priority applications, with all of the benefits that designation confers.
Expedited Programs
The FDA is required to facilitate the development, and expedite the review, of drug products that are intended for the treatment of a serious or life-threatening disease or condition for which there is no effective treatment and which demonstrate the potential to address unmet medical needs for the condition. Fast track designation may be granted for products that are intended to treat a serious or life-threatening disease or condition for which there is no effective treatment and preclinical or clinical data demonstrate the potential to address unmet medical needs for the condition. Fast track designation applies to both the product and the specific indication for which it is being studied. Any product submitted to the FDA for marketing, including under a fast track program, may be eligible for other types of FDA programs intended to expedite development and review, such as priority review.
Priority review may be granted for products that are intended to treat a serious or life-threatening condition and, if approved, would provide a significant improvement in safety and effectiveness compared to available therapies. The FDA will attempt to direct additional resources to the evaluation of an application designated for priority review in an effort to facilitate the review.
The FDA is also required to expedite the development and review of applications for approval of products that are intended to treat a serious or life-threatening disease or condition where preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints. Under the breakthrough therapy program, the sponsor of a new product candidate may request that the FDA designate the product candidate for a specific indication as a breakthrough therapy concurrent with, or after, the submission of the IND for the product candidate. The FDA must determine if the product candidate qualifies for breakthrough therapy designation within 60 days of receipt of the sponsor’s request. The FDA may take certain actions with respect to breakthrough therapies, including holding meetings with the sponsor throughout the development process, providing timely advice to the product sponsor regarding development and approval, involving more senior staff in the review process, assigning a cross disciplinary project lead for the review team and taking other steps to design the clinical studies in an efficient manner.
Orphan Drug Designation
Pursuant to the Orphan Drug Act, the FDA may grant special status, or orphan designation, to a drug intended to treat a rare disease or condition, which is defined as a disease or condition that affects fewer than 200,000 individuals in the United States, or there is no reasonable expectation that the sales of the product will offset the cost of developing and making the drug available in the United States. A request for orphan drug designation must be submitted before the NDA is submitted. Following the grant of orphan designation, the FDA will publicly disclose the identity of the therapeutic drug candidate and its potential orphan use. Orphan designation does not shorten the duration of the regulatory review and approval process.
15
If a drug candidate with orphan designation subsequently receives the first FDA approval for the disease or condition for which it has orphan designation, the drug is entitled to a seven-year period of market exclusivity subject to certain exceptions (e.g., clinical superiority of a subsequent product). This means that the FDA may not approve another drug application authorizing another manufacturer to market the same drug for the same indication for seven years. This does not preclude competitors from receiving approval of the same product that has orphan exclusivity for a different indication or a different product for the same indication for which the orphan product has exclusivity. The orphan designation of a drug also provides the sponsor with certain financial incentives including tax credits and waiver of PDUFA fees.
The granting of an orphan drug designation does not shorten the duration of, the regulatory review and approval process nor does it guarantee regulatory approval. The first applicant of a new drug application, or NDA, to receive FDA, approval for a particular active ingredient to treat a particular disease with FDA orphan drug designation is entitled to a seven year exclusive marketing period in the United States for that product for that indication.
Rare Pediatric Disease Priority Review Voucher Program
Under the Rare Pediatric Disease Priority Review Voucher program, the FDA may award a priority review voucher to the sponsor of an approved marketing application for a product that treats or prevents a rare pediatric disease. The voucher entitles the sponsor to priority review of one subsequent marketing application.
A voucher may be awarded only for an approved rare pediatric disease product application. A rare pediatric disease product application is an NDA for a product that treats or prevents a serious or life-threatening disease in which the serious or life-threatening manifestations primarily affect individuals aged from birth to 18 years; in general, the disease must affect fewer than 200,000 such individuals in the U.S.; the NDA must be deemed eligible for priority review; the NDA must not seek approval for a different adult indication (i.e., for a different disease/condition); the product must not contain an active ingredient that has been previously approved by the FDA; and the NDA must rely on clinical data derived from studies examining a pediatric population such that the approved product can be adequately labeled for the pediatric population. Before NDA approval, the FDA may designate a product in development as a product for a rare pediatric disease, but such designation is not required to receive a voucher.
To receive a rare pediatric disease priority review voucher, a sponsor must notify the FDA, upon submission of the NDA, of its intent to request a voucher. If the FDA determines that the NDA is a rare pediatric disease product application and grants priority review, and if the NDA is approved, the FDA will award the sponsor of the NDA a voucher upon approval of the NDA. The FDA may revoke a rare pediatric disease priority review voucher if the product for which it was awarded is not marketed in the U.S. within 365 days of the product’s approval.
The voucher, which is transferable to another sponsor, may be submitted with a subsequent NDA or biologics license application (“BLA”) and entitles the holder to priority review of the accompanying NDA or BLA. The sponsor submitting the priority review voucher must notify the FDA of its intent to submit the voucher with the NDA or BLA at least 90 days prior to submission of the NDA or BLA and must pay a priority review user fee in addition to any other required user fee. The FDA must take action on an NDA or BLA under priority review within six months of receipt of the NDA or BLA.
The Rare Pediatric Disease Priority Review Voucher program was reauthorized in the Creating Hope Reauthorization Act in December 2020. Under the current statutory sunset provisions, after December 20, 2024, the FDA may only award a priority review voucher for an approved rare pediatric disease application if the sponsor has rare pediatric disease designation for the drug that is the subject of such application, and that designation was granted by December 20, 2024. After September 30, 2026, the FDA may not award any rare pediatric disease priority review vouchers, unless the program is extended. Although legislation to extend the rare pediatric disease priority review voucher program has been proposed, Congress has not yet, and may never, pass a bill to reauthorize the program and extend the sunset dates.
Post-Marketing Obligations
All approved drug products are subject to continuing regulation by the FDA, including record-keeping requirements, reporting of adverse experiences with the product, sampling and distribution requirements, notifying the FDA and gaining approval for certain manufacturing or labeling changes, complying with certain electronic records and signature requirements, submitting periodic reports to the FDA, maintaining and providing updated safety and efficacy information to the FDA, and complying with FDA promotion and advertising requirements. Failure to comply with the statutory and regulatory requirements can subject a manufacturer to possible legal or regulatory action, such as warning letters, suspension of manufacturing, seizure of product, injunctive action, criminal prosecution, or civil penalties.
16
The FDA may require post-marketing studies or clinical trials to develop additional information regarding the safety of a product. These studies or trials may involve continued testing of a product and development of data, including clinical data, about the product’s effects in various populations and any side-effects associated with long-term use. The FDA may require post-marketing studies or trials to investigate known serious risks or signals of serious risks or identify unexpected serious risks and may require periodic status reports if new safety information develops. Failure to conduct these studies in a timely manner may result in substantial civil fines.
Drug manufacturers and their subcontractors are required to register their establishments with the FDA and certain state agencies, and to list their products with the FDA. The FDA periodically inspects manufacturing facilities in the United States and abroad in order to assure compliance with the applicable cGMP regulations and other requirements. Facilities also are subject to inspections by other federal, foreign, state or local agencies. In complying with the cGMP regulations, manufacturers must continue to assure that the product meets applicable specifications, regulations and other post-marketing requirements. Any third-party manufacturers must also maintain compliance with all applicable regulations and requirements. Failure to comply with these requirements subjects the manufacturer to possible legal or regulatory action, such as suspension of manufacturing or recall or seizure of product.
Also, newly discovered or developed safety or efficacy data may require changes to a product’s approved labeling, including the addition of new warnings and contraindications, additional pre-clinical or clinical studies, or even in some instances, revocation or withdrawal of the approval. Violations of regulatory requirements at any stage, including after approval, may result in various adverse consequences, including the FDA’s withdrawal of an approved product from the market, other voluntary or FDA-initiated action that could delay or restrict further marketing, and the imposition of civil fines and criminal penalties against the NDA holder. In addition, later discovery of previously unknown problems may result in restrictions on the product or NDA holder, including withdrawal of the product from the market. Furthermore, new government requirements may be established that could delay or prevent regulatory approval of our products under development, or affect the conditions under which approved products are marketed.
Data Privacy
We are subject to various laws and regulations globally regarding privacy and data protection, including laws and regulations relating to the collection, storage, handling, use, disclosure, transfer and security of personal information. The legislative and regulatory environment regarding privacy and data protection is continuously evolving and developing and the subject of significant attention globally. Certain privacy and data protection laws, such as the Health Insurance Portability and Accountability Act (HIPAA) and the California Consumer Privacy Act (CCPA), may not apply to us directly at this time, but those laws may apply to the investigators, health care professionals, third party payors, and business partners with whom we have relationships and so may apply to our processing of personal information that we receive from or share with such third parties. We may also engage service providers, such as contract research organizations, to process personal information on our behalf. We cannot ensure that all our contractors, vendors, licensees, business partners or collaborators will comply with all applicable privacy and data protection laws and regulations. The failure to comply with these current and future laws could result in significant penalties and reputational harm and could have a material adverse effect on our business and results of operations.
Commercial Product Pricing
In the United States and some foreign jurisdictions, many of the markets in which we may do business in the future, the prices of pharmaceutical products are subject to direct price controls (by law) and to drug reimbursement programs with varying price control mechanisms.
In the United States, the Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or Medicare Modernization Act, changed the way Medicare covers and pays for pharmaceutical products. The legislation expanded Medicare coverage for drug purchases by the elderly and introduced a new reimbursement methodology based on average sales prices for physician administered drugs. In addition, this legislation provided authority for limiting the number of drugs that will be covered in any therapeutic class in certain cases. Cost reduction initiatives and other provisions of this and other more recent legislation could decrease the coverage and reimbursement that is provided for any approved products. While the Medicare Modernization Act applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own reimbursement rates. Therefore, any reduction in reimbursement that results from the Medicare Modernization Act or other more recent legislation may result in a similar reduction in payments from private payors.
17
Healthcare Reform
Healthcare reforms that have been adopted, and that may be adopted in the future, could result in further reductions in coverage and levels of reimbursement for pharmaceutical products, increases in rebates payable under U.S. government rebate programs and additional downward pressure on pharmaceutical product prices. Recently, healthcare reform initiatives culminated in the enactment of the Inflation Reduction Act (“IRA”) in August 2022, which will, among other things, allow U.S. Department of Health and Human Services (“HHS”) to negotiate the selling price of certain drugs and biologics that the Centers for Medicare & Medicaid Services (“CMS”) reimburses under Medicare Part B and Part D, although only high-expenditure single-source drugs that have been approved for at least 7 years (11 years for biologics) can be selected by CMS for negotiation, with the negotiated price taking effect two years after the selection year. The negotiated prices, which will first become effective in 2026, will be capped at a statutory ceiling price. Beginning in October 2023, the IRA will also penalize drug manufacturers that increase prices of Medicare Part B and Part D drugs at a rate greater than the rate of inflation. The IRA permits the Secretary of HHS to implement many of these provisions through guidance, as opposed to regulation, for the initial years. Manufacturers that fail to comply with the IRA may be subject to various penalties, including civil monetary penalties. The IRA also extends enhanced subsidies for individuals purchasing health insurance coverage in ACA marketplaces through plan year 2025. These provisions will take effect progressively starting in 2023, although they may be subject to legal challenges. It is unclear to what extent additional statutory, regulatory, and administrative initiatives will be enacted and implemented.
European Regulatory Authorities
In the European Union, governments influence the price of pharmaceutical products through their pricing and reimbursement rules and control of national healthcare systems that fund a large part of the cost of such products to consumers. The approach taken varies from member state to member state. Some jurisdictions operate positive and/or negative list systems under which products may be marketed only once a reimbursement price has been agreed. Other member states allow companies to fix their own prices for medicines, but monitor and control company profits. The downward pressure on healthcare costs in general, particularly prescription drugs, has become very intense. As a result, increasingly high barriers are being erected to the entry of new products, as exemplified by the role of the National Institute for Health and Clinical Excellence in the United Kingdom, which evaluates the data supporting new medicines and passes reimbursement recommendations to the government. In addition, in some countries cross-border imports from low-priced markets (parallel imports) exert commercial pressure on pricing within a country.
Environmental and Safety Laws
We do not use, handle, store, or dispose of hazardous materials and our operations do not produce hazardous waste. Accordingly, we are not subject to federal, state and local regulations relating to the use, handling, storage and disposal of hazardous materials. Any waste generated is non-hazardous and is disposed of by third party contractors. Likewise, given that we have less than 10 employees, we are not subject to the recordkeeping requirements under the Occupational Safety and Health Administration (“OSHA”) although other OSHA regulations may apply. OSHA and/or the Environmental Protection Agency may promulgate regulations that may affect our research and development programs.
We are also subject to various laws and regulations governing laboratory practices and the experimental use of animals.
Human Capital
As of December 31, 2024, we had three full-time employees and two part-time employees. Our employees are not represented by any collective bargaining agreements, and we have never experienced an organized work stoppage.
Enforceability of Civil Liabilities
To the extent any of our shareholders may seek to enforce a U.S. judgment in Israel against us or our executive officers and directors, or to assert U.S. securities law claims in Israel, shareholders may have difficulties enforcing such a U.S. judgment, including judgments based upon the civil liability provisions of the U.S. federal securities laws, in Israel.
We have been informed by our legal counsel in Israel that it may be difficult to assert U.S. securities laws claims in original actions instituted in Israel. Israeli courts may refuse to hear a claim based on a violation of U.S. securities laws because Israel is not the most appropriate forum in which to bring such a claim. In addition, even if an Israeli court agrees to hear a claim, it may determine that Israeli law and not U.S. law is applicable to the claim. If U.S. law is found to be applicable, the content of applicable U.S. law must be proven as a fact which can be a time-consuming and costly process. Matters of procedure will also be governed by Israeli law.
18
We have irrevocably appointed Quoin Pharmaceuticals, Inc., as our agent to receive service of process in any action against us in any U.S. federal or state court arising out of this offering or any purchase or sale of securities in connection with this offering. Subject to specified time limitations and legal procedures, Israeli courts may enforce a U.S. judgment in a civil matter which is non-appealable, including a judgment based upon the civil liability provisions of the Securities Act or the Exchange Act and including a monetary or compensatory judgment in a non-civil matter, provided that, among other things:
| ● | the judgment was rendered by a court of competent jurisdiction, according to the laws of the state in which the judgment is given; |
| ● | the judgment is enforceable according to the laws of Israel and according to the law of the foreign state in which the relief was granted; and |
| ● | the judgment is not contrary to public policy of Israel. |
Even if such conditions are met, an Israeli court may not declare a foreign civil judgment enforceable if:
| ● | the prevailing law of the foreign state in which the judgment is rendered does not allow for the enforcement of judgments of Israeli courts (subject to exceptional cases); |
| ● | the defendant did not have a reasonable opportunity to be heard and to present his or her evidence, in the opinion of the Israeli court; |
| ● | the enforcement of the civil liabilities set forth in the judgment is likely to impair the security or sovereignty of Israel; |
| ● | the judgment was obtained by fraud; |
| ● | the judgment was rendered by a court not competent to render it according to the rules of private international law prevailing in Israel; |
| ● | the judgment conflicts with any other valid judgment in the same matter between the same parties; or |
| ● | an action between the same parties in the same matter was pending in any Israeli court or tribunal at the time at which the lawsuit was instituted in the foreign court. |
If a foreign judgment is enforced by an Israeli court, it generally will be payable in Israeli currency, which can then be converted into non-Israeli currency and transferred out of Israel. The usual practice in an action before an Israeli court to recover an amount in a non-Israeli currency is for the Israeli court to issue a judgment for the equivalent amount in Israeli currency at the rate of exchange in force on the date of the judgment, but the judgment debtor may make payment in foreign currency. Pending collection, the amount of the judgment of an Israeli court stated in Israeli currency ordinarily will be linked to the Israeli consumer price index plus interest at the annual statutory rate set by Israeli regulations prevailing at the time. Judgment creditors must bear the risk of unfavorable exchange rates.
Company Information
We were incorporated under the laws of the State of Israel in 1986 under the name Montiger Ltd. Between 1986 and 2021, we underwent several name changes, including the name change to Cellect Biotechnology Ltd. (“Cellect”). On October 28, 2021, Cellect completed the business combination with Quoin Pharmaceuticals, Inc., a Delaware corporation (“Quoin Inc.”), in accordance with the terms of the Agreement and Plan of Merger and Reorganization, dated as of March 24, 2021 (the “Merger Agreement”), by and among Cellect, Quoin Inc. and CellMSC, Inc., a Delaware corporation and wholly-owned subsidiary of Cellect (“Merger Sub”), pursuant to which Merger Sub merged with and into Quoin Inc., with Quoin Inc. surviving as a wholly-owned subsidiary of Cellect (the “Merger”). Immediately after completion of the Merger, Cellect changed its name to “Quoin Pharmaceuticals Ltd.” In addition, on October 28, 2021, Cellect sold the entire share capital of its subsidiary, Cellect Biotherapeutics Ltd., which essentially included all of Cellect’s then existing net assets, to EnCellX Inc. (“EnCellX”), a newly formed U.S. privately held company. We have no interests in EnCellX subsequent to the closing of the Merger.
19
Prior to January 1, 2023, we qualified as a “foreign private issuer” as such term is defined in Rule 405 under the Securities Act. Since January 1, 2023, we have been obligated to file or furnish reports, proxy statements, and other information on U.S. domestic issuer forms with the Securities and Exchange Commission (the “SEC”), which are more detailed and extensive in certain respects, and which must be filed more promptly, than the forms available to a foreign private issuer.
The address of our executive corporate offices is 42127 Pleasant Forest Ct., Ashburn, VA 20148, and our telephone number is (703) 980-4182. Our website is www.quoinpharma.com. Information contained on or accessible through this website is not incorporated by reference in, or otherwise a part of, this Annual Report, and any references to this website are intended to be inactive textual references only.
Available Information
We are subject to the informational requirements of the Exchange Act and in accordance therewith, we file reports, proxy and information statements and other information with the SEC. You can read our SEC filings over the Internet at the SEC’s website at www.sec.gov.Our filings with the SEC are also available free of charge on the investors section of our website at www.quoinpharma.com. Our filings are available free of charge as soon as reasonably practicable after we electronically file them with, or furnish them to, the SEC. From time to time, we also use multiple social media channels to communicate with the public about Quoin and its products. It is possible that the information we post on social media could be deemed to be material information. Therefore, we encourage you to review the information we post on such social media channels as our LinkedIn page (https://www.linkedin.com/company/quoin-pharmaceuticals/) and our Twitter account (@Quoinpharma). This list may be updated from time to time on our investor relations website.
Information contained on or accessible through the websites and social media channels referred to above is not incorporated by reference in, or otherwise a part of, this Annual Report, and any references to these websites and social media channels are intended to be inactive textual references only.
Smaller Reporting Company
We are a “smaller reporting company” as defined in the Securities Exchange Act of 1934, as amended (the “Exchange Act”). As a result, we may take advantage of certain reduced disclosure obligations available to smaller reporting companies, including the exemption from compliance with the auditor attestation requirements pursuant to the Sarbanes-Oxley Act of 2022, reduced disclosure about our executive compensation arrangements and the requirements to provide only two years of audited financial statements in our annual reports and registration statements. We will continue to be a “smaller reporting company” as long as (1) we have a public float (i.e., the market value of our ADSs held by non-affiliates) less than $250 million calculated as of the last business day of our most recently completed second fiscal quarter, or (2) our annual revenues are less than $100 million for our previous fiscal year and we have either no public float or a public float of less than $700 million as of the end of that fiscal year’s second fiscal quarter. Decreased disclosures in our SEC filings due to our status as a “smaller reporting company” may make it harder for investors to analyze our results of operations and financial prospects.
20
Item 1A. Risk Factors
Investing in our securities involves a high degree of risk. You should carefully consider the risk factors discussed below as well as other information we include in this Annual Report, including our consolidated financial statements and the related notes and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” If any of the following risks occur, our business, financial condition, results of operations and prospects could be materially and adversely affected. In that case, the market price of our securities could decline and you could lose all or part of your investment. Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also materially harm our business, operating results and financial condition and could result in a complete loss of your investment. This Annual Report also contains forward-looking statements that involve risks and uncertainties. Our results could materially differ from those anticipated in these forward-looking statements as a result of certain factors including the risks described below and elsewhere in this Annual Report and our other SEC filings. For a summary of the risk factors included in this Item 1A and for further details on our forward-looking statements, see “Cautionary Note Regarding Forward-Looking Statements and Summary of Risk Factors” on page 1.
Risks Related to Our Financial Position and Capital Requirements
We have a limited operating history that you can use to evaluate us, and the likelihood of our success must be considered in light of the problems, expenses, difficulties, complications and delays frequently encountered by a small developing company.
Our wholly owned subsidiary, Quoin Inc., commenced operations in 2018. As such, we have a limited operating history and our operations are subject to all of the risks inherent in the establishment of a new business enterprise, including a lack of operating history. Since inception, our operations have been primarily limited to acquiring and licensing intellectual property rights, undertaking research and conducting preclinical and clinical studies for our initial programs and negotiating and executing the Merger and financings. We have not yet obtained regulatory approval for any product candidates. Consequently, any predictions about our future success or viability, or any evaluation of our business and prospects, may not be accurate. The likelihood of our success must be considered in light of the problems, expenses, difficulties, complications and delays frequently encountered by a small developing company starting a new business enterprise and the highly competitive environment in which we will operate. Since we have a limited operating history, we cannot assure you that our business will be profitable or that we will ever generate sufficient revenues to meet our expenses and support our anticipated activities. In addition, there is no guarantee that any of our product candidates with ever receive approval from the U.S. Food and Drug Administration, or the “FDA.” We cannot be certain that our business strategy will be successful or that we will be solvent at any particular time. Our likelihood of success must be considered in light of the problems, expenses, difficulties, complications and delays frequently encountered in connection with the early stages of the development of any company. If we fail to address any of these risks or difficulties adequately, our business will likely suffer. Because of the numerous risks and uncertainties associated with developing and commercializing our products, we are unable to predict the extent of any future losses or when we will become profitable, if ever. We may never become profitable and investors may never receive a return on an investment in our securities. An investor in our securities must carefully consider the substantial challenges, risks and uncertainties inherent in the attempted development and commercialization of products in the medical and pharmaceutical industries. We may never successfully commercialize our products and our business may fail.
We have incurred significant losses since our inception and have limited cash available for our operations.
To date, we have not commercialized any products and have not generated any revenue. We have incurred net losses every year since inception and we have an accumulated deficit of approximately $55.2 million at December 31, 2024. We have historically funded our operations through debt and equity financings. At December 31, 2024, we had cash balances totaling $3.6 million and investments of $10.4 million. We believe that we have sufficient cash for operating our business for at least the next twelve months from the date of filing this Form 10-K. However, we are subject to risks common to late-stage clinical specialty pharmaceutical companies including, but not limited to, unanticipated or higher than expected clinical trial costs and the ability to estimate such occurrences, if any, on our cash, liquidity, additional financing requirements, and availability. Accordingly, we may need to raise additional funds during this period. We have devoted a majority of our financial resources to research and development, including our preclinical and ongoing clinical development activities. To date, we have funded our operations primarily through our founders’ funding expenditures and the sale of equity and convertible securities.
We do not expect to generate revenue from product sales unless and until we successfully complete development and obtain marketing approval for one or more of our product candidates or these candidates participate in Early Access or Named Patient programs, which we expect will take a number of years and is subject to significant uncertainty. Additional financing will be required to complete the research and development of our product candidates and our other operating requirements, which may not be available at acceptable
21
terms, if at all. If we are unable to obtain additional funding when it becomes necessary, the development of our product candidates will be impacted and we would likely be forced to delay, reduce, or terminate some or all of our development programs, all of which could have a material adverse effect on our business, results of operations and financial condition.
We expect to continue to incur significant expenses and increasing operating losses for the foreseeable future. The net losses we incur may fluctuate significantly from quarter to quarter. We anticipate that our expenses will increase substantially if and as we:
| ● | continue and/or initiate clinical development of our product candidates, including—QRX003—a topical lotion comprised of a broad-spectrum serine protease inhibitor, formulated with the proprietary Invisicare® technology, which is under clinical development as a potential treatment for Netherton Syndrome (“NS”); |
| ● | further enhance our internal control systems; |
| ● | initiate the development of additional product candidates for other rare disease indications; |
| ● | acquire or in-license other products and technologies and advance those product candidates into clinical trials; |
| ● | seek marketing approvals for our product candidates that successfully complete clinical trials; |
| ● | ultimately establish a sales, marketing and distribution infrastructure to commercialize any products for which we may obtain marketing approval; |
| ● | maintain, expand and protect our intellectual property portfolio; |
| ● | hire additional clinical, regulatory, research, executive and administrative personnel; and |
| ● | create additional infrastructure to support our operations and our product development and planned future commercialization efforts. |
We have never generated any revenue from product sales or any other sources since inception, and may never be profitable.
Our ability to generate revenue and achieve profitability depends on our ability, alone or with strategic alliance partners, to successfully complete the development of, obtain the necessary regulatory approvals for and commercialize our product candidates. We do not anticipate generating revenues from sales of our products until regulatory approval has been obtained, if ever. Our ability to generate future revenues from product sales depends heavily on our success in:
| ● | completing our research and preclinical development of product candidates; |
| ● | initiating and completing clinical trials for product candidates with favorable results; |
| ● | seeking, obtaining, and maintaining marketing approvals for product candidates that successfully complete clinical trials; |
| ● | establishing and maintaining supply and manufacturing relationships with third parties; |
| ● | launching and commercializing product candidates for which we may obtain marketing approval, with an alliance partner or, if launched independently, successfully establishing a sales force, marketing and distribution infrastructure; |
| ● | maintaining, protecting and expanding our intellectual property portfolio; and |
| ● | attracting, hiring and retaining qualified personnel. |
22
Because of the numerous risks and uncertainties associated with pharmaceutical product development, we are unable to predict the timing or amount of increased expenses and when we will be able to achieve or maintain profitability, if ever. In addition, our expenses could increase beyond expectations if we are required by the FDA or other foreign regulatory agencies to perform studies and trials in addition to those that we currently anticipate.
Even if one or more of the product candidates that we independently develop is approved for commercial sale, we may incur significant costs associated with commercializing any approved product. Even if we are able to generate revenues from the sale of any approved products, we may not become profitable and may need to obtain additional funding to continue operations.
We expect that we will need to raise additional capital, which may not be available on acceptable terms, or at all.
Developing pharmaceutical products, including conducting preclinical studies and clinical trials, is expensive. We expect our research and development expenses to substantially increase in connection with our ongoing activities, particularly as we advance our product candidates towards or through clinical trials. We may need to raise additional capital to support our operations and such funding may not be available to us on acceptable terms, or at all. We cannot provide assurances that our plans will not change or that changed circumstances will not result in the depletion of our capital resources more rapidly than we currently anticipate. For example, our preclinical or clinical trials may encounter technical difficulties or be subject to delays or other issues. Any of these events may increase our development costs more than we expect. In order to support our long-term plans, we may need to raise additional capital or otherwise obtain funding through additional strategic alliances if we choose to initiate preclinical or clinical trials for new product candidates other than programs currently partnered. In any event, we will require additional capital to obtain regulatory approval for, and to commercialize, future product candidates.
Any additional fundraising efforts may divert our management from our day-to-day activities, which may adversely affect our ability to develop and commercialize future product candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. If we are unable to raise additional capital when required or on acceptable terms, we may be required to:
| ● | significantly delay, scale back or discontinue the development or commercialization of any future product candidates; |
| ● | seek strategic alliances for research and development programs at an earlier stage than otherwise would be desirable or on terms that are less favorable than might otherwise be available; or |
| ● | relinquish or license on unfavorable terms, our rights to technologies or any future product candidates that we otherwise would seek to develop or commercialize ourselves. |
If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we will be prevented from pursuing development and commercialization efforts, which will have a material adverse effect on our business, operating results and prospects.
Risks Related to the Discovery and Development of Product Candidates
Preclinical and clinical studies of our product candidates may not be successful. If we are unable to generate successful results from preclinical and clinical studies of our product candidates, or experience significant delays in doing so, our business may be materially harmed.
We have no products approved for commercial marketing and most of our product candidates are in preclinical and clinical development as is the case with our lead asset for NS, which is currently being tested in four separate clinical studies in NS patients. Moreover, the clinical development process can take several years, and there is no assurance that our clinical trials will be successful or that we will obtain marketing approvals for any of our product candidates from either the FDA or the EMA. Our ability to achieve and sustain profitability depends on obtaining regulatory approvals for and, if approved, successfully commercializing our product candidates, either alone or with third parties. Before obtaining regulatory approval for the commercial distribution of our product candidates, we or an existing or future collaborator must conduct extensive preclinical tests and clinical trials to demonstrate the safety and efficacy of our product candidates.
23
The success of our product candidates will depend on several factors, including the following:
| ● | successfully implementing preclinical studies which may be predictive of clinical outcomes; |
| ● | successful enrollment in clinical trials and completion of those trials with favorable results; |
| ● | receipt of marketing approvals from applicable regulatory authorities; |
| ● | obtaining and maintaining patent and trade secret protection for current and future product candidates; |
| ● | establishing and maintaining manufacturing relationships with third parties or establishing our own manufacturing capability; and |
| ● | successfully commercializing our products, if approved, including successfully establishing a sales force, marketing and distribution infrastructure, whether alone or in collaboration with others. |
If we do not achieve one or more of these factors in a timely manner or at all, we could experience significant delays or an inability to successfully complete the development or commercialization of our product candidates, which would materially harm our business.
We may not be successful in our efforts to identify or develop potential product candidates.
The success of our business depends primarily upon our ability to identify, develop and commercialize our product candidates. Our research programs may initially show promise in identifying potential product candidates, yet fail to yield product candidates for clinical development for a number of reasons, including:
| ● | our research methodology may be unsuccessful in identifying potential product candidates; or |
| ● | potential product candidates may be shown to have harmful side effects or may have other characteristics that may make the products unsuitable for administration in patients in clinical trials, unlikely to receive marketing approval or unmarketable. |
If any of these events occur, we may be forced to abandon our development efforts for a program or programs, which would have a material adverse effect on our business and could potentially cause us to cease operations. Research programs to identify new product candidates require substantial technical, financial and human resources. We may focus our efforts and resources on potential programs or product candidates that ultimately prove to be unsuccessful.
If clinical trials of our product candidates fail to demonstrate safety and efficacy to the satisfaction of regulatory authorities or do not otherwise produce positive results, we may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our product candidates.
Before obtaining marketing approval from regulatory authorities for the sale of product candidates, we must conduct extensive clinical trials to demonstrate the safety and efficacy of the product candidates in humans. Clinical trials are expensive, difficult to design and implement, can take many years to complete and are uncertain as to the outcome. A failure of one or more clinical trials can occur at any stage of testing. The outcome of preclinical studies and early clinical trials may not be predictive of the success of later clinical trials, and preliminary results or planned interim analyses of a clinical trial do not necessarily predict final results. Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses, and many companies that have believed their product candidates performed satisfactorily in preclinical studies and clinical trials have nonetheless failed to obtain marketing approval for their products.
Events which may result in a delay or unsuccessful completion of clinical development include:
| ● | delays in reaching an agreement with the FDA or other regulatory authorities on final trial design, including selection of dose and clinical outcome assessments and related efficacy endpoints |
| ● | delays in obtaining from the FDA, or comparable foreign regulatory authority, authorization to administer an investigational new drug product to humans through the submission or acceptance of an IND or similar foreign application; |
24
| ● | imposition of a clinical hold of clinical trial operations or trial sites by the FDA or other regulatory authorities; |
| ● | delays in reaching agreement on acceptable terms with prospective contract research organizations (“CROs”) and clinical trial sites; |
| ● | our inability to adhere to clinical trial requirements directly or with third parties such as CROs; |
| ● | clinical trial site or CRO non-compliance with good clinical practices (“GCPs”), good laboratory practices, or other regulatory requirements; |
| ● | inability or failure of clinical trial sites to adhere to the clinical trial protocol; |
| ● | delays in obtaining required IRB approval at each clinical trial site, or an IRB reversing such approval resulting in the suspension or termination of a trial at that site; |
| ● | delays in recruiting and retaining suitable patients to participate in a trial particularly for a rare disease such as NS; |
| ● | delays in the testing, validation, manufacturing and delivery of the product candidates to the clinical sites; |
| ● | delays in having patients complete participation in a trial or return for post-treatment follow-up; |
| ● | delays caused by patients dropping out of a trial due to protocol procedures or requirements, product side effects or disease progression; |
| ● | clinical sites dropping out of a trial to the detriment of enrollment; |
| ● | time required to add new clinical sites; or |
| ● | delays by our contract manufacturers to produce and deliver sufficient supply of clinical trial materials. |
Accordingly, we cannot be sure that we will submit INDs on our expected timelines and we cannot be certain the FDA or foreign regulatory agencies such as the EMA, will allow us to progress into clinical trials based on the submission of any IND.
If we are required to conduct additional clinical trials or other testing of any product candidates beyond those that are currently contemplated, are unable to successfully complete clinical trials of any such product candidates or other testing, or if the results of these trials or tests are not positive, are only modestly positive or if there are safety concerns, we may:
| ● | be delayed in obtaining marketing approval for our future product candidates; |
| ● | not obtain marketing approval at all; |
| ● | obtain approval for indications or patient populations that are not as broad as originally intended or desired; |
| ● | obtain approval with labeling that includes significant use or distribution restrictions or safety warnings; |
| ● | be subject to additional post-marketing testing requirements; or |
| ● | have the product removed from the market after obtaining marketing approval. |
Our product development costs will also increase if we experience delays in testing or marketing approvals. We do not know whether any clinical trials will begin as planned, will need to be restructured or will be completed on schedule, or at all. Significant clinical trial delays also could shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before we do, which would impair our ability to successfully
25
commercialize our product candidates and may harm our business and results of operations. Any inability to successfully complete preclinical and clinical development could result in additional costs to us or impair our ability to generate revenues from product sales.
Any of our product candidates may cause undesirable side effects or have other properties impacting safety that could delay or prevent their regulatory approval or limit the scope of any approved label or market acceptance.
Undesirable side effects caused by our product candidates could cause us or regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval by the FDA or other regulatory authorities. Results of our clinical trials could reveal a high and unacceptable severity level and prevalence of side effects. In such an event, our trials could be suspended or terminated and the FDA or comparable foreign regulatory authorities could order us to cease further development of or deny approval of our product candidates for any or all targeted indications. Such side effects could also affect patient recruitment, the ability of enrolled patients to complete the trial or result in potential product liability claims. Any of these occurrences may materially and adversely affect our business, financial condition, results of operations and prospects.
Further, clinical trials by their nature test product candidates in only small samples of the potential patient populations. With a limited number of patients and limited duration of exposure in such trials, rare and potentially severe side effects of our product candidates may not be uncovered until a significantly larger number of patients are exposed to the product candidate.
If any of our product candidates receive marketing approval, and causes serious, unexpected, or undesired side effects, a number of potentially significant negative consequences could result, including:
| ● | regulatory authorities may withdraw, suspend, or limit their approval of the product or impose restrictions on its distribution in the form of a modified risk evaluation and mitigation strategy; |
| ● | regulatory authorities may require the addition of labeling statements, such as black box warnings or contraindications; |
| ● | we may be required to change the way the product is administered or conduct additional clinical trials or post-marketing surveillance; |
| ● | we could be sued and held liable for harm caused to patients; or |
| ● | our reputation may suffer. |
Any of these events could prevent us from achieving or maintaining market acceptance of the affected product and could substantially increase the costs of commercializing our future products and impair our ability to generate revenues from the commercialization of these products.
Even if we complete the necessary preclinical studies and clinical trials, we cannot predict whether or when we will obtain regulatory approval to commercialize a product candidate and we cannot, therefore, predict the timing of any revenue from a future product.
We cannot commercialize a product until the appropriate regulatory authorities, such as the FDA, have reviewed and approved the product candidate. The regulatory authorities may not complete their review processes in a timely manner, or we may not be able to obtain regulatory approval for many reasons including:
| ● | regulatory authorities disagreeing with the design or implementation of our clinical trials; |
| ● | such authorities may disagree with our interpretation of data from preclinical studies or clinical trials; |
| ● | such authorities may not accept clinical data from trials which are conducted at clinical facilities or in countries where the standard of care is potentially different from that of the United States; |
| ● | unfavorable or unclear results from our clinical trials or results that may not meet the level of statistical significance required by the FDA or comparable foreign regulatory agencies for approval; |
26
| ● | serious and unexpected drug-related side effects experienced by participants in our clinical trials or by individuals using drugs similar to our product candidates; |
| ● | the population studied in the clinical trial may not be sufficiently broad or representative to assure safety in the full population for which we seek approval; |
| ● | we may be unable to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks; |
| ● | such authorities may not agree that the data collected from clinical trials of our product candidates are acceptable or sufficient to support the submission of a New Drug Application (“NDA”) or other submission or to obtain regulatory approval in the United States or elsewhere, and such authorities may impose requirements for additional preclinical studies or clinical trials; |
| ● | such authorities may disagree regarding the formulation, labeling and/or the specifications of our product candidates; |
| ● | such authorities may find deficiencies in the manufacturing processes, testing systems or facilities of our third-party manufacturers with which we contract for clinical and commercial supplies; or |
| ● | regulations of such authorities may significantly change in a manner rendering our clinical data insufficient for approval. |
Additional delays may result if an FDA advisory committee recommends restrictions on approval or recommends non-approval. In addition, we may experience delays or rejections based upon additional government regulation from future legislation or administrative action, or changes in regulatory agency policy during the period of product development, clinical trials and the review process.
Even if we obtain regulatory approval for a product candidate, we will still face extensive regulatory requirements and our products may face future development and regulatory challenges.
Even if we obtain regulatory approval in the United States, the FDA may still impose significant restrictions on the indicated uses or marketing of our product candidates, or impose ongoing requirements for potentially costly post-approval studies or post-market surveillance. The FDA may also require risk evaluation and mitigation strategies as a condition of approval of our product candidates, which could include requirements for a medication guide, physician communication plans or additional elements to ensure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. Additionally, the manufacturing processes, packaging, distribution, adverse event reporting, labeling, advertising, promotion, and recordkeeping for the product will be subject to extensive and ongoing FDA regulatory requirements, in addition to other potentially applicable federal and state laws. These requirements include monitoring and reporting of adverse events (“AEs”) and other post-marketing information and reports, registration, as well as continued compliance with current good manufacturing practice (“cGMP”) regulations. The holder of an approved NDA must also submit new or supplemental applications and obtain FDA approval for certain changes to the approved product, product labeling or manufacturing process. If we or a regulatory agency discovers previously unknown problems with a product such as AEs of unanticipated severity or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictions relative to that product or the manufacturing facility, including requiring recall or withdrawal of the product from the market or suspension of manufacturing.
If we fail to comply with applicable regulatory requirements following approval of any of our product candidates, a regulatory agency may:
| ● | issue a warning or untitled letter asserting that we are in violation of the law; |
| ● | seek an injunction or impose civil or criminal penalties or monetary fines; |
| ● | suspend or withdraw regulatory approval; |
| ● | suspend any ongoing clinical trials; |
| ● | refuse to approve a pending NDA or supplements to an NDA submitted by us; |
27
| ● | seize product or require a product recall; or |
| ● | refuse to allow us to enter into supply contracts, including government contracts. |
Any government investigation of alleged violations of law could require us to expend significant time and resources in response and could generate negative publicity. The occurrence of any event or penalty described above may inhibit our ability to commercialize our future products, if approved, and generate revenues.
We may not be able to obtain or maintain orphan drug designation or exclusivity for our product candidates.
Regulatory authorities in some jurisdictions, including the United States, may designate drugs for relatively small patient populations as orphan drugs. Under the Orphan Drug Act, the FDA may designate a drug as an orphan drug if it is intended to treat a rare disease or condition, which is generally defined as a patient population of fewer than 200,000 individuals in the United States, or if the disease or condition affects more than 200,000 individuals in the United States and there is no reasonable expectation that the cost of developing the drug for the type of disease or condition will be recovered from sales of the product in the United States.
Orphan drug designation entitles a party to financial incentives, such as tax advantages and user fee waivers. Additionally, if a product that has orphan designation subsequently receives the first FDA approval for the disease or condition for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications to market the same drug for the same indication for seven years, except in certain circumstances, such as a showing of clinical superiority (i.e., another product is safer, more effective or makes a major contribution to patient care) over the product with orphan exclusivity or where the manufacturer is unable to assure sufficient product quantity. Competitors, however, may receive approval of different products for the same indication for which the orphan product has exclusivity, or obtain approval for the same product but for a different indication than that for which the orphan product has exclusivity.
We intend to apply for orphan drug designation in the United States for QRX003 for the treatment of NS. However, obtaining an orphan drug designation can be difficult, and we may not be successful in doing so. Even if we obtain orphan drug designation for a product candidate in specific indications, we may not be the first to obtain regulatory approval of the product candidate for the orphan-designated indication. In addition, exclusive marketing rights in the United States may be limited if we seek approval for an indication broader than the orphan-designated indication or may be lost if the FDA later determines that the request for orphan designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition. Orphan drug designation does not ensure that we will receive marketing exclusivity in a particular market, and we cannot assure you that any future application for orphan drug designation in any other geography or with respect to any other future product candidate will be granted. Orphan drug designation neither shortens the development time or regulatory review time of a drug, nor gives the drug any advantage in the regulatory review or approval process.
We may pursue Rare Pediatric Disease designation for QRX003 for the treatment of NS or other of our product candidates. There is no assurance that we will obtain such designation. Moreover, a Rare Pediatric Disease designation by the FDA does not guarantee that the NDA for the product will qualify for a priority review voucher upon approval, and it does not lead to a faster development or regulatory review process, or increase the likelihood that any of our product candidates will receive marketing approval.
Under the Rare Pediatric Disease Priority Review Voucher program, upon the approval of a qualifying NDA for the treatment of a rare pediatric disease, the sponsor of such an application may be awarded a transferable rare pediatric disease priority review voucher that can be used to obtain priority review for a subsequent NDA or BLA. We intend to pursue Rare Pediatric Disease designation for QRX003 for the treatment of NS, but there is no assurance that we will receive such designation. Under the current statutory sunset provisions, the FDA may only award a priority review voucher for an approved rare pediatric disease application if the sponsor has a rare pediatric disease designation for a drug that is the subject of an application, and that designation was granted by December 20, 2024. The drug also must receive FDA approval no later than September 30, 2026 to qualify for the priority review vouchers. Although legislation to extend the rare pediatric disease priority review voucher program has been proposed, Congress has not yet, and may never, pass a bill to reauthorize the program and extend the sunset dates. There is no guarantee that the rare pediatric disease priority review voucher program will be extended or that any of our product candidates will obtain such designation and, therefore, we may not be in a position to obtain a priority review voucher. Additionally, designation of a drug for a rare pediatric disease does not guarantee that an NDA will meet the other eligibility criteria for a rare pediatric disease priority review voucher at the time the application is approved. Finally, a Rare Pediatric Disease designation does not lead to faster development or regulatory review of the product, or increase the likelihood that it will receive marketing approval.
28
We may use our financial and human resources to pursue a particular research program or product candidate and fail to capitalize on programs or product candidates that may be more profitable or for which there is a greater likelihood of success.
As a result of our limited financial and human resources, we will have to make strategic decisions as to which product candidates to pursue and may forego or delay pursuit of opportunities with other product candidates or for other indications that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities. Our spending on research and development programs and product candidates for specific indications may not yield any commercially viable products. If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product candidate through strategic alliance, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such product candidate, or we may allocate internal resources to a product candidate in a therapeutic area in which it would have been more advantageous to enter into a partnering arrangement.
We expect competition in the marketplace for our product candidates, should any of them receive regulatory approval.
If successfully developed and approved, our product candidates may face competition. We may not be able to compete successfully against organizations with competitive products, particularly large pharmaceutical companies. Many of our potential competitors have significantly greater financial, technical and human resources than us, and may be better equipped to develop, manufacture, market and distribute products. Many of these companies operate large, well-funded research, development and commercialization programs, have extensive experience in nonclinical and clinical studies, obtaining FDA and other regulatory approvals and manufacturing and marketing products, and have multiple products that have been approved or are in late-stage development. These advantages may enable them to receive approval from the FDA or any foreign regulatory agency before us.
Currently, there are no approved products to treat NS. However, to our knowledge, there are a number of therapeutic products at various stages of development for the treatment of NS, including candidates from LifeMax Laboratories, Inc., Krystal Biotech, Inc., Sixera Pharmaceuticals, ResVita Bio, and Azitra Inc. As of now, to the best of our knowledge, out of these companies only Azitra is actively dosing subject in clinical studies on NS patients under an open IND.
We face significant competition from other biotechnology and pharmaceutical companies and our operating results will suffer if we fail to compete effectively.
The biotechnology and pharmaceutical industries are intensely competitive. We have competitors both in the United States and internationally, including major multinational pharmaceutical companies, biotechnology companies, universities and other research institutions. Our competitors may have substantially greater financial, technical and other resources, such as larger research and development staff and experienced marketing and manufacturing organizations. Additional mergers and acquisitions in the biotechnology and pharmaceutical industries may result in even more resources being concentrated in our competitors. Competition may increase further as a result of advances in the commercial applicability of technologies and greater availability of capital for investment in these industries. Our competitors may succeed in developing, acquiring or licensing on an exclusive basis, drug products that are more effective or less costly than any product candidate that we may develop.
All of our product candidates are in either preclinical or clinical development and targeted toward indications for which there may be other product candidates in clinical development. We may face competition from other drugs currently approved or that may be approved in the future for the same therapeutic indications as our product candidates. Our ability to compete successfully will depend largely on our ability to leverage our experience in drug development to:
| ● | develop therapeutics that are superior to other products in the market; |
| ● | attract qualified scientific, product development and commercial personnel; |
| ● | obtain patent and/or other proprietary protection for our product candidates; |
| ● | obtain required regulatory approvals; and |
| ● | successfully collaborate with pharmaceutical companies in the discovery, development and commercialization of new therapeutics. |
29
The availability of our competitors’ products could limit the demand, and the price we are able to charge, for any products that we may develop and commercialize. We will not achieve our business plan if the acceptance of any of these products is inhibited by price competition or the reluctance of physicians to switch from existing drug products to our products, or if physicians switch to other new drug products or choose to reserve our future products for use in limited circumstances. The inability to compete with existing or subsequently introduced drug products would have a material adverse impact on our business, financial condition and prospects.
Established pharmaceutical companies may invest heavily to accelerate discovery and development of novel compounds or to in-license novel compounds that could make our product candidates less competitive. In addition, any new product that competes with an approved product must demonstrate compelling advantages in efficacy, convenience, tolerability and safety in order to overcome price competition and to be commercially successful. Accordingly, our competitors may succeed in obtaining patent protection, receiving FDA approval or discovering, developing and commercializing product candidates before we do, which would have a material adverse impact on our business.
The commercial success of our product candidates will depend upon the acceptance of these product candidates by the medical community, including physicians, patients and healthcare payors.
The degree of market acceptance of any product candidates will depend on a number of factors, including:
| ● | demonstration of clinical safety and efficacy compared to other products; |
| ● | the relative convenience, ease of administration and acceptance by physicians, patients and healthcare payors; |
| ● | the prevalence and severity of any AEs; |
| ● | limitations or warnings contained in the FDA-approved label for such products; |
| ● | availability of alternative treatments; |
| ● | pricing and cost-effectiveness; |
| ● | the effectiveness of our, or any of our collaborators’, sales and marketing strategies; |
| ● | our ability to obtain hospital or payor formulary approval; |
| ● | our ability to obtain and maintain sufficient third-party coverage and adequate reimbursement; and |
| ● | the willingness of patients to pay out-of-pocket in the absence of third-party coverage. |
If a product is approved but does not achieve an adequate level of acceptance by physicians, patients and healthcare payors, we may not generate sufficient revenues from such product and we may not become or remain profitable. Such increased competition may decrease any future potential revenue for future product candidates due to increasing pressure for lower pricing and higher discounts in the commercialization of our product.
If we are unable to establish sales and marketing capabilities or enter into agreements with third parties to market and sell our product candidates, we may be unable to generate any revenues.
We currently do not have an organization for the sales, marketing and distribution of pharmaceutical products and the cost of establishing and maintaining such an organization may exceed the cost-effectiveness of doing so. In order to market any products that may be approved, we must build our sales, marketing, managerial and other non-technical capabilities or make arrangements with third parties to perform these services. With respect to future programs, we may rely completely on an alliance partner for sales and marketing. In addition, we may enter into strategic alliances with third parties to commercialize other product candidates, if approved, including in markets outside of the United States and Europe or for other large markets that are beyond our resources. Although we intend to establish a sales organization if we are able to obtain approval to market any product candidates in the United States, and Europe we will also consider the option to enter into strategic alliances for future product candidates in the United States and Europe if commercialization requirements exceed our available resources. This will reduce the revenue generated from the sales of these products.
30
Any future strategic alliance partners may not dedicate sufficient resources to the commercialization of our product candidates, if approved, or may otherwise fail in their commercialization due to factors beyond our control. If we are unable to establish effective alliances to enable the sale of our product candidates, if approved, to healthcare professionals and in geographical regions, including the United States and Europe, that will not be covered by our own marketing and sales force, or if our potential future strategic alliance partners do not successfully commercialize the product candidates that may be approved, our ability to generate revenues from product sales will be adversely affected.
If we are unable to establish adequate sales, marketing and distribution capabilities, whether independently or with third parties, we may not be able to generate sufficient product revenue and may not become profitable. We will be competing with many companies that currently have extensive and well-funded marketing and sales operations. Without an internal team or the support of a third party to perform marketing and sales functions, we may be unable to compete successfully against these more established companies.
If we obtain approval to commercialize any approved products outside of the United States and Europe, a variety of risks associated with international operations could materially adversely affect our business.
If we obtain approval to commercialize any approved products outside of the United States and Europe, we expect that we will be subject to additional risks related to entering into international business relationships, including:
| ● | different regulatory requirements for drug approvals in foreign countries; |
| ● | differing payor reimbursement regimes, governmental payors or patient self-pay systems and price controls; |
| ● | reduced protection for intellectual property rights; |
| ● | unexpected changes in tariffs, trade barriers and regulatory requirements; |
| ● | economic weakness, including inflation, or political instability in particular foreign economies and markets; |
| ● | compliance with tax, employment, immigration and labor laws for employees living or traveling abroad; |
| ● | foreign taxes, including withholding of payroll taxes; |
| ● | foreign currency fluctuations, which could result in increased operating expenses and reduced revenues, and other obligations incident to doing business in another country; |
| ● | workforce uncertainty in countries where labor unrest is more common than in the United States; |
| ● | production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad; and |
| ● | business interruptions resulting from geopolitical actions, including war and terrorism, or natural disasters including earthquakes, typhoons, floods and fires. |
31
Coverage and adequate reimbursement may not be available for our product candidates, if approved, which could make it difficult for us to sell products profitably.
Market acceptance and sales of any product candidates that we develop will depend on coverage and reimbursement policies and may be affected by future healthcare reform measures. Government authorities and third-party payors, such as private health insurers, government payors and health maintenance organizations, decide which drugs they will pay for and establish reimbursement levels. We cannot be sure that coverage and adequate reimbursement will be available for any future product candidates. In the United States, the Centers for Medicare & Medicaid Services (“CMS”), an agency within the U.S. Department of Health and Human Services, decides whether and to what extent a new drug will be covered and reimbursed under Medicare. Private payors tend to follow the coverage reimbursement policies established by CMS to a substantial degree. It is difficult to predict what CMS will decide with respect to reimbursement for novel product candidates. Inadequate reimbursement amounts may reduce the demand for, or the price of, our future products. Further, one payor’s determination to provide coverage for a product does not assure that other payors will also provide coverage for the product. If reimbursement is not available, or is available only at limited levels, we may not be able to successfully commercialize product candidates that we develop and that may be approved. Thus, even if we succeed in bringing a product to market, it may not be considered medically necessary or cost-effective, and the amount reimbursed for any products may be insufficient to allow us to sell our products on a competitive basis.
There have been a number of legislative and regulatory proposals to change the healthcare system in the United States and in some foreign jurisdictions that could affect our ability to sell products profitably. These legislative and/or regulatory changes may negatively impact the reimbursement for drug products, following approval. The availability of numerous generic treatments may also substantially reduce the likelihood of reimbursement for our future products. We expect to experience pricing pressures in connection with the sale of any products that we develop, due to the trend toward managed healthcare, the increasing influence of health maintenance organizations and additional legislative changes. The downward pressure on healthcare costs in general, and prescription drugs in particular, has and is expected to continue to increase in the future. For instance, government and private payors who reimburse patients or healthcare providers are increasingly seeking greater upfront discounts, additional rebates and other concessions to reduce prices for pharmaceutical products. If we fail to successfully secure and maintain reimbursement coverage for our future products or are significantly delayed in doing so, we will have difficulty achieving market acceptance of our future products and our business will be harmed.
In addition, in some non-U.S. jurisdictions, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, the EU provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A member state may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the EU do not follow price structures of the U.S. and generally tend to be priced significantly lower.
Risks Related to Our Reliance on Third Parties
We rely on third parties to conduct some aspects of our compound formulation, research and preclinical studies, and those third parties may not perform satisfactorily, including failing to meet deadlines for the completion of such formulation, research or testing.
We do not expect to independently conduct all aspects of our drug development activities, compound formulation research or preclinical studies of product candidates. We currently rely and expect to continue to rely on third parties to conduct some or all aspects of our preclinical studies and formulation development.
Any of these third parties may terminate their engagements with us at any time. If we need to enter into alternative arrangements, it could delay our product development activities. Our reliance on these third parties for research and development activities will reduce our control over these activities but will not relieve us of our responsibilities.
If these third parties do not successfully carry out their contractual duties, meet expected deadlines or conduct our studies in accordance with regulatory requirements or our stated study plans and protocols, we will not be able to complete, or may be delayed in completing, the necessary preclinical studies to enable us to select viable product candidates for IND submissions and will not be able to, or may be delayed in our efforts to, successfully develop and commercialize such product candidates.
32
We rely, or will rely, on third-party manufacturers to produce the supply of our preclinical product, clinical product candidates and commercial supplies of any approved product candidates.
Reliance on third-party manufacturers entails risks, including risks that we would not be subject to if we manufactured the product candidates ourselves.
Third-party manufacturers may not be able to comply with cGMP regulations or similar regulatory requirements outside of the United States. If the FDA determines that our third-party manufacturers are not in compliance with FDA laws and regulations, including those governing cGMPs, the FDA may not approve an NDA until the deficiencies are corrected or we replace the manufacturer in our application with a manufacturer that is in compliance. Moreover, our failure, or the failure of our third-party manufacturers and suppliers, to comply with applicable regulations could result in sanctions being imposed on us, including clinical holds, fines, injunctions, civil penalties, seizures or recalls of product candidates or products, operating restrictions and criminal prosecutions, any of which could significantly and adversely affect supplies of our products. In addition, approved products and the facilities at which they are manufactured are required to maintain ongoing compliance with extensive FDA requirements and the requirements of other similar agencies, including ensuring that quality control and manufacturing procedures conform to cGMP requirements. As such, our third-party manufacturers are subject to continual review and periodic inspections to assess compliance with cGMPs. Furthermore, although we do not have day-to-day control over the operations of our third-party manufacturers, we are responsible for ensuring compliance with applicable laws and regulations, including cGMPs.
Other risks of reliance on third-party manufacturers include:
| ● | the inability to meet any product specifications and quality requirements consistently; |
| ● | a delay or inability to procure or expand sufficient manufacturing capacity; |
| ● | manufacturing and product quality issues related to scale-up of manufacturing; |
| ● | costs and validation of new equipment and facilities required for scale-up; |
| ● | the inability to negotiate manufacturing or supply agreements with third parties under commercially reasonable terms; |
| ● | termination or nonrenewal of manufacturing agreements with third parties in a manner or at a time that is costly or damaging to us; |
| ● | the reliance on a limited number of sources, and in some cases, single sources for raw materials, such that if we are unable to secure a sufficient supply of these product components, we will be unable to manufacture and sell future product candidates in a timely fashion, in sufficient quantities or under acceptable terms; |
| ● | the lack of qualified backup suppliers for any raw materials that are currently purchased from a single source supplier; |
| ● | operations of our third-party manufacturers or suppliers could be disrupted by conditions unrelated to our business or operations, including the bankruptcy of the manufacturer or supplier; |
| ● | carrier disruptions or increased costs that are beyond our control; and |
| ● | the failure to deliver products under specified storage conditions and in a timely manner. |
Any of these events could lead to clinical study delays or failure to obtain regulatory approval, or impact our ability to successfully commercialize future products, if approved. Some of these events could be the basis for FDA action, including injunction, recall, seizure or total or partial suspension of production.
33
We rely on limited sources of supply for the drug substance of product candidates and any disruption in the chain of supply may cause a delay in developing and commercializing these product candidates.
We have established manufacturing relationships with a limited number of suppliers to manufacture raw materials and the drug substance used to create our product candidates. The availability of such suppliers to manufacture raw materials and drug substance for our product candidates in sufficient quantities for evaluation in preclinical or clinical studies or, if our product candidates are approved, for commercial supply may be limited. Further, each supplier may require licenses to manufacture such components if such processes are not owned by the supplier or in the public domain. As part of any marketing approval, a manufacturer and its processes are required to be qualified by the FDA prior to commercialization. If product supply from any manufacturer approved in the NDA is interrupted, there could be a significant disruption in commercial supply. An alternative manufacturer would need to be qualified through an NDA supplement which could result in further delay. The FDA or other regulatory agencies outside of the United States may also require additional studies if a new manufacturer is relied upon for commercial production. Switching manufacturers may involve substantial costs and is likely to result in a delay in our desired clinical and commercial timelines.
These factors could cause the delay of clinical trials, regulatory submissions, required approvals or commercialization of our product candidates, cause us to incur higher costs and prevent us from commercializing our products successfully. Furthermore, if our suppliers fail to deliver the required commercial quantities of active pharmaceutical ingredients on a timely basis and at commercially reasonable prices, and we are unable to secure one or more replacement suppliers capable of production in a timely manner at a substantially equivalent cost, our clinical trials may be delayed, or we could lose potential revenue.
Manufacturing issues may arise that could increase product and regulatory approval costs or delay commercialization.
Manufacturing of product candidates and conducting required stability testing, product, packaging, equipment and process-related issues may require refinement or resolution in order to proceed with any clinical trials and obtain regulatory approval for commercial marketing. We may identify significant impurities, which could result in increased scrutiny by the regulatory agencies, delays in clinical programs and regulatory approval, increases in our operating expenses, or failure to obtain or maintain approval for product candidates or any approved products.
We intend to rely on third parties to conduct, supervise and monitor our clinical trials, and if those third parties perform in an unsatisfactory manner, it may harm our business.
We intend to rely on CROs and clinical trial sites to ensure the proper and timely conduct of our clinical trials. While we will have agreements governing their activities, we have limited influence over their actual performance. We will control only certain aspects of our CROs’ activities. Nevertheless, we will be responsible for ensuring that each of our clinical trials are conducted in accordance with the applicable protocol, legal, regulatory and scientific standards and our reliance on the CROs will not relieve us of our regulatory responsibilities.
We and our CROs will be required to comply with the FDA’s or other regulatory agency’s GCPs, for conducting, recording and reporting the results of IND-enabling studies and clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of clinical trial participants are protected. The FDA and non-U.S. regulatory agencies enforce these GCPs through periodic inspections of trial sponsors, CROs, principal investigators and clinical trial sites. If we or our CROs fail to comply with applicable GCPs, the clinical data generated in our clinical trials may be deemed unreliable and the FDA or applicable non-U.S. regulatory agency may require us to perform additional clinical trials before approving any marketing applications for the relevant jurisdiction. Upon inspection, the FDA or applicable non-U.S. regulatory agency may determine that our clinical trials did not comply with GCPs. In addition, our clinical trials will require a sufficiently large number of test subjects to evaluate the safety and effectiveness of a potential drug product. Accordingly, if our CROs fail to comply with these regulations or fail to recruit a sufficient number of patients, we may be required to repeat such clinical trials, which would delay the regulatory approval process.
Our CROs will not be our employees, and we will not be able to control whether or not they devote sufficient time and resources to our clinical and nonclinical programs. These CROs may also have relationships with other commercial entities, including our competitors, for whom they may also be conducting clinical trials, or other drug development activities which could harm our competitive position. If our CROs do not successfully carry out their contractual duties or obligations, fail to meet expected deadlines, or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols or regulatory requirements, or for any other reasons, our clinical trials may be extended, delayed or terminated, and we may not be able to obtain regulatory approval for, or successfully commercialize our product candidates. As a result, our financial results and the
34
commercial prospects for such products and any product candidates that we develop would be harmed, our costs could increase, and our ability to generate revenues could be delayed.
We intend to rely on other third parties to package, store and deliver drug products to the clinical trial sites for any clinical trials that we may conduct. Any performance failure on the part of these third parties could delay clinical development or marketing approval of our product candidates or commercialization of our products, if approved, producing additional losses and depriving us of potential product revenue.
Risks Related to Our Intellectual Property
If we are unable to obtain or protect intellectual property rights related to our future products and product candidates, we may not be able to compete effectively in our markets.
Our success depends in part on our ability to obtain and maintain patents and other forms of intellectual property rights, including in-licenses of intellectual property rights of others, for our product candidates, methods used to develop and manufacture our product candidates and methods for treating patients using our product candidates, as well as our ability to preserve our trade secrets, to prevent third parties from infringing upon our proprietary rights and to operate without infringing upon the proprietary rights of others. The strength of patents in the biotechnology and pharmaceutical field involves complex legal and scientific questions and can be uncertain. Our patent applications may fail to result in patents with claims that cover the products in the United States or in other countries. There is no assurance that all of the potentially relevant prior art relating to patents and patent applications that we use in our business has been found; such prior art can invalidate a patent or prevent a patent from issuing based on a pending patent application. Even if patents do successfully issue, third parties may challenge their validity, enforceability or scope, which may result in such patents being narrowed or invalidated. Furthermore, even if they are unchallenged, our patents and patent applications may not adequately protect our intellectual property or prevent others from designing around our claims.
If the patent applications we hold or patents we have in-licensed with respect to our programs or product candidates fail to issue or if their breadth or strength of protection is threatened, as applicable, it could dissuade companies from collaborating with us to develop product candidates, and threaten our ability to commercialize, future products. We cannot offer any assurances about which, if any, patents will issue or whether any issued patents will be found invalid and unenforceable or will be threatened by third parties. A patent may be challenged through one or more of several administrative proceedings including post-grant challenges, re-examination or opposition before the USPTO or foreign patent offices. Any successful challenge of patents or any other patents owned by or licensed to us could deprive us of rights necessary for the successful commercialization of any product candidates that we may develop.
Since patent applications in the United States and most other countries are confidential for a period of time after filing, and some remain so until issued, we cannot be certain that we were the first to file any patent application related to a product candidate. Furthermore, in certain situations, if we and one or more third parties have filed patent applications in the United States and claiming the same subject matter, an administrative proceeding, known as an interference, can be initiated to determine which applicant is entitled to the patent on that subject matter. Such an interference proceeding provoked by third parties or brought by us may be necessary to determine the priority of inventions with respect to our patents or patent applications, or those of our licensors. An unfavorable outcome could require us to cease using the related technology or to require us to license rights to it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license at all, or on commercially reasonable terms. Our defense of a patent or patent application in such a proceeding may not be successful and, even if successful, may result in substantial costs and distract our management.
In addition, patents have a limited lifespan. In the United States, the natural expiration of a patent is generally 20 years after it is filed. Various extensions may be available, however the life of a patent, and the protection it affords is limited. Once the patent life has expired for a product, we may be open to competition from generic medications. Further, if we encounter delays in regulatory approvals, the period of time during which we could market a product candidate under patent protection could be reduced. One of our patents expired in March 2024 and another is set to expire in July 2025.
In addition to the protection afforded by patents, we rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable, including processes for which patents are difficult to enforce and any other elements of our drug discovery and development processes that involve proprietary know-how, information or technology that is not covered by patents. Although we expect all our employees to assign their inventions to us, and all our employees, consultants, advisors and any third parties who have access to our proprietary know-how, information or technology to enter into confidential information and invention agreements, we cannot provide any assurances that all such agreements have been or will be duly executed or will be enforceable. In
35
addition, others may independently discover our trade secrets and proprietary information. For example, the FDA, as part of its Transparency Initiative, is currently considering whether to make additional information publicly available on a routine basis, including information that we may consider to be trade secrets or other proprietary information, and it is not clear at the present time how the FDA’s disclosure policies may change in the future, if at all.
Further, the laws of some foreign countries do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant problems in protecting and defending our intellectual property both in the United States and abroad. If we are unable to prevent material disclosure of the non-patented intellectual property related to our technologies to third parties, and there is no guarantee that we will have any such enforceable trade secret protection, we may not be able to establish or maintain a competitive advantage in our market, which could materially adversely affect our business, results of operations and financial condition.
Third-party claims of intellectual property infringement may prevent or delay our development and commercialization efforts.
Our commercial success depends in part on our avoiding infringement of the patents and proprietary rights of third parties. There is a substantial amount of litigation, both within and outside the United States, involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits. Numerous U.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields in which we are pursuing development candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that our product candidates may be subject to claims of infringement of the patent rights of third parties.
Third parties may assert that we are employing their proprietary technology without authorization. There may be third-party patents or patent applications with claims to materials, formulations, methods of manufacture or methods for treatment related to the use or manufacture of our product candidates. Because patent applications can take many years to issue, there may be currently pending patent applications which may later result in patents that our product candidates may infringe. In addition, third parties may obtain patents in the future and claim that use of our technologies infringes upon these patents. If any third-party patents were held by a court of competent jurisdiction to cover the manufacturing process of any of our product candidates, any molecules formed during the manufacturing process or any final product itself, the holders of any such patents may be able to block our ability to commercialize such product candidate unless we obtained a license under the applicable patents, or until such patents expire. Similarly, if any third-party patents were held by a court of competent jurisdiction to cover aspects of our formulations, processes for manufacture or methods of use, including combination therapy, the holders of any such patents may be able to block our ability to develop and commercialize the applicable product candidate unless we obtained a license or until such patent expires. In either case, such a license may not be available on commercially reasonable terms or at all.
Parties making claims against us may obtain injunctive or other equitable relief, which could effectively block our ability to further develop and commercialize one or more of our product candidates. Defense of these claims, regardless of their merit, would involve substantial litigation expense and would be a substantial diversion of management resources from our business. In the event of a successful claim of infringement against us, we may have to pay substantial damages, including treble damages and attorneys’ fees for willful infringement, pay royalties, redesign our infringing products or obtain one or more licenses from third parties, which may be impossible or require substantial time and monetary expenditure.
If we fail to obtain licenses or comply with our obligations in these agreements under which we license intellectual property rights from third parties or otherwise experience disruptions to our business relationships with our licensors, we could lose license rights that are important to our business.
We are a party to intellectual property license agreements that are important to our business and expect to enter into additional license agreements in the future. Our existing license agreements impose, and we expect that future license agreements will impose, various obligations on us.
We may need to obtain licenses from third parties to advance our research or allow commercialization of our product candidates, and we have done so from time to time. We may fail to obtain any of these licenses at a reasonable cost or on reasonable terms, if at all. In that event, we would be unable to further develop and commercialize one or more of our product candidates, which could harm our business significantly. We cannot provide any assurances that third-party patents do not exist which might be enforced against our future products, resulting in either an injunction prohibiting our sales, or, with respect to our sales, an obligation on our part to pay royalties and/or other forms of compensation to third parties.
36
We may be involved in lawsuits to protect or enforce our patents or the patents of our licensors, which could be expensive, time consuming and unsuccessful.
Competitors may infringe our patents or the patents of our licensors. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive and time-consuming. In addition, in an infringement proceeding, a court may decide that a patent of ours or of our licensors is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation or defense proceedings could put one or more of our patents at risk of being invalidated or interpreted narrowly and could put our patent applications at risk of not issuing.
Our defense in a lawsuit may fail and, even if successful, may result in substantial costs and distract our management. We may not be able to prevent, alone or with our licensors, misappropriation of our intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our ordinary shares.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties.
We have or may employ individuals who were previously employed at other biotechnology or pharmaceutical companies. We may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise used or disclosed confidential information of our employees’ former employers or other third parties. We may also be subject to claims that former employers or other third parties have an ownership interest in our patents. Litigation may be necessary to defend against these claims. There is no guarantee of success in defending these claims, and if we are successful, litigation could result in substantial cost and be a distraction to management and other employees.
Other Risks Related to Our Business Operations and Industry
Our future success depends on our ability to attract and retain key executives and to attract, retain and motivate qualified personnel.
We are highly dependent on principal members of our executive team, and any reduction or loss of their services may adversely impact the achievement of our objectives. While we have entered into employment agreements with each of our executive officers, any of them could leave our employment at any time. Recruiting and retaining other qualified employees for our business, including scientific and technical personnel, will also be critical to our success. There is currently a shortage of skilled executives in our industry, which is likely to continue. As a result, competition for skilled personnel is intense and the turnover rate can be high. We may not be able to attract and retain personnel on acceptable terms given the competition among numerous pharmaceutical companies for individuals with similar skill sets. In addition, failure to succeed in preclinical studies and clinical trials may make it more challenging to recruit and retain qualified personnel. The inability to recruit any executive or key employee or the loss of the services of any executive or key employee might impede the progress of our research, development and commercialization objectives.
We may need to expand our organization and may experience difficulties in managing our growth, which could disrupt our operations.
In the future we may expand our employee base to increase our managerial, scientific, operational, commercial, financial and other resources and we may hire more consultants and contractors. Future growth would impose significant additional responsibilities on our management, including the need to identify, recruit, maintain, motivate and integrate additional employees, consultants and contractors. Also, our management may need to divert a disproportionate amount of its attention away from our day-to-day activities and devote a substantial amount of time to managing these growth activities. Our expected growth could require significant capital expenditures and may divert financial resources from other projects, such as the development of additional product candidates. Moreover, if our management is unable to effectively manage our growth, our expenses may increase more than expected, our ability to generate and/or grow revenues could be reduced, and we may not be able to implement our business strategy. Our future financial performance and our ability to commercialize product candidates and compete effectively will depend, in part, on our ability to effectively manage any future growth.
37
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements and insider trading.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include intentional or nonintentional failures to comply with the regulations of the FDA and non-U.S. regulators, to provide accurate information to the FDA and non-U.S. regulators, to comply with healthcare fraud and abuse laws and regulations in the United States and abroad, to report financial information or data accurately or to disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements.
Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and cause serious harm to our reputation. We have adopted a code of ethics and business conduct, but it is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of civil, criminal and administrative penalties, damages, fines, possible exclusion from Medicare, Medicaid and other government healthcare programs, additional reporting requirements and/or oversight, particularly if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance, disgorgement, imprisonment, and contractual damages. Even if we are ultimately successful in defending against any such action, we could be required to divert financial and managerial resources in doing so and adverse publicity could result, all of which could harm our business.
Future relationships with customers and third-party payors as well as certain of our business operations may be subject, directly or indirectly, to federal and state healthcare fraud and abuse laws, false claims laws and health information privacy and security laws. If we are unable to comply, or have not fully complied, with such laws, we could face criminal sanctions, civil penalties, contractual damages, reputational harm and diminished profits and future earnings.
If we obtain FDA approval for any of our product candidates and begin commercializing those products in the United States, our operations may be directly, or indirectly through our customers, further subject to various federal and state fraud and abuse laws, including, without limitation, the federal Anti-Kickback Statute and the federal False Claims Act. These laws may impact, among other things, our proposed sales, marketing and education programs. In addition, we may be subject to patient privacy regulation by the federal government and by the U.S. states and foreign jurisdictions in which we conduct our business. The healthcare laws and regulations that may affect our ability to operate include:
| ● | The federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce, or in return for, either the referral of an individual, or the purchase or recommendation of an item or service for which payment may be made under a federal healthcare program, such as the Medicare and Medicaid programs. Remuneration has been interpreted broadly to include anything of value. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution, the exemptions and safe harbors are drawn narrowly, and those activities may be subject to scrutiny or penalty if they do not qualify for an exemption or safe harbor. A conviction for violation of the Anti-Kickback Statute requires mandatory exclusion from participation in federal healthcare programs. This statute has been applied to arrangements between pharmaceutical manufacturers and those in a position to purchase products or refer others, including prescribers, patients, purchasers and formulary managers. In addition, the Affordable Care Act amended the Social Security Act to provide that the U.S. government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act penalties for which are described below. |
| ● | Federal civil and criminal false claims laws and civil monetary penalty laws, including the federal False Claims Act (“FCA”), which imposes criminal or civil penalties, including through civil whistleblower or qui tam actions, against individuals or entities for, among other things, knowingly presenting, or causing to be presented, claims for payment to the federal government, including Medicare or Medicaid, that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government. FCA liability is potentially significant in the healthcare industry because the statute provides for treble damages and mandatory penalties per false claim or statement. |
38
| ● | The civil monetary penalties statute, which imposes penalties against any person or entity who, among other things, is determined to have presented or caused to be presented a claim to a federal healthcare program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent. |
| ● | The federal Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), which imposes civil and criminal penalties for, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations or promises, any money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payor (e.g., public or private), knowingly and willfully embezzling or stealing from a health care benefit program, willfully obstructing a criminal investigation of a healthcare offense and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare. |
| ● | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (“HITECH”), and its implementing regulations, which imposes certain requirements on certain types of individuals and entities, such as healthcare providers, health plans and healthcare clearing houses, known as “covered entities,” as well as their “business associates,” independent contractors or agents of covered entities that receive or obtain individually identifiable health information in connection with providing a service on behalf of a covered entity, relating to the privacy, security and transmission of individually identifiable health information. |
| ● | The federal Physician Payments Sunshine Act, which requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, with specific exceptions, to report annually to CMS, information related to payments or other transfers of value made to physicians, physician assistants, certain types of advance practice nurses and teaching hospitals, and further requires applicable manufacturers and applicable group purchasing organizations to report annually to CMS ownership and investment interests held by physicians and their immediate family members. Failure to submit timely, accurately and completely the required information for all covered payments, transfers of value and ownership or investment interests may result in civil monetary penalties; and |
| ● | Many state and foreign law equivalents of each of the above federal laws, such as: anti-kickback and false claims laws which may apply to items or services reimbursed by any third party payor, including commercial insurers; state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government; state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; state and local laws that require the registration of pharmaceutical sales representatives; and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. |
In addition, the European Union (“EU”) has established its own data security and privacy legal framework, including but not limited to Directive 95/46/EC (the “Data Protection Directive”). The European General Data Protection Regulation (“GDPR”) contains new provisions specifically directed at the processing of health information, higher sanctions and extra-territoriality measures intended to bring non-EU companies under the regulation. We anticipate that over time we may expand our business operations to include additional operations in the EU, including potentially conducting preclinical and clinical trials. With such expansion, we would be subject to increased governmental regulation in the EU countries in which we might operate, including regulation due to the GDPR.
If our operations are found to be in violation of any of the laws described above or any other governmental regulations or laws that apply to us, we may be subject to penalties, including, without limitation, civil, criminal and administrative penalties, damages, fines, possible exclusion from Medicare, Medicaid and other government healthcare programs, additional reporting requirements and/or oversight, particularly if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance, disgorgement, imprisonment, contractual damages, reputational harm, diminished profits and future earnings, and curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
39
Our operations may be impacted from changes to current regulations and future legislation.
The United States and many foreign jurisdictions have enacted or proposed legislative and regulatory changes affecting the healthcare system that could prevent or delay approval of our products, restrict or regulate post-approval activities and affect our ability to profitably sell a drug product for which we obtain approval. Changes in regulations, statutes or the interpretation of existing regulations could impact our business in the future by requiring, for example:
| ● | changes to manufacturing arrangements; |
| ● | additions or modifications to product labeling; |
| ● | the recall or discontinuation of products; or |
| ● | additional record-keeping requirements. |
If any such changes were to be imposed, they could adversely affect the operation of our business. In the United States, there have been and continue to be a number of legislative initiatives to contain healthcare costs. For example, in March 2010, the Patient Protection and Affordable Care Act of 2010, as amended by the Health Care and Education Reconciliation Act of 2010, collectively, ACA, was passed, which substantially changed the way healthcare is financed by both governmental and private insurers, and significantly impacted the U.S. pharmaceutical industry. The ACA contained provisions that may reduce the profitability of drug products through increased rebates for drugs reimbursed by Medicaid programs, extension of Medicaid rebates to Medicaid managed care plans, mandatory discounts for certain Medicare Part D beneficiaries and annual fees based on pharmaceutical companies’ share of sales to federal health care programs. In December 2020, the U.S. Centers for Medicare & Medicaid Services, or CMS, issued a final rule implementing significant manufacturer price reporting changes under the Medicaid Drug Rebate Program, including regulations that affect manufacturer-sponsored patient assistance programs subject to pharmacy benefit manager accumulator programs and Best Price reporting related to certain value-based purchasing arrangements. The American Rescue Plan Act of 2021 eliminated the statutory cap on Medicaid Drug Rebate Program rebates that manufacturers pay to state Medicaid programs. Elimination of this cap may require pharmaceutical manufacturers to pay more in rebates than it receives on the sale of products.
In June 2021, the United States Supreme Court held that Texas and other challengers had no legal standing to challenge the ACA, dismissing the case without specifically ruling on the constitutionality of the ACA. Further, on August 16, 2022, President Biden signed the Inflation Reduction Act, or IRA, into law, which among other things, extends enhanced subsidies for individuals purchasing health insurance coverage in ACA marketplaces through plan year 2025. The IRA also eliminates the “donut hole” under the Medicare Part D program beginning in 2025 by significantly lowering the beneficiary maximum out-of-pocket cost through a newly established manufacturer discount program. It is possible that the ACA will be subject to judicial or Congressional challenges in the future. It is unclear how additional challenges and healthcare reform measures under the Trump administration will impact the ACA. Complying with any new legislation and regulatory requirements could be time-intensive and expensive.
The Bipartisan Budget Act of 2018 also amended the ACA, effective January 1, 2019, by increasing the point-of-sale discount that is owed by pharmaceutical manufacturers who participate in Medicare Part D and closing the coverage gap in most Medicare drug plans, commonly referred to as the “donut hole.” CMS published a final rule permitting further collections and payments to and from certain ACA qualified health plans and health insurance issuers under the ACA risk adjustment program in response to the outcome of federal district court litigation regarding the method CMS uses to determine this risk adjustment. In addition, CMS has published a final rule to give states greater flexibility, starting in 2020, in setting benchmarks for insurers in the individual and small group marketplaces, which may have the effect of relaxing the essential health benefits required under the ACA for plans sold through such marketplaces. The American Taxpayer Relief Act of 2012, among other things, reduced Medicare payments to several providers, including hospitals, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. Other legislative changes include aggregate reductions to Medicare payments to providers of up to 2% per fiscal year pursuant to the Budget Control Act of 2011, which began in 2013 and will remain in effect through 2032, with the exception of a temporary suspension implemented under various COVID-19 relief legislation, unless Congress takes further action.
There has been increasing legislative and enforcement interest in the United States with respect to specialty drug pricing practices. Specifically, there have been several recent U.S. Congressional inquiries and proposed federal and state legislation designed to, among other things, bring more transparency to drug pricing, reduce the cost of prescription drugs under Medicare, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drugs. For
40
example, in August 2022, Congress passed the IRA, which includes prescription drug provisions that have significant implications for the pharmaceutical industry and Medicare beneficiaries, including allowing the federal government to negotiate a maximum fair price for certain high-priced single source Medicare drugs, imposing penalties and excise tax for manufacturers that fail to comply with the drug price negotiation requirements, requiring inflation rebates for all Medicare Part B and Part D drugs, with limited exceptions, if their drug prices increase faster than inflation, and redesigning Medicare Part D to reduce out-of-pocket prescription drug costs for beneficiaries, among other changes. Various industry stakeholders, including pharmaceutical companies and the Pharmaceutical Research and Manufacturers of America, have initiated lawsuits against the federal government asserting that the price negotiation provisions of IRA are unconstitutional. The impact of these judicial challenges as well as other legislative, executive, and administrative actions and any future healthcare measures is unclear. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize our product candidates, if approved.
Our revenue prospects could be affected by changes in healthcare spending and policy in the United States and abroad. We operate in a highly regulated industry and new laws, regulations or judicial decisions, or new interpretations of existing laws, regulations or decisions, related to healthcare availability, the method of delivery or payment for healthcare products and services could negatively impact our business and operations.
There have been, and likely will continue to be, legislative and regulatory proposals at the foreign and federal levels directed at broadening the availability of healthcare and containing or lowering the cost of healthcare. We cannot predict the initiatives that may be adopted in the future, including repeal, replacement or significant revisions to the ACA. The continuing efforts of the government, insurance companies, managed care organizations and other payors of healthcare services to contain or reduce costs of healthcare or impose price controls may adversely affect our ability to set a price that we believe is fair for our products. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our product candidates.
Legislative and regulatory proposals have been made to expand post-approval requirements and restrict sales and promotional activities for biotechnology products. We cannot be sure to what extent the trajectory of these legislative and regulatory proposals will be implemented, whether additional legislative changes will be enacted, whether FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on the marketing approvals of our product candidates, if any, may be. In addition, increased scrutiny by Congress of the FDA’s approval process may significantly delay or prevent approval, as well as subject us to more stringent product labeling and post-marketing testing and other requirements.
We face potential product liability, and, if successful claims are brought against us, we may incur substantial liability and costs.
The use of our product candidates in clinical trials and the sale of any products for which we obtain marketing approval exposes us to the risk of product liability claims. Product liability claims might be brought against us by consumers, healthcare providers, pharmaceutical companies or others selling or otherwise coming into contact with our products. For example, unanticipated adverse effects could result from the use of our future products or product candidates which may result in a potential product liability claim. If we cannot successfully defend against product liability claims, we could incur substantial liability and costs. In addition, regardless of merit or eventual outcome, product liability claims may result in:
| ● | impairment of our business reputation; |
| ● | withdrawal of clinical trial participants; |
| ● | costs due to related litigation; |
| ● | distraction of management’s attention from our primary business; |
| ● | substantial monetary awards to patients or other claimants; |
| ● | the inability to commercialize our product candidates; and |
| ● | decreased demand for our product candidates, if approved for commercial sale. |
41
We plan to obtain product liability insurance relating to the use of our therapeutics in clinical trials. However, such insurance coverage may not be sufficient to reimburse us for any expenses or losses we may suffer. Moreover, insurance coverage is becoming increasingly expensive and in the future we may not be able to obtain or maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability. If and when we obtain marketing approval for product candidates, we intend to expand our insurance coverage to include the sale of commercial products; however, we may be unable to obtain product liability insurance on commercially reasonable terms or in adequate amounts. On occasion, large judgments have been awarded in class action lawsuits based on drugs that had unanticipated adverse effects. A successful product liability claim or series of claims brought against us could cause our share price to decline and, if judgments exceed our insurance coverage, could adversely affect our results of operations and business.
Cyber security risks and the failure to maintain the confidentiality, integrity, and availability of our computer hardware, software, and Internet applications and related tools and functions could result in damage to our reputation and/or subject us to costs, fines or lawsuits.
Our business and operations would suffer in the event of computer system failures, cyber-attacks or a deficiency in our cyber-security. Despite the implementation of security measures, our internal computer systems, and those of third parties on which we rely, are vulnerable to damage from computer viruses, malware, supply chain attacks, ransomware attacks, natural disasters, terrorism, war, telecommunication and electrical failures, cyber-attacks or cyber-intrusions over the Internet, attachments to emails, persons inside our organization or inside external organizations on which we rely for support, systems, or hardware. The risk of a security breach or disruption, particularly through cyber-attacks or cyber intrusion, including by computer hackers, foreign governments, and cyber terrorists, has generally increased as the number, intensity and sophistication of attempted attacks and intrusions from around the world have increased. If such an event were to occur and cause interruptions in our operations, it could result in a material disruption of business. Maintaining safeguards to comply with evolving security laws and to protect our systems and data may increase our operating costs. To the extent that any disruption or security breach was to result in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur material legal claims and liability, damage to our reputation, and a delay in development of our drug candidates.
We have been, and may in the future be, adversely affected by health epidemics and pandemics which may significantly harm our business, prospects, financial condition and operating results.
We face risks related to health epidemics and other outbreaks. For example, during 2020, the spread of the novel coronavirus led to disruption and volatility in the global capital markets. If such disruption and volatility recurs, there could be an increase to our cost of capital and an adverse effect on our ability to access the capital markets. The extent to which a pandemic, epidemic or outbreak of an infectious disease impacts our operations, including our clinical trials, will depend on future occurrences, which are highly uncertain and cannot be predicted with confidence, including the duration of any outbreak and the actions to contain or treat its impact, among others. Any negative impact infectious diseases have on patient enrollment or treatment or the execution of our product candidates could cause costly delays to clinical trial activities, which could adversely affect our ability to obtain regulatory approval for and to commercialize our product candidates, increase our operating expenses, and have a material adverse effect on our financial results.
Business interruptions could delay us in the process of developing our future products.
We are vulnerable to natural disasters such as earthquakes and wildfires, as well as other events that could disrupt our operations. We do not carry insurance for earthquakes or other natural disasters and we may not carry sufficient business interruption insurance to compensate us for losses that may occur. Any losses or damages we incur could have a material adverse effect on our business operations.
Risks Related to Us Being an Israeli Company
Shareholders may have difficulties enforcing a U.S. judgment, including judgments based upon the civil liability provisions of the U.S. federal securities laws, against us or our executive officers and directors, or asserting U.S. securities laws claims in Israel.
Service of process upon us in Israel or upon our non-U.S. resident directors and officers may be difficult to obtain within the United States and it may be difficult to enforce judgments obtained in the United States against our non-U.S. directors and executive officers. In addition, we have been informed by our legal counsel in Israel that it may be difficult to assert claims under U.S. securities laws in original actions instituted in Israel or obtain a judgment based on the civil liability provisions of U.S. federal securities laws. Israeli courts may refuse to hear a claim based on a violation of U.S. securities laws against us or our officers and directors because Israel may not be the most appropriate forum to bring such a claim. In addition, even if an Israeli court agrees to hear a claim, it may determine
42
that Israeli law and not U.S. law is applicable to the claim. If U.S. law is found to be applicable, the content of applicable U.S. law must be proved as a fact, which can be a time-consuming and costly process. Certain matters of procedure will also be governed by Israeli law. There is little binding case law in Israel addressing the matters described above. Israeli courts might not enforce judgments rendered outside Israel, which may make it difficult to collect on judgments rendered against us or our officers and directors in Israel.
Moreover, an Israeli court will not enforce a foreign judgment if it was given in a state whose laws do not provide for the enforcement of judgments of Israeli courts (subject to exceptional cases) or if its enforcement is likely to prejudice the sovereignty or security of the State of Israel or due to, among other reasons, absence of due process, or the existence of a judgment which is at variance with another judgment that was given in the same matter if a suit in the same matter between the same parties was pending before a court or tribunal in Israel.
The rights and responsibilities of our shareholders are governed by Israeli law, which may differ in some respects from the rights and responsibilities of shareholders of U.S. corporations.
Since we are incorporated under Israeli law, the rights and responsibilities of our shareholders are governed by our articles of association and Israeli law. These rights and responsibilities differ in some respects from the rights and responsibilities of shareholders of U.S.-based corporations. In particular, a shareholder of an Israeli company, such as us, has a duty to act in good faith and in a customary manner in exercising its rights and performing its obligations towards us and other shareholders and to refrain from abusing its power in us, including, among other things, in voting at the general meeting of shareholders on certain matters, such as an amendment to our articles of association, an increase of our authorized share capital, a merger, and approval of related party transactions that require shareholder approval. A shareholder also has a general duty to refrain from taking advantage of other shareholders. In addition, a controlling shareholder (as defined below), or any shareholder who knows that it possesses the power to determine the outcome of a shareholders’ vote, or who has the power to appoint or prevent the appointment of one of our office holders (as defined below), or who holds any other power in our regard, has a duty to act in fairness towards us. However, Israeli law does not define the substance of this duty of fairness. There is limited case law available to assist in understanding the implications of these provisions that govern shareholder behavior.
Provisions of Israeli law may delay, prevent or otherwise impede a merger with, or an acquisition of, our company, which could prevent a change of control, even when the terms of such a transaction are favorable to us and our shareholders.
Israeli corporate law regulates mergers, requires tender offers for acquisitions of shares above specified thresholds, requires special approvals for transactions involving directors, officers or significant shareholders, and regulates other matters that may be relevant to these types of transactions. For example, a merger may not be consummated unless at least 50 days have passed from the date that a merger proposal was filed by each merging company with the Israel Registrar of Companies, and at least 30 days from the date that the shareholders of both merging companies approved the merger. In addition, the holder of a majority of each class of securities of the target company must approve a merger. Moreover, a full tender offer can only be completed if the acquirer receives at least 95% of the issued share capital (provided that a majority of the offerees that do not have a personal interest in such tender offer shall have approved the tender offer, except that if the total votes to reject the tender offer represent less than 2% of the company’s issued and outstanding share capital, in the aggregate, approval by a majority of the offerees that do not have a personal interest in such tender offer is not required to complete the tender offer), and the shareholders, including those who indicated their acceptance of the tender offer, may, at any time within six months following the completion of the tender offer, petition the court to alter the consideration for the acquisition (unless the acquirer stipulated in the tender offer that a shareholder that accepts the offer may not seek appraisal rights).
Furthermore, Israeli tax considerations may make potential transactions unappealing to us or to those of our shareholders whose country of residence does not have a tax treaty with Israel exempting such shareholders from Israeli tax. For example, Israeli tax law does not recognize tax-free share exchanges to the same extent as U.S. tax law. With respect to mergers, Israeli tax law allows for tax deferral in certain circumstances, but makes the deferral contingent on the fulfillment of numerous conditions, including a holding period of two years from the date of the transaction during which sales and dispositions of shares of the participating companies are restricted. Moreover, with respect to certain share swap transactions, the tax deferral is limited in time, and when such time expires, the tax becomes payable even if no actual disposition of the shares has occurred. Additional tax considerations or exemptions from the foregoing may apply to certain non-Israeli tax resident shareholders.
These and other similar provisions could delay, prevent or impede an acquisition of us or our merger with another company, even if such an acquisition or merger would be beneficial to us or to our shareholders.
43
Risks Related to Ownership of Our ADSs and Ordinary Shares
We do not know whether a market for our securities will be sustained and as a result it may be difficult for shareholders to sell our securities.
Although our ADSs trade on Nasdaq, an active trading market for the ADSs may not be sustained. It may be difficult for shareholders to sell their ADSs without depressing the market price for the ADSs. As a result of these and other factors, shareholders may not be able to sell their ADSs. Further, an inactive market may also impair our ability to raise capital by issuing securities and may impair our ability to enter into strategic partnerships or acquire companies or products by using our equity as consideration.
The requirements of being a publicly traded company may strain our resources and divert management’s attention.
As a publicly traded company, we have incurred, and will continue to incur, significant legal, accounting and other expenses that we did not incur as a private company. In addition, the Sarbanes-Oxley Act and the Dodd-Frank Wall Street Reform and Consumer Protection Act (the “Dodd-Frank Act”), as well as rules subsequently implemented by the SEC and Nasdaq under such acts have imposed various requirements on public companies. Shareholder activism, the current political environment and the current high level of government regulatory reform may lead to substantial new regulations and disclosure obligations, which may lead to additional compliance costs and impact the manner in which we operate our business in ways we cannot currently anticipate. Our management will need to devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations will increase our legal and financial compliance costs and will make some activities more time-consuming and costly. For example, we expect these rules and regulations to make it more difficult and more expensive for us to obtain director and officer liability insurance and we may be required to incur substantial costs to maintain our current levels of such coverage.
Failure to achieve and maintain effective internal controls in accordance with Section 404 of the Sarbanes-Oxley Act could have a material adverse effect on our business, results of operation or financial condition. In addition, current and potential shareholders could lose confidence in our financial reporting, which could have a material adverse effect on the price of the ADSs.
Effective internal controls are necessary for us to provide reliable financial reports and effectively prevent fraud. We are required to document and test our internal control procedures in order to satisfy the requirements of Section 404 of the Sarbanes-Oxley Act, which requires annual management assessments of the effectiveness of our internal control over financial reporting. In addition, if we fail to maintain the adequacy of our internal controls, as such standards are modified, supplemented or amended from time to time, we may not be able to ensure that we can conclude on an ongoing basis that we have effective internal controls over financial reporting in accordance with Section 404. Disclosing deficiencies or weaknesses in our internal controls, failing to remediate these deficiencies or weaknesses in a timely fashion or failing to achieve and maintain an effective internal control environment may cause investors to lose confidence in our reported financial information, which could have a material adverse effect on the price of the ADSs. If we cannot provide reliable financial reports or prevent fraud, our operating results could be harmed.
Our failure to meet the continued listing requirements of The Nasdaq Capital Market could result in a delisting of our ADSs.
Our ADSs are currently listed on the Nasdaq Capital Market. In order to maintain this listing, we must satisfy minimum financial and other continued listing requirements and standards, including a maintaining minimum closing bid price requirement for our ADSs of $1.00 per ADS as required by Nasdaq Listing Rule 5550(a)(2) (“Minimum Bid Price Requirement”). There can be no assurance that we will be able to comply with the applicable listing standards.
On April 29, 2024, we received a deficiency letter from the Listing Qualifications Department of Nasdaq notifying us that for the preceding 31 consecutive business days (March 14, 2024 through April 26, 2024), our ADSs did not maintain a minimum closing bid price of $1.00 per ADS as required by the Minimum Bid Price Requirement. In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we had a compliance period of 180 calendar days, or until October 28, 2024, to regain compliance with Nasdaq Listing Rule 5550(a)(2). On October 16, 2024, the Company submitted a letter to Nasdaq requesting an additional 180-day grace period to regain compliance with the Minimum Bid Price Requirement. On October 29, 2024, the Company received a letter from the Listing Qualifications Department of Nasdaq granting the Company an additional 180 calendar day grace period, or until April 28, 2025, to regain compliance. The Staff’s determination in granting the Company the extension was based on the Company meeting the continued listing requirement for market value of publicly held shares and all other applicable requirements for initial listing on the Nasdaq Capital Market with the exception of the Minimum Bid Price Requirement, and the Company’s written notice of its intention to cure the deficiency during the second compliance period by effecting a reverse split, if necessary. Compliance may be achieved without further action if the closing bid price of the Company’s ADS is at or above $1.00 for a minimum of ten consecutive business days at any time during the second
44
compliance period, in which case Nasdaq will notify the Company if it determines the Company is in compliance and the matter will be closed; however Nasdaq could require the closing bid price to equal or to exceed the $1.00 minimum bid price requirement for more than 10 consecutive business days before determining that the Company complies. If compliance cannot be demonstrated by April 28, 2025, the Staff will provide written notification that the Company’s securities will be delisted. At that time, the Company may appeal the Staff’s determination to a Hearings Panel.
If we cannot regain compliance with the Minimum Bid Price Requirement or if we otherwise fail to meet any of Nasdaq’s listing standards, our ADSs will be subject to delisting. If that were to occur, our ADSs would be subject to rules that impose additional sales practice requirements on broker-dealers who sell our securities. The additional burdens imposed upon broker-dealers by these requirements could discourage broker-dealers from effecting transactions in our ADSs. This would adversely affect the ability of investors to trade our ADSs and would adversely affect the value of our ADSs. Delisting from Nasdaq would cause us to pursue eligibility for trading of our ADSs on other markets or exchanges, or on an over-the-counter market. In such case, our shareholders’ ability to trade or obtain quotations of the market value of our ADSs would be severely limited because of lower trading volumes and transaction delays. These factors could contribute to lower prices and larger spreads in the bid and ask prices of these securities. There can be no assurance that our ADSs, if delisted from the Nasdaq, would be listed on a national securities exchange, a national quotation service or the over-the-counter markets. Delisting from the Nasdaq could also result in negative publicity, adversely affect the market liquidity of our ADSs, decrease securities analysts’ coverage of us or diminish investor, supplier and employee confidence.
The delisting of our ADSs from Nasdaq may make it more difficult for us to raise capital on favorable terms in the future, or at all. Such a delisting would likely have a negative effect on the price of our ADSs and would impair shareholders’ ability to sell or purchase our ADSs when they wish to do so. Further, if our ADSs were to be delisted from Nasdaq, our ADSs would cease to be recognized as a covered security, and we would be subject to additional regulation in each state in which we offer our securities. Moreover, there is no assurance that any actions that we take to restore our compliance with the Nasdaq Minimum Bid Price Requirement would stabilize the market price or improve the liquidity of our ADSs, prevent our ADSs from falling below the Nasdaq minimum bid price required for continued listing again or prevent future non-compliance with other applicable Nasdaq listing requirements, including maintaining minimum levels of stockholders’ equity or market values of our ADSs, our ADSs could be delisted.
The market price for our ADSs may be volatile.
The market price for our ADSs is likely to be highly volatile and subject to wide fluctuations in response to numerous factors including the following:
| ● | our failure to obtain the approvals necessary to commence clinical trials; |
| ● | results of clinical and preclinical studies; |
| ● | announcements of regulatory approval or the failure to obtain it, or changes or delays in the regulatory review process; |
| ● | announcements of new products or product enhancements by us or others; |
| ● | adverse actions taken by regulatory agencies with respect to our clinical trials, manufacturing supply chain or sales and marketing activities; |
| ● | changes or developments in laws, regulations or decisions applicable to our product candidates or patents; |
| ● | any adverse changes to our relationship with manufacturers or suppliers; |
| ● | announcements concerning our competitors or healthcare industries in general; |
| ● | achievement of expected product sales and profitability or our failure to meet expectations; |
| ● | our commencement of or results of, or involvement in, litigation, including, but not limited to, any product liability actions or intellectual property infringement actions; |
| ● | any major changes in our board of directors, management or other key personnel; |
45
| ● | announcements by us of significant strategic partnerships, out-licensing, in-licensing, joint ventures, acquisitions or capital commitments; |
| ● | expiration or terminations of licenses, research contracts or other collaboration agreements; |
| ● | public concern as to the safety of our products that we, our licensors or others develop; |
| ● | success of research and development projects; |
| ● | developments concerning intellectual property rights or regulatory approvals; |
| ● | variations in our and our competitors’ results of operations; |
| ● | changes in earnings estimates or recommendations by securities analysts; |
| ● | future issuances of ordinary shares, ADSs or other securities; |
| ● | general market conditions and other factors, including factors unrelated to our operating performance, such as natural disasters and political and economic instability, including wars, terrorism, political unrest, results of certain elections and votes, emergence of a pandemic, or other widespread health emergencies (or concerns over the possibility of such an emergency), boycotts, adoption or expansion of government trade restrictions, and other business restrictions; and |
| ● | the other factors described in this “Risk Factors” section. |
These factors and any corresponding price fluctuations may materially and adversely affect the market price of the ADSs, which would result in substantial losses by our investors. In addition, the securities market has from time to time experienced significant price and volume fluctuations that are not related to the operating performance of any particular company. These market fluctuations may also have a material adverse effect on the market price of the ADSs.
We may be at risk of securities class action litigation.
We may be at risk of securities class action litigation. This risk is especially relevant for us due to our dependence on positive clinical trial outcomes and regulatory approvals of our product candidates. In the past, medical, biotechnology and pharmaceutical companies have experienced significant stock price volatility, particularly when associated with such events such as clinical trials and product approvals. If we face such litigation, it could result in substantial costs, divert management’s attention and resources, and have a material adverse effect on our business, operating results and prospects.
Substantial future sales or perceived potential sales of our ADSs in the public market could cause the price of our ADSs decline.
Substantial sales of our ADSs on Nasdaq may cause the market price of our ADSs to decline. Sales by us or our security holders of substantial amounts of our ADSs or the perception that these sales may occur in the future, could cause a reduction in the market price of our ADSs. The issuance of any additional ordinary shares or any additional ADSs, or any securities that are exercisable for or convertible into our ordinary shares or ADSs, may have an adverse effect on the market price of our ADSs and will have a dilutive effect on our existing shareholders.
Our shareholders may experience substantial dilution as a result of future issuances of our equity securities.
We expect that significant additional capital will be needed in the future to continue our planned operations. To the extent we raise additional capital by issuing equity securities, our shareholders may experience substantial dilution. Pursuant to our equity incentive plan, the Board may grant options to employees, directors and consultants. We may sell ordinary shares represented by ADSs, convertible securities or other equity securities in one or more transactions at prices and in a manner we determine from time to time, any of which may result in material dilution to our existing shareholders. New investors could also be issued securities with rights superior to those of our existing shareholders.
46
We have not paid, and do not intend to pay, dividends on our ordinary shares and, therefore, unless our traded securities appreciate in value, our investors may not benefit from holding our securities.
We have not paid any cash dividends on our ordinary shares, and we do not anticipate paying any cash dividends on our ordinary shares in the foreseeable future. Moreover, the Israeli Companies Law, 5759-1999 (the “Companies Law”) imposes certain restrictions on our ability to declare and pay dividends. As a result, investors in our ADSs or ordinary shares will not be able to benefit from owning these securities unless their market price becomes greater than the price paid by such investors and they are able to sell such securities. We cannot assure you that shareholders will ever be able to resell our securities at a price in excess of the price paid.
If we pay dividends or other distributions, an ADS holder may not receive the same distributions or dividends as those we make to the holders of our ordinary shares, and, in some limited circumstances, an ADS holder may not receive dividends or other distributions on our ordinary shares and an ADS holder may not receive any value for them, if it is illegal or impractical to make them available to such ADS holder.
The depositary for the ADSs has agreed to pay to the ADSs holders the cash dividends or other distributions it or the custodian receives on ordinary shares or other deposited securities underlying the ADSs, after deducting its fees and expenses. ADS holders will receive these distributions, if any, in proportion to the number of ordinary shares their ADSs represent. However, the depositary is not responsible if it decides that it is unlawful or impractical to make a distribution available to any holders of ADSs. For example, it would be unlawful to make a distribution to a holder of ADSs if it consists of securities that require registration under the Securities Act, but that are not properly registered or distributed under an applicable exemption from registration. In these cases, the depositary may determine not to distribute such property and hold it as “deposited securities” or may seek to effect a substitute dividend or distribution, including net cash proceeds from the sale of the dividends that the depositary deems an equitable and practicable substitute. We have no obligation to register under U.S. securities laws any ADSs, ordinary shares, rights or other securities received through such distributions. We also have no obligation to take any other action to permit the distribution of ADSs, ordinary shares, rights or anything else to holders of ADSs. In addition, the depositary may withhold from such dividends or distributions its fees and an amount on account of taxes or other governmental charges to the extent the depositary believes it is required to make such withholding. This means that holders of ADSs may not receive the same distributions or dividends as those we make to the holders of our ordinary shares, and, in some limited circumstances, holders of ADSs may not receive any value for such distributions or dividends if it is illegal or impractical for us to make them available to such holders. These restrictions may cause a material decline in the value of the ADSs.
Holders of ADSs must act through the depositary to exercise their rights.
Holders of ADSs do not have the same rights as holders of our ordinary shares and may only exercise the voting rights with respect to the underlying ordinary shares in accordance with the provisions of the deposit agreement for the ADSs. Under Israeli law and our articles of association, the minimum notice period required to convene a shareholders meeting is not less than 35 or 21 calendar days, depending on the proposals on the agenda for the shareholders meeting. When a shareholder meeting is convened, holders of the ADSs may not receive sufficient notice of a shareholders meeting to permit them to withdraw their ordinary shares to allow them to cast their vote with respect to any specific matter. In addition, the depositary and its agents may not be able to send voting instructions to holders of the ADSs or carry out their voting instructions in a timely manner. We will make all reasonable efforts to cause the depositary to extend voting rights to holders of the ADSs in a timely manner, but we cannot assure holders that they will receive the voting materials in time to ensure that they can instruct the depositary to vote their ADSs. Furthermore, the depositary and its agents will not be responsible for any failure to carry out any instructions to vote, for the manner in which any vote is cast or for the effect of any such vote. As a result, holders of the ADSs may not be able to exercise their right to vote and they may lack recourse if their ADSs are not voted as they requested.
Holders of ADSs may be subject to limitations on the transfer of their ADSs.
ADSs are transferable on the books of the depositary. However, the depositary may close its transfer books at any time or from time to time when it deems expedient in connection with the performance of its duties. In addition, the depositary may refuse to deliver, transfer or register transfers of ADSs generally when our books or the books of the depositary are closed, or at any time if we or the depositary deems it advisable to do so because of any requirement of law or of any government or governmental body, or under any provision of the deposit agreement, or for any other reason in accordance with the terms of the deposit agreement.
Item 1B. Unresolved Staff Comments
None.
47
Item 1C. Cybersecurity
We depend on a variety of information systems and technologies (including cloud technologies) to manage our business. We
The underlying processes and controls of our cyber risk management program incorporate recognized best practices and standards for cybersecurity and information technology, including the National Institute of Standards and Technology (“NIST”) Cybersecurity Framework (“CSF”). We have undertaken, to conduct an annual assessment of our cyber risk management program and controls to identify, quantify, and categorize material cyber risks. In addition, we have developed a risk mitigation plan to address such risks, and where necessary, remediate potential vulnerabilities identified through the annual assessment process.
In addition, we maintain policies over areas such as information security, access on/offboarding, and access and account management, to help govern the processes put in place by management designed to protect our IT assets, data, and services from threats and vulnerabilities. Our cybersecurity risk management strategy and infrastructure includes maintenance of an IT assets inventory, periodic vulnerability scanning, identity access management controls including restricted access of privileged accounts, network integrity safeguarded by employing web-based software, industry-standard encryption protocols, critical data backups, infrastructure maintenance, incident response, cybersecurity strategy, and cyber risk advisory, assessment and remediation.
We face risks from cybersecurity threats that could have a material adverse effect on our business, financial condition, results of operations, cash flows or reputation. We acknowledge that the risk of cyber incident is prevalent in the current threat landscape and that a future cyber incident may occur in the normal course of our business. To date,
Item 2. Properties
We do not own any property, and we do not have any contracts or options to acquire or lease any property in the future. We are operating out of a virtual office, which is adequate for our present and planned future operations, as our corporate staff has been working remotely.
Item 3. Legal Proceedings
From time to time, we may become involved in legal proceedings or be subject to claims arising in the ordinary course of our business. We are currently not a party to any material legal or administrative proceedings, and we are not aware of any pending or threatened material legal or administrative proceedings against us.
Item 4. Mine Safety Disclosures
Not applicable.
48
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information
Our ADSs are currently listed on The Nasdaq Capital Market under the symbol “QNRX,” with each ADS representing one ordinary share.
Holders
As of March 10, 2025, our ADSs were held by 7 holders of record, and our ordinary shares were held by 5 holders of record. Bank of New York Mellon (“BNY”) is the depositary for our ADR program, and Computershare Trust Company, N.A. is our transfer agent. The number of record holders was determined from the records of our depositary and transfer agent and does not include beneficial owners of ADSs or ordinary shares whose shares are held in the names of various securities brokers, dealers and registered clearing agencies.
Dividends
We have never declared or paid any dividends on our ordinary shares. We do not anticipate paying any dividends in the foreseeable future. We currently intend to retain future earnings, if any, to finance operations and expand our business. Our board of directors has sole discretion whether to pay dividends. If our board of directors decides to pay dividends, the form, frequency and amount will depend upon our future operations and earnings, capital requirements and surplus, general financial condition, contractual restrictions and other factors that our directors may deem relevant. The Companies Law imposes restrictions on our ability to declare and pay dividends.
Equity Compensation Plan Table
The information included in our Equity Compensation Plan Table under Item 12 of Part III of this Annual Report is hereby incorporated by reference into this Item 5 of Part II of the Annual Report.
Item 6. [Reserved]
49
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of our financial condition and results of operations in conjunction with our audited consolidated financial statements and related notes to those statements included in this Annual Report. Our consolidated financial statements have been prepared in accordance with generally accepted accounting principles in the United States (“GAAP”), reflect the operations of Quoin Pharmaceuticals Inc. (“Quoin Inc.”) since inception and include the accounts of Quoin Ltd. since the closing of the Merger (as defined below). In addition to historical financial information, the following discussion contains forward-looking statements that reflect our plans, estimates, beliefs and expectations that involve risks and uncertainties. Our actual results and the timing of events could differ materially from those discussed in these forward-looking statements. Important factors that could cause or contribute to these differences include those discussed below and elsewhere in this Annual Report, particularly in Part I, Item 1A. “Risk Factors” and the section entitled “Cautionary Note Regarding Forward-Looking Statements and Summary of Risk Factors.”
Overview
We are a late-stage clinical specialty pharmaceutical company focused on the development and commercialization of therapeutic products that treat rare and orphan diseases for which there are currently no approved treatments or cures. Our initial focus is on the development of products, using our proprietary owned and in-licensed drug delivery technologies, that could help address rare genetic diseases. Our lead product, QRX003, is under clinical development as a potential treatment for Netherton Syndrome, a rare hereditary genetic disease. QRX003 is currently being tested in three regulatory clinical studies under an open IND application with the FDA. We have opened five clinical sites in the US and intend to open a sixth clinical site at Northwestern University. We are expanding our trials internationally into the Middle East, the United Kingdom and additional countries in Western and Eastern Europe. QRX003 is currently being tested in a pediatric NS patient at the Children’s Hospital in Dublin, Ireland and we intend to expand this study to include additional children with NS in Spain, the United Kingdom and potentially other countries. QRX003 is also being developed as a potential treatment for Peeling Skin Syndrome with the first subject being treated in New Zealand. In addition, we entered into two separate Research Agreements with the Queensland University of Technology, under which we have obtained an option for global licenses to QRX007 for the potential treatment of NS and QRX008 for the potential treatment of scleroderma, as well as a Research Agreement with the University College Cork for the development of novel topical formulations of Rapamycin (sirolimus) as potential treatments for a number of rare and orphan diseases for which there are currently no approved therapies or cures. Other development products in our pipeline include QRX004 as a potential treatment for Recessive Dystrophic Epidermolysis Bullosa.
Our mission is to develop and commercialize proprietary therapeutic drug products that treat rare and orphan diseases, particularly those where none currently exists. To achieve this, we plan to:
| ● | complete the late-stage clinical testing of QRX003 in NS and, if successful, file for marketing approval in the United States and other territories; |
| ● | prepare to commercialize QRX003 by establishing our own sales infrastructure in the U.S. and Europe and entering into distribution partnerships in other territories such as those currently established for Canada, Australia/New Zealand, the Middle East, China, Hong Kong, Taiwan, Latin America, Central and Eastern Europe, Turkey and Singapore; and |
| ● | pursue business development activities by seeking partnering, licensing, merger and acquisition opportunities or other transactions to further expand our pipeline and drug-development capabilities. |
To date, no products have been commercialized and no revenue has been generated. We do not expect to generate revenue from product sales unless and until we successfully complete development and obtain marketing approval for one or more of our product candidates, which we expect will take a number of years and is subject to significant uncertainty. Accordingly, we will need to raise additional capital prior to the commercialization of QRX003 or any other product candidate. Until such time, if ever, as we can generate substantial revenue from product sales, we expect to finance our operating activities through a combination of equity offerings, debt financings, government or other third-party funding, commercialization, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. Our failure to raise capital or enter into such other arrangements as and when needed would have a negative impact on our financial condition and our ability to continue our operations. See “Liquidity and Capital Resources”.
50
Key Events
Public Offerings
On February 24, 2023 (the “2023 Closing Date”), we completed an offering (the “2023 Offering”) of 412,500 ordinary shares represented by 412,500 ADSs at a purchase price of $12.00 per ADS and a pre-funded warrant (the “2023 Pre-Funded Warrant”) to purchase 170,833 ordinary shares represented by 170,833 ADSs at a per pre-funded warrant price of $11.9988, with each ADS and 2023 Pre-Funded Warrant accompanied by an ordinary warrant (the “2023 Ordinary Warrant”) for aggregate gross proceeds of $7.0 million, resulting in net proceeds of approximately $5.8 million, after deducting the placement agent’s fees and offering expenses paid by us, and excluding the proceeds, if any, from the subsequent exercise of the 2023 Ordinary Warrants. Each 2023 Ordinary Warrant has an exercise price of $12.00 per ADS and expires on the fifth anniversary of the 2023 Closing Date. On the 2023 Closing Date, the holder of the 2023 Pre-Funded Warrant exercised its Pre-Funded Warrant in full.
In connection with the 2023 Offering, we entered into an Amendment No. 1 to Warrant to Purchase Ordinary Shares Represented by American Depositary Shares, dated February 24, 2023 (collectively, the “2023 Warrant Amendments”), with each of the purchasers (the “2022 Purchasers”) who participated in both our August 2022 public offering (the “2022 Offering”) and the 2023 Offering. The 2023 Warrant Amendments amended certain terms of the ordinary warrants issued to such 2022 Purchasers in the 2022 Offering. Specifically, the 2023 Warrant Amendments reduced the exercise price of such warrants to $13.20 and extended the term during which those warrants could remain exercisable until February 24, 2028.
On March 7, 2024, we completed an offering (the “March 2024 Offering”) of the following securities (i) 811,250 ordinary shares represented by ADSs, (ii) 4,062,500 Series D warrants (the “Series D Warrants”) to purchase 4,062,500 ordinary shares represented by ADSs, (iii) 4,062,500 Series E warrants (the “Series E Warrants”) to purchase 4,062,500 ordinary shares represented by ADSs, and (iv) 3,251,250 pre-funded warrants (the “March 2024 Pre-Funded Warrants”) to purchase 3,251,250 ordinary shares represented by ADSs for aggregate gross proceeds of approximately $6.5 million, resulting in net proceeds of approximately $5.5 million, after deducting the placement agent’s fees and offering expenses paid by us. Each ADS (or March 2024 Pre-Funded Warrant to purchase one ADS in lieu thereof) was sold together with a Series D Warrant to purchase one ADS and a Series E Warrant to purchase one ADS. The ADSs and accompanying Series D Warrants and the Series E Warrants were sold at a combined public offering price of $1.60 and the March 2024 Pre-Funded Warrants and accompanying Series D Warrants and the Series E Warrants were sold at a combined public offering price of $1.5999, which is equal to the combined purchase price per ADS and accompanying Series D Warrants and the Series E Warrants, minus the exercise price of each March 2024 Pre-Funded Warrant of $0.0001. As of December 31, 2024 all March 2024 Pre-Funded Warrants have been exercised and are included in issued and outstanding ADSs. The Series D Warrants and the Series E Warrants have an exercise price of $1.60 per share, were exercisable immediately following the closing of the March 2024 Offering and expire in two years and five years, respectively, from the closing of the March 2024 Offering.
On March 7, 2024, we also entered into privately negotiated agreements with the holders of certain existing outstanding warrants to purchase up to 638,834 ADSs to, among other things, reduce the exercise price of such warrants to $1.60 and to extend the expiration date of such warrants until March 7, 2029. The incremental fair value of the modified warrants was approximately $209,000, which was accounted for as an offering expense in connection with the March 2024 Offering.
51
On December 23, 2024, we completed an offering (the “December 2024 Offering” and, together with the March 2024 Offering, the “2024 Offerings”) of the following securities (i) 3,137,778 ordinary shares represented by ADSs, (ii) 15,111,110 Series F warrants (the “Series F Warrants”) to purchase 15,111,110 ordinary shares represented by ADSs, (iii) 15,111,110 Series G warrants (the “Series G Warrants” and together with the Series F Warrants, the “December 2024 Warrants”) to purchase 15,111,110 ordinary shares represented by ADSs, and (iv) 11,973,332 pre-funded warrants (the “December 2024 Pre-Funded Warrants”) to purchase 11,973,332 ordinary shares represented by ADSs for aggregate gross proceeds of approximately $6.8 million, resulting in net proceeds of approximately $5.8 million, after deducting the placement agent’s fees and offering expenses paid by us. Each ADS (or December 2024 Pre-Funded Warrant to purchase one ADS in lieu thereof) was sold together with a Series F Warrant to purchase one ADS and a Series G Warrant to purchase one ADS. The ADSs and accompanying Series F Warrants and Series G Warrants were sold at a combined public offering price of $0.45 and the December 2024 Pre-Funded Warrants and accompanying Series F Warrants and Series G Warrants were sold at a combined public offering price of $0.4499, which is equal to the combined purchase price per ADS and accompanying Series F Warrants and Series G Warrants, minus the exercise price of each December 2024 Pre-Funded Warrant of $0.0001. As of December 31, 2024, 760,666 December 2024 Pre-Funded Warrants, have been exercised and are included in issued and outstanding ADSs. The December 2024 Pre-Funded Warrants were immediately exercisable upon issuance and may be exercised at any time until exercised in full. The Series F Warrants and the Series G Warrants have an exercise price of $0.45 per share, were exercisable immediately upon issuance and expire in two years and five years, respectively, from the closing of the December 2024 Offering. In January and February 2025, certain investors in the December 2024 Offering exercised (i) the remaining outstanding 11,212,666 December 2024 Pre-Funded Warrants, (ii) 320,000 Series F Warrants, and (iii) 105,000 Series G Warrants.
In connection with the 2024 Offerings, we entered into Securities Purchase Agreements (the “2024 Purchase Agreements”) dated March 7, 2024 and December 23, 2024, respectively, with certain institutional investors signatory thereto, pursuant to which we agreed to issue and sell to such investors, certain of the ADSs, pre-funded warrants and ordinary warrants sold in the 2024 Offerings. Pursuant to the terms of each of the 2024 Purchase Agreements, we agreed, subject to certain exceptions, (i) to not enter into variable rate financings for a period of 180 days following the closing of such 2024 Offering, and (ii) to not enter into any equity financings for 90 days from closing of such 2024 Offering.
Certain of our officers and directors purchased an aggregate of 1,333,333 of our ADSs and accompanying December 2024 Warrants to purchase 2,666,666 of our ADSs, for a total purchase price of approximately $600,000, at the public offering price and on the same terms as the other purchasers in the December 2024 Offering.
On December 20, 2024, we also entered into privately negotiated agreements with the holders of certain existing outstanding warrants to purchase up to 7,002,500 ADSs to, among other things, reduce the exercise price of such warrants to $0.45 and to extend the expiration date of such warrants until December 23, 2029. The incremental fair value of the modified warrants was approximately $1.5 million, which was accounted for as an offering expense in connection with the December 2024 Offering.
Alumni Equity Line and Purchase Agreement
On January 25, 2024, we entered into a Purchase Agreement (the “Alumni Purchase Agreement”) with Alumni Capital LP (“Alumni”). Pursuant to the Alumni Purchase Agreement, we have the right to sell to Alumni up to $8,000,000 (the “Commitment Amount”) of newly issued ordinary shares that are represented by ADS (the “Purchase Notice Securities”), subject to certain conditions and limitations, from time to time during the term of the Alumni Purchase Agreement.
We do not have the right to commence any sales of ordinary shares represented by ADSs to Alumni under the Alumni Purchase Agreement until the date, which we refer to as the Commencement Date, that all of the conditions set forth in the Alumni Purchase Agreement have been satisfied, including that the registration statement we agreed to file with the SEC pursuant to the Alumni Purchase Agreement is declared effective by the SEC, and our shareholders have approved of the issuance of ADSs under the Alumni Purchase Agreement, which approval was obtained on April 5, 2024.
From and after the Commencement Date, we may, from time to time and at our sole discretion for a period of three months, which we at our sole discretion may increase by an additional three months (such period, including any extension, the “Commitment Period”), on any business day that we select, direct Alumni to purchase ordinary shares represented by ADSs. The purchase price for the ordinary shares represented by ADSs we may sell to Alumni will be based upon formulas set forth in the Alumni Purchase Agreement based on the then current market price of the ADSs as computed under the Alumni Purchase Agreement and will depend on the type of purchase notice we submit to Alumni from time to time. There is no upper limit on the price per share that Alumni could be obligated to pay for the ADSs under the Alumni Purchase Agreement; provided, however at no time can the purchase price be below a floor price of $1.00
52
per share (subject to adjustment). We agreed to issue purchase notices for an aggregate of at least $4,000,000 of the Commitment Amount prior to the end of the Commitment Period.
As consideration for Alumni’s irrevocable commitment to purchase ADSs under the Alumni Purchase Agreement, we agreed to issue to Alumni, at the times set forth in the Alumni Purchase Agreement beginning with the trading day after the Commencement Date, a number of ADSs with a value at the time of issuance not to exceed $240,000 in the aggregate (the “Commitment Securities”). The ADSs to be issued will be valued at the average of the closing prices of the ADSs on Nasdaq for the five trading days immediately prior to the date such ADSs are issued. We may pay cash in lieu of issuing all or any portion of the Commitment Securities.
Per mutual agreement between the parties, we have not filed the required registration statement or sold any ADSs to Alumni under the Alumni Purchase Agreement. The Company is restricted from selling ADSs under the Alumni Purchase Agreement for 180 days from the closing of the December 2024 Offering, see above.
Nasdaq Listing
On April 29, 2024, we received a letter from the Listing Qualifications Department of Nasdaq (notifying us that the closing bid price per ADS of the Company was below the required minimum of $1.00 for a period of 31 consecutive business days and that we did not meet the minimum bid price requirements set forth in Nasdaq Rule 5550(a)(2).
Pursuant to Nasdaq Rule 5810(c)(3)(A), we had a period of one hundred eighty (180) calendar days, or until October 28, 2024, to regain compliance with Nasdaq’s minimum bid price requirement. On October 16, 2024, the Company submitted a letter to Nasdaq requesting an additional 180-day grace period to regain compliance with the minimum bid price requirement. On October 29, 2024, the Company received a letter from the Listing Qualifications Department of Nasdaq granting the Company an additional 180 calendar day grace period, or until April 28, 2025, to regain compliance. The Staff’s determination in granting the Company the extension was based on the Company meeting the continued listing requirement for market value of publicly held shares and all other applicable requirements for initial listing on the Nasdaq Capital Market with the exception of the minimum bid price requirement, and the Company’s written notice of its intention to cure the deficiency during the second compliance period by effecting a reverse split, if necessary. Accordingly, there is no immediate effect on the listing or trading of the Company’s ADSs on the Nasdaq Capital Market under the symbol “QNRX.”
Compliance may be achieved without further action if the closing bid price of the Company’s ADS is at or above $1.00 for a minimum of ten consecutive business days at any time during the second compliance period, in which case Nasdaq will notify the Company if it determines the Company is in compliance and the matter will be closed; however Nasdaq could require the closing bid price to equal or to exceed the $1.00 minimum bid price requirement for more than 10 consecutive business days before determining that the Company complies. If compliance cannot be demonstrated by April 28, 2025, the Staff will provide written notification that the Company’s securities will be delisted. At that time, the Company may appeal the Staff’s determination to a Hearings Panel. If the Company appeals it will be asked to provide a plan to regain compliance to the Panel. Historically, Panels have generally viewed a near-term reverse split as the only definitive plan acceptable to resolve a minimum bid price deficiency.
The Company intends to actively monitor the bid price of its ADSs and will consider available options to regain compliance with Nasdaq’s listing requirements.
Components of Our Results of Operations
Operating Expenses
Our current operating expenses consist of two components - research and development expenses, and general and administrative expenses.
Research and Development Expenses
Research and development costs are expensed as incurred. Research and development expenses include personnel costs associated with research and development activities, including third-party contractors to perform research, conduct clinical trials and manufacture
53
drug supplies and materials. We utilize outside consultants and third parties to conduct the majority of our research and development, under the supervision of our management team.
Future research and development expenses may include:
| ● | employee-related expenses, such as salaries, bonuses and benefits, consultant-related expenses, share-based compensation, overhead related expenses and travel related expenses for our research and development personnel; |
| ● | expenses incurred under agreements with CROs, as well as consultants that support the implementation of the clinical studies described above; |
| ● | manufacturing and packaging costs in connection with conducting clinical trials and for stability and other studies required to support the NDA filing as well as manufacturing drug product for commercial launch; |
| ● | formulation, research and development expenses related to QRX003; and other product candidates we may choose to develop; and |
| ● | costs for sponsored research. |
Research and development activities will continue to be central to our business plan. Product candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical trials. We expect our research and development expenses to be significant over the next several years as personnel and compensation costs increase and we conduct late-stage clinical studies and prepare to seek regulatory approval for QRX003 and any other future product candidate.
The duration, costs and timing of clinical trials of QRX003 and any other future product candidate will depend on a variety of factors that include, but are not limited to:
| ● | the number of trials required for approval; |
| ● | the per patient trial costs; |
| ● | the number of patients that participate in the trials; |
| ● | the number of sites included in the trials; |
| ● | the countries in which the trial is conducted; |
| ● | the length of time required to enroll eligible patients; |
| ● | the number of doses that patients receive; |
| ● | the drop-out or discontinuation rates of patients; |
| ● | the potential additional safety monitoring or other studies requested by regulatory agencies; |
| ● | the duration of patient follow-up; |
| ● | the timing and receipt of regulatory approvals; and |
| ● | the efficacy and safety profile of our product candidates. |
54
General and Administrative Expenses
General and administrative expenses consist primarily of compensation and employee related expenses including non-cash stock-based compensation, professional fees and other corporate expenses. We anticipate that our general and administrative expenses will increase in the future to support our continued research and development activities. These increases will likely include compensation and employee-related expenses including stock-based compensation, increased costs related to the potential hiring of personnel, travel costs and fees to outside consultants, lawyers and accountants.
Other Expenses (income)
Other expenses (income) consist primarily of interest income and unrealized loss (gain) on investments.
Results of Operations - Year ended December 31, 2024 compared to Year ended December 31, 2023
The following table sets forth our results of operations for the year ended December 31, 2024, compared to the year ended December 31, 2023:
Year ended December 31, | |||||||||
| 2024 |
| 2023 |
| Change | ||||
Operating Expenses |
|
|
|
|
|
| |||
General and administrative | $ | 5,925,833 | $ | 6,070,517 | $ | (144,684) | |||
Research and development |
| 3,602,632 |
| 3,307,987 |
| 294,645 | |||
Total operating expenses |
| 9,528,465 |
| 9,378,504 |
| 149,961 | |||
Other (income) and expenses |
|
|
|
|
|
| |||
Unrealized gain (loss) | (7,502) | 2,683 | (10,185) | ||||||
Realized and accrued interest income |
| (558,491) |
| (694,614) |
| 136,123 | |||
Total other income |
| (565,993) |
| (691,931) |
| 125,938 | |||
Net loss | $ | (8,962,472) | $ | (8,686,573) | $ | (275,899) | |||
General and Administrative Expenses
General and administrative expenses were approximately $5,926,000 and $6,071,000, in the year ended December 31, 2024 and 2023, respectively, representing a decrease of $145,000, or 2.4%. The decrease was primarily due to a decrease in travel allocated to general and administrative expenses of $168,000, a decrease in insurance of $124,000, offset with an increase in payroll and benefits of $105,000, an increase in legal fees and other public company expenses of $49,000, and an increase in non-cash stock-based compensation expense of $24,000.
Research and Development Expenses
Our research and development expenses during the year ended December 31, 2024 and 2023 were approximately $3,603,000 and $3,308,000, respectively, representing an increase of $295,000, or approximately 8.9%. The increase was primarily due to an increase of $176,000 worth of expenditures on our development programs, including work related to the clinical studies for the development of QRX003, manufacturing costs for material used in our clinical studies an increased allocation of internal compensation and travel costs to our research and development programs. The increase also included approximately $140,000 in non-cash stock-based compensation expense. We expect to continue our research and development efforts by conducting the remaining studies necessary for the development and approval of QRX003, see “Components of Our Results of Operations - Research and Development Expenses” above.
We amortize licensed or acquired intellectual property over its expected useful life, included in research and development expenses set out above. The license from Skinvisible was obtained in October 2019, see “Research and Development, Patents and Licenses.” Amortization of intangible assets was approximately $100,000 and $104,000 in each of the years ended December 31, 2024 and 2023. As of December 31, 2023 we determined that the Polytherapeutics asset was no longer of use and reduced the carrying value to zero, incurring an impairment charge of approximately $18,000.
55
Other Expenses:
Interest and financing expense
We earned approximately $558,000 in interest income and approximately $7,500 in unrealized gain, and earned approximately $695,000 in interest income and incurred approximately $3,000 in unrealized loss, in the year ended December 31, 2024 and December 31 2023, respectively, from our cash and cash equivalents and investments in marketable debt securities. The decrease in interest income in the year ending December 31, 2024 is the result of lower average aggregate cash and investment balances and a decrease in interest rates.
Liquidity and Capital Resources
We have incurred net losses every year since inception. We believe that we have sufficient resources to effect our business plan for at least one year from the issuance of the consolidated financial statements included in this report; however, the Company is subject to risks common to development stage biopharmaceutical companies including, but not limited to, unanticipated clinical trial costs and the ability to estimate such occurrences, if any, on the Company’s cash, liquidity, additional financing requirements, and availability. Accordingly, we may need to raise additional funds sooner than planned. We do not expect to generate revenue from product sales unless and until we successfully complete development and obtain marketing approval for one or more of our product candidates, which we expect will take a number of years and is subject to significant uncertainty. Additional financing will be required to complete the research and development of our therapeutic targets and our other operating requirements, which may not be available at acceptable terms, if at all. If we are unable to obtain additional funding when it becomes necessary, the development of our product candidates will be impacted and we would likely be forced to delay, reduce, or terminate some or all of our development programs, all of which could have a material adverse effect on our business, results of operations and financial condition.
Our net losses may fluctuate significantly from quarter-to-quarter and year-to-year, depending on the timing of planned clinical trials and our expenditures on other research and development activities.
Future Funding Requirements
We will need to obtain further funding through public or private offerings of our capital stock, debt financing, collaboration and licensing arrangements or other sources, the requirements for which will depend on many factors, including:
| ● | the scope, timing, rate of progress and costs of our drug development efforts, preclinical development activities, the timing of laboratory testing and clinical trials for our product candidates; |
| ● | the number and scope of clinical programs we decide to pursue; |
| ● | the cost, timing and outcome of preparing for and undergoing regulatory review of our product candidates; |
| ● | the scope and costs of development and commercial manufacturing activities; |
| ● | the cost and timing associated with commercializing our product candidates, if they receive marketing approval; |
| ● | the extent to which we acquire or in-license other product candidates and technologies; |
| ● | the costs of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending intellectual property-related claims; |
| ● | our ability to establish and maintain collaborations on favorable terms, if at all; |
| ● | our efforts to enhance operational systems and our ability to attract, hire and retain qualified personnel, including personnel to support the development of our product candidates and, ultimately, the sale of our products, following FDA approval; |
| ● | our implementation of operational, financial and management systems; and |
56
| ● | the costs associated with being a public company. |
Adequate additional funding may not be available to us on acceptable terms, or at all. If we are unable to raise capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, scale back or discontinue the development or commercialization of QRX003, any future product candidate, or potentially discontinue operations.
To the extent that we raise additional capital through the sale of our equity or convertible debt securities, and pursuant to the exercise of the warrants issued to our investors in the 2022 Offering, the 2023 Offering and the 2024 Offerings, the ownership interest of our equity holders will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our equity holders. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends.
If we raise additional funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, we may be required to relinquish valuable rights to our technologies, future revenue streams, research programs or proposed products, or to grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our drug development or future commercialization efforts or grant rights to develop and market any future product that we would otherwise prefer to develop and market ourselves.
Summary Statement of Cash Flows – Year ended December 31, 2024 compared to Year ended December 31, 2023
As of December 31, 2024, we had approximately $14,057,000 in cash and investments in marketable securities. The table below presents our cash flows for the year ended December 31, 2024 and 2023:
Year ended December 31, | ||||||
| 2024 |
| 2023 | |||
Net cash used in operating activities | $ | (7,857,309) | $ | (7,864,429) | ||
Net cash provided (used) in investing activities |
| (1,888,282) |
| 2,188,316 | ||
Net cash provided by financing activities |
| 10,967,736 |
| 5,216,683 | ||
Net change in cash and cash equivalents | $ | 1,222,145 | $ | (459,430) | ||
Operating Activities
Net cash used in operating activities was approximately $7,857,000 and $7,864,000 for the year ended December 31, 2024 and 2023, respectively. The decrease for the year ended December 31, 2024 was primarily due to an increase in non-cash based stock compensation and an increase in accounts payable and accrued expenses, offset by an increase in operating expense and an increase in prepaid expenses and other assets.
Investing Activities
Net cash used in investing activities in the year ended December 31, 2024 was approximately $1,888,000 and net cash provided by investing activities in the year ended December 31, 2023 was approximately $2,188,000 in each period consisting of net proceeds from maturity and purchases of short maturity US Treasury Bills and Notes.
Financing Activities
Net cash provided by financing activities was approximately $10,968,000 for the year ended December 31, 2024, consisting of approximately $11,568,000 in net proceeds from the March 2024 and December 2024 Offerings, partially offset by repayments of amounts due to officers of $600,000. Net cash provided by financing activities was approximately $5,217,000 for the year ended December 31, 2023, consisting of approximately $5,849,000 in net proceeds from the 2023 Offering partially offset by repayments of amounts due to officers of $600,000, and $33,000 in deferred financing costs.
57
Research and Development Commitments
In October 2019, Quoin Inc. entered into the Exclusive Licensing Agreement (as amended from time to time, the “License Agreement”) with Skinvisible Pharmaceuticals, Inc. (“Skinvisible”), under which Skinvisible granted us an exclusive royalty-bearing license relating to the production and manufacture of prescription drug products related to certain patents held by Skinvisible, including those related to QRX003 and QRX004. We made Skinvisible a one-time non-refundable, non-creditable license fee of $1 million (the “License Fee”). In addition, we agreed to pay Skinvisible a single digit royalty percentage of our net sales revenues for any licensed product covered by the patent rights licensed under the License Agreement. We also agreed to pay Skinvisible 25% of any revenues we receive as royalties in the event that we sublicense any licensed products to a third party. The License Agreement also requires that we make a $5 million payment to Skinvisible upon receiving approval in the U.S. or European Union, whichever occurs first, for the first drug product developed using intellectual property licensed thereunder.
In November 2020, Quoin Inc. entered into a Master Service Agreement with Therapeutics Inc. for the management of the preclinical and clinical development of QRX003 for Netherton Syndrome. The initial term of the agreement was three years with automatic one year extensions, and the agreement required the execution of individual work orders. Quoin Inc. may terminate any work order for any reason with 90 days written notice subject to costs incurred through termination and a defined termination fee, unless there is a material breach by Therapeutics Inc. A work order was entered into in June 2022 for the first QRX003 clinical study at an expected estimated cost of approximately $4.4 million through 2024. An additional change order was entered into in December 2022 for a second QRX003 clinical study at an expected estimated cost of approximately $830,000. An amended and restated change order for the two studies was entered into in December 2024 at an estimated total remaining cost from August 2024 of approximately $3.6 million for the two studies combined. In the years ended December 31, 2024 and 2023, we incurred research and development costs under these agreements of approximately $1.1 million and $1.5 million, respectively. During the year ended December 31, 2024, the Company received a credit of approximately $83,000 applied to prior expenses incurred during the period of January 2024 to March 2024. During the year ended December 31, 2023, the Company received a credit of approximately $278,000 applied to prior expenses incurred during the period of March 2023 to July 2023.
In November 2021, we entered into a research agreement with Queensland University of Technology (QUT) for a pre-clinical research program for the development of a product to treat Netherton Syndrome of approximately $250,000. In May 2022, we entered into a second research agreement with QUT for the development of a product to treat Scleroderma of approximately $610,000. Each agreement remains in place until the completion of the research program, which in each case was initially anticipated to be 18 months from execution. For the years December 31, 2024 and 2023, we incurred research and development costs related to these agreements of approximately $0 and $361,000 respectively. Quoin is planning to schedule a meeting with QUT to discuss the future direction of both research programs.
On June 10, 2024, we entered into a research agreement with The School of Pharmacy at UCC. The scope of the agreement encompasses the development of novel topical formulations of Rapamycin (sirolimus) as potential treatments for a number of rare and orphan diseases for which there are currently no approved therapies or cures. Under the terms of the agreement, based on the achievement of certain milestones, we will fund up to approximately €567,000 ($608,000) plus VAT over an anticipated 2-1/2 year period to support the UCC research program to investigate the development of a number of topical rapamycin formulations for future development as potential treatments for several rare and orphan diseases. Following completion of the research program, we will have the option to advance the clinical development of rapamycin formulations developed by UCC. Work on this research project commenced in December 2024 and we accrued $10,000 in research and development costs for the year ended December 31, 2024.
Critical Accounting Estimates
Critical accounting estimates are those that, in management’s view, are most important to the portrayal of a company’s financial condition and results of operations and most demanding on their calls on judgment, often as a result of the need to make estimates about the effect of matters that are inherently uncertain and may change in subsequent periods. We believe our most critical accounting estimates relate to:
58
Research and Development
Research and development costs are expensed as incurred. Research and development expenses include personnel costs associated with research and development activities, including third-party contractors to perform research, conduct clinical trials and manufacture drug supplies and materials. We accrue for costs incurred by external service providers, including contract research organizations and clinical investigators, based on its estimates of service performed and costs incurred. These estimates include the level of services performed by third parties, patient enrollment in clinical trials when applicable, administrative costs incurred by third parties, and other indicators of the services completed. Based on the timing of amounts invoiced by service providers, we may also record payments made to those providers as prepaid expenses that will be recognized as expense in future periods as the related services are rendered.
Stock based compensation:
We recognize compensation costs resulting from the issuance of stock-based awards to employees, non-employees and directors as an expense in the consolidated statements of operations over the requisite service period based on a measurement of fair value for each stock-based award. The fair value of each option grant is estimated as of the date of grant using the Black-Scholes option-pricing model, net of actual forfeitures. The fair value is amortized as compensation cost on a straight-line basis over the requisite service period of the awards, which is generally the vesting period.
Since we have a limited history of trading as a public company, our expected stock volatility is based on a weighting of its historical volatility along with a group of a publicly traded set of peer companies. We utilize the simplified method to estimate the expected term. The risk-free interest rate was determined by reference to the U.S. Treasury yield curve in effect at the time of grant of the award for time periods approximately equal to the expected term of the award. The expected dividend yield was assumed to be zero as we have not paid dividends since our inception and we do not anticipate paying dividends in the foreseeable future.
Long-lived assets
Long-lived assets are comprised of acquired technology and licensed rights to use technology, which are considered platform technology with alternative future uses beyond the current products in development. Such intangible assets are being amortized on a straight-line basis over their expected useful life of 10 years.
We assess the impairment for long-lived assets whenever events or circumstances indicate the carrying value may not be recoverable. Factors we consider that could trigger an impairment review include the following:
| ● | Significant changes in the manner of our use of the acquired assets or the strategy for our overall business, |
| ● | Significant underperformance relative to expected historical or projected development milestones, |
| ● | Significant negative regulatory or economic trends, and |
| ● | Significant technological changes which could render the platform technology obsolete. |
We recognize impairment when the sum of the expected undiscounted future cash flows is less than the carrying amount of the asset. Impairment losses, if any, are measured as the excess of the carrying amount of the asset over its estimated fair value. During the year ended December 31, 2024 there was no impairment indicator which required an impairment loss measurement. During the year ended December 31, 2023, there was one impairment indicator which required an impairment loss measurement (see Note 10 to the Consolidated Financial Statements included in this Annual Report).
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
We are a smaller reporting company as defined in Rule 12b-2 of the Exchange Act and are not required to provide the information otherwise required under this Item 7A.
Item 8. Financial Statements and Supplementary Data
The information required by this Item is set forth in the consolidated financial statements and notes thereto in Item 15 of Part IV of this Annual Report.
59
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
Not applicable.
Item 9A. Controls and Procedures
Disclosure Controls and Procedures
We maintain disclosure controls and procedures, which are designed to provide reasonable assurance that information required to be disclosed in the reports we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC rules and forms, and that such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate to allow timely decisions regarding required disclosure. Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, conducted an evaluation, as of the end of the period covered by this report, of the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15e under the Exchange Act). Based on that evaluation, our Chief Executive Officer and Chief Financial Officer have concluded that, as of the end of the period covered by this report, our disclosure controls and procedures were effective at the reasonable assurance level.
Management’s Annual Report on Internal Control over Financial Reporting
Our management, including our Chief Executive Officer and Chief Financial Officer, are responsible for establishing and maintaining adequate internal control over financial reporting (as defined in Rules 13a-15(f) under the Exchange Act). Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Internal control over financial reporting includes policies and procedures that:
| ● | pertain to the maintenance of records that in reasonable detail accurately and fairly reflect our transactions and asset dispositions; |
| ● | provide reasonable assurance that transactions are recorded as necessary to permit the preparation of our financial statements in accordance with generally accepted accounting principles; |
| ● | provide reasonable assurance that receipts and expenditures are made only in accordance with authorizations of our management and board of directors (as appropriate); and |
| ● | provide reasonable assurance regarding the prevention or timely detection of unauthorized acquisition, use or disposition of assets that could have a material effect on our financial statements. |
Due to its inherent limitations, any system of internal control over financial reporting, no matter how well defined, may not prevent or detect misstatements. In addition, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, assessed the effectiveness of our internal control over financial reporting as of December 31, 2024, based on the framework set forth in Internal Control — Integrated Framework by The Committee of Sponsoring Organizations of the Treadway Commission (COSO) (2013). Based on this assessment using this framework, our management concluded that our internal control over financial reporting was effective as of December 31, 2024.
60
Attestation Report of the Registered Public Accounting Firm
This Annual Report does not include an attestation report of our independent registered public accounting firm regarding internal control over financial reporting because Quoin Pharmaceuticals Ltd. is not an accelerated filer or a large accelerated filer, and it is not subject to the attestation requirement.
Changes in Internal Control over Financial Reporting
There was no change in our internal control over financial reporting (as defined in Rule 13a-15(f) under the Exchange Act), that occurred during the quarter ended December 31, 2024 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
During the fourth quarter of 2024, none of our directors or executive officers
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
61
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
Set forth below is certain information regarding the members of our board of directors (the “Board” or the “Board of Directors”) and our executive officers. Each director is entitled to serve until the 2025 annual meeting of shareholders and until a successor is duly elected and qualified or until his or her earlier retirement, resignation or removal.
Name |
| Age |
| Position(s) |
Dr. Michael Myers |
| 63 |
| Chairman of the Board and Chief Executive Officer |
Denise Carter |
| 56 |
| Director and Chief Operating Officer |
Joseph Cooper(1)(3) |
| 67 |
| Director |
James Culverwell(2)(4) |
| 68 |
| Director |
Dr. Dennis H. Langer(5) |
| 73 |
| Director |
Natalie Leong(1)(6) |
| 39 |
| Director |
Michael Sember(2) | 75 | Director | ||
Gordon Dunn |
| 60 |
| Chief Financial Officer |
| (1) | Member of our Audit Committee. |
| (2) | Member of our Compensation Committee. |
| (3) | Member of our Nominating and Governance Committee. |
| (4) | Chairperson of our Audit Committee |
| (5) | Chairperson of our Compensation Committee |
| (6) | Chairperson of our Nominating and Governance Committee |
Set forth below is a summary of the business experience of each of our directors and executive officers.
Dr. Michael Myers, Chief Executive Officer and Director. Dr. Myers is the co-founder of Quoin Inc. and has served as Chairman and Chief Executive Officer of Quoin Inc. since its inception in 2018. Dr. Myers has served as Chairman and Chief Executive Officer of Quoin Ltd. since October 28, 2021. Dr. Myers has over 36 years of industry experience in the drug delivery and specialty pharmaceutical sectors. From 2003 to October 2015, he served as Chief Executive Officer of Innocoll AG (n/k/a Innocoll Biotherapeutics N.A. Inc.), a biotherapeutics pharmaceutical company, and was responsible for taking that company public in 2014. From 2001 to 2002, he served as President of the drug delivery division of West Pharmaceutical Services, Inc., a publicly traded company and a designer and manufacturer of injectable pharmaceutical packaging and delivery systems. From 1996 to 1999, Dr. Myers served as the President of Pharmaceutical Operations for Fuisz Technologies (Biovail), a developer of food and drug delivery systems and technologies. From 2000 to 2001, Dr. Myers served as Executive Vice President and Chief Commercial Officer of Flamel Technologies (n/k/a Avadel Pharmaceuticals PLC, a publicly traded company and a specialty pharmaceutical company. From 1987 to 1995, Dr. Myers served as the Head of Pharmaceutical Development for Elan Corporation, a biotechnology drug company. Since 2019, Dr. Myers has served as a director of Sonoran Bioscience and Wellesley Pharmaceuticals, each a specialty pharmaceutical company. Dr. Myers has served as a director of Cranial Devices, a clinical stage medical device company since 2023. Dr. Myers earned his Ph.D. in Chemistry from University College Cork, Ireland. We believe Dr. Myers is qualified to serve on our Board due to his extensive knowledge as one of Quoin Inc.’s co-founders and Chief Executive Officer, and his extensive clinical development, commercial and management experience with both public and private life sciences companies.
Denise Carter, Chief Operating Officer and Director. Ms. Carter is the co-founder of Quoin Inc. and has served as a director and Chief Operating Officer of Quoin Inc. since its inception in 2018. Ms. Carter has served as a director and Chief Operating Officer of Quoin Ltd. since October 28, 2021. Ms. Denise Carter has over 30 years of experience in the drug delivery and specialty pharmaceutical industries. From June 2003 to October 2015, Ms. Carter held various positions at Innocoll AG (n/k/a Innocoll Biotherapeutics N.A. Inc.), including President of Innocoll Pharmaceuticals and Executive Vice President of Business Development and Corporate Affairs of Innocoll AG. From 2001 to 2003, Ms. Carter was the Vice President of Business Development of the drug delivery division of West Pharmaceuticals, Inc., a publicly traded company. From 2000 to 2001, she was the Senior Director of Business Development of Eurand, a specialty pharmaceutical company. From 1996 to 1999, Ms. Carter was the Director of Business Development and Alliance Management of Fuisz Technologies (Biovail). From 1999 to 2000, Ms. Carter was the Director of Business Development of Cardinal Health, Inc., a multi-national health care service company. Ms. Carter earned her MBA from Wharton School of Business, University of Pennsylvania and a B.S. in Chemistry from the College of William and Mary.
62
We believe Ms. Carter is qualified to serve on our Board due to her extensive knowledge as one of Quoin Inc.’s co-founders and Chief Operating Officer, and her extensive business development, sales and marketing and fundraising experience in the life sciences industry.
Joseph Cooper, Director. Mr. Cooper has served as a director of Quoin Inc. since May 2021. Mr. Cooper has served as a director of Quoin Ltd. since October 28, 2021. Mr. Cooper has significant experience in finance, operation, corporate development and general management roles within the pharmaceutical and healthcare industry. Since July 2023, Mr. Cooper has served as Chief Financial Officer for Hydrinity Skin Sciences, a medical aesthetics company. From 2012 to 2023, Mr. Cooper served as the President of Boulder Cove LC, a pharmaceutical and healthcare consulting company. From September 2019 to December 2022, Mr. Cooper served as the Chief of Strategy and Corporate Development for Resonea, Inc., a digital health company. From August 2018 to December 2019, Mr. Cooper served as the Chief Business Officer of NuvOx Pharmaceuticals, a clinical stage pharmaceutical company. From January 2015 to August 2018, Mr. Cooper served as Chief Financial and Operating Officer for First Place, AZ, a non-profit healthcare services organization. From 1996 to 2010, Mr. Cooper served as the Executive Vice President of Corporate and Product Development of Medicis Pharmaceutical Corp., a publicly traded pharmaceutical and medical aesthetics company. Since January 2018, Mr. Cooper has served as a director of Sonoran Biosciences, a specialty pharmaceutical company. From 2006 to 2007, Mr. Cooper served as a director of Bioenvision, a publicly traded pharmaceutical company. Mr. Cooper holds an MBA from the WP Carey School of Business at Arizona State University and a BA from Northeastern Illinois University. We believe Mr. Cooper is qualified to serve on our Board due to his extensive executive and board experience with pharmaceutical and healthcare companies.
James Culverwell, Director. Mr. Culverwell has served as a director of Quoin Inc. since April 2021. Mr. Culverwell has served as a director of Quoin Ltd. since October 28, 2021. Since May 2013, Mr. Culverwell has served as the Chief Executive Officer and is currently Chairman of the Board of Directors of HOX Therapeutics, a prostate cancer research company. In 2005, Mr. Culverwell founded Sudbrook Associates, which provided strategic advice and fund raising services for life science companies. From 1992 to 2004, Mr. Culverwell was Senior Vice President and Global Coordinator Healthcare Research at Merrill Lynch. From 1982 to 1992, Mr. Culverwell was Director of Healthcare Equity Research at ABN Amro Bank N.V., a private banking company. Since February 2022, Mr. Culverwell has served as a director and Audit Committee Chairman of TC BioPharm (Holdings) plc, a publicly traded company and a cancer treatment development company. Since January 2005, Mr. Culverwell has served as a director, Audit Committee Chairman, and member of the Compensation Committee of SafeGuard Biosystems, a high throughput molecular diagnostics company. From April 2016 to September 2019, Mr. Culverwell served as a director and Audit Committee Chairman of Amryt Pharma PLC, a publicly traded company and a commercial-stage biopharmaceutical company. From February 2013 to July 2017, Mr. Culverwell served as a director and Audit Committee Chairman of Innocoll AG. He received an MSc with honors from the University of Aberdeen. We believe Mr. Culverwell is qualified to serve on our Board due to his extensive experience serving on the audit and compensation committees for multiple public and private life sciences and healthcare companies.
Dennis H. Langer, M.D., J.D., Director. Dr. Langer has served as a director of Quoin Inc. since 2019. Dr. Langer has served as a director of Quoin Ltd. Since October 28, 2021. From 2005 to 2010, Dr. Langer served as the Managing Partner at Phoenix IP Ventures, LLC, a private equity and venture capital fund specializing in life sciences companies. From 2004 to 2005, Dr. Langer was the President, North America for Dr. Reddy’s Laboratories, Inc., a multi-national pharmaceutical company. Dr. Langer was with GlaxoSmithKline, a multi-national pharmaceutical and biotechnology company, from 1994 - 2004, where he served as Senior Vice President, Project, Portfolio and Alliance Management, Senior Vice President, Product Development Strategy, and Senior Vice President, Healthcare Services R&D. From 1991 to 1994, he served as President and Chief Executive Officer at Neose Technologies, Inc., a clinical stage biopharmaceutical company. From 2004 to June 2022, Dr. Langer served as a director of Myriad Genetics, Inc., a publicly traded company and a genetic testing and precision medicine company. From 2021 to June 2022, Dr. Langer served as a director of Brooklyn ImmunoTherapeutics, Inc. (n/k/a Eterna Therapeutics Inc.), a publicly traded company and a biotechnology company. From 2007 to 2019, Dr. Langer served as a director of Dicerna Pharmaceuticals Inc., a publicly traded company and a biopharmaceutical company. Dr. Langer has served on the Dean’s Advisory Board of Harvard Law School since 2010, and as a Director of the Whitehead Institute for Biomedical Research since 2020. He received an M.D. from Georgetown University School of Medicine, a J.D. from Harvard Law School, and a B.A. in Biology from Columbia University. We believe Dr. Langer is qualified to serve on our Board due to his extensive experience as an executive and board member of public and private life sciences and healthcare companies.
Natalie Leong, Director. Ms. Leong has served as a director of Quoin Inc. since April 2021. Ms. Leong has served as a director of Quoin Ltd. since October 28, 2021. Since January 2023, Ms. Leong has been the Senior Vice President of Product Management for B.S.D. Capital, Inc. (d/b/a Lendistry), a minority-led small business lender. Ms. Leong was the Head of Finance and Product Strategy (October 2019 – October 2020) and subsequently Head of Product Management (October 2020 – November 2022) for LoanStreet Inc., a financial SaaS company. From May 2016 to July 2019, Ms. Leong served as the Lead for the Asset Liability Committee for the US at RBC Capital Markets. In addition, from August 2018 to October 2019, she served as the Lead for Global Originations FP&A for RBC Capital Markets. From October 2011 to May 2016, Ms. Leong worked as the Vice President of Capital Insights at National Australia
63
Bank. From February 2008 to October 2011, Ms. Leong served as a Senior Auditor at National Australia Bank. Ms. Leong earned her MBA at The Wharton School, University of Pennsylvania. She earned a B.Comm degree (Finance and Economics) and a B.A. degree (French and Literature) from the University of Melbourne in 2007. We believe Ms. Leong is qualified to serve on our Board due to her extensive financial and business management experience.
Michael Sember, Director. Mr. Sember has served as a director of Quoin Inc. since May 2021. Mr. Sember has served as a director of Quoin Ltd. since October 28, 2021. Since 2007, he has served as a Principal of Accela Advisors, a biopharmaceutical consulting firm specializing in strategic planning, business development and coaching for startups. From 2022 until 2023, Mr. Sember served as the Chief Executive Officer of RaeSedo, Inc, a startup therapeutics company spin out of the University of Arizona. From January 2018 to October 2020, Mr. Sember served as the Chief Executive Officer of Regulonix Holding, Inc., a drug development company. From October 2015 to March 2019, he served as the Mentor in Residence to companies formed from inventions discovered at the University of Arizona. From 2013 to 2015, Mr. Sember was the Corporate Turnaround Specialist and Chief Executive Officer of Palyon Medical Corporation, a drug delivery system company. From 1991 to 2002, Mr. Sember was Executive Vice President of Corporate Business Development for Élan Corporation, responsible for strategic collaborations and mergers and acquisitions. From 1973 to 1991, Mr. Sember served as the Senior director of Global Program Management at Marion Laboratories (later Marion Merrell Dow). From 2013 to 2015, Mr. Sember was the Chairman of the Board of Paylon Medical Corporation, a drug delivery system company. From 2012 to 2013, Mr. Sember was the Chairman of the Board of BioIndustry Organization of Southern Arizona, a non-profit trade group. Mr. Sember earned a Bachelor of Science degree from the University of Pittsburgh and an MBA from Rockhurst University. We believe Mr. Sember is qualified to serve on our Board due to his broad executive and capital raising experience in the life sciences industry.
Gordon Dunn, Chief Financial Officer. Mr. Dunn has served as Chief Financial Officer of Quoin Ltd. since November 1, 2021. Mr. Dunn has over 30 years of finance experience. He served as Chief Financial Officer of Health Technologies Ltd. (d/b/a Qured), a UK-based healthcare provider, from March 2020 to October 2021, and as Chief Financial Officer of U-Research, an online company information platform, from July 2017 to March 2020. Mr. Dunn also served as Chief Financial Officer of Anton Corporation, a film and media finance company, from September 2016 to July 2017, and as Chief Financial Officer of Innocoll AG from 2012 to 2016. Prior to these roles, he had deep experience in investment banking and private equity, serving as Portfolio Manager of NewSmith Asset Management, a private equity fund from 2004 to 2014, and as Director of Investment Banking and Co-Head of Private Equity at Merrill Lynch, in addition to other roles, from 1994 to 2003. Mr. Dunn also serves as a director of Oddonos Gelati Italiani Ltd. Mr. Dunn was an associate at Morrison & Foerster LLP from 1991 to 1993. Mr. Dunn earned his JD from New York University School of Law and a BA from Stanford University.
Code of Ethics
We have adopted a Code of Ethics and Business Conduct (the “Code of Ethics”) that applies to all of our directors, officers and employees, including our principal executive officer and our principal financial and accounting officer. A copy of our Code of Ethics has been posted to the “Investors—Corporate Governance” section of our website www.quoinpharma.com, and it is attached as an exhibit to this Annual Report. If we make any amendment to the Code of Ethics or grant any waivers, including any implicit waiver, from a provision of the Code of Ethics, we will disclose the nature of such amendment or waiver on our website www.quoinpharma.com. to the extent required by the rules and regulations of the SEC. The information on the website is not and should not be considered part of this Form 10-K and is not incorporated by reference in this Form 10-K.
Board of Directors
The Board of Directors has established three standing committees: the Audit Committee, the Compensation Committee and the Nominating and Governance Committee.
Audit Committee
The Audit Committee of the Board of Directors consists of Joseph Cooper, James Culverwell, and Natalie Leong, with Mr. Culverwell chairing the committee.
Under the Nasdaq listing standards, we are required to maintain an audit committee consisting of at least three independent directors, each of whom is financially literate and one of whom has accounting or related financial management expertise. Our Board has determined that each member of the Audit Committee satisfies the independence requirements under Nasdaq listing standards and Rule 10A-3(b)(1) of the Exchange Act, has the requisite financial sophistication as required by the Nasdaq listing standards and is an audit committee financial expert, as defined by the SEC rules.
64
Our Board adopted the Amended and Restated Charter of the Audit Committee that sets forth the responsibilities of the Audit Committee under Nasdaq listing standards, as well as the requirements for such committee under the Companies Law, including the following:
| ● | overseeing our independent registered public accounting firm and recommending the engagement, compensation or termination of engagement of our independent registered public accounting firm to the board of directors in accordance with Israeli law; |
| ● | recommending the engagement or termination of the person filling the office of our internal auditor; |
| ● | recommending the terms of audit and non-audit services provided by the independent registered public accounting firm for pre-approval by our board of directors; |
| ● | determining whether there are deficiencies in the business management practices of our company, including in consultation with our internal auditor or the independent auditor, and making recommendations to the board of directors to improve such practices; |
| ● | determining the approval process for transactions that are ‘non-negligible’ (i.e., transactions with a controlling shareholder that are classified by the audit committee as non-negligible, even though they are not deemed extraordinary transactions), as well as determining which types of transactions would require the approval of the audit committee, which determination may be based on annually pre-determined criteria; |
| ● | determining whether to approve certain related party transactions (including transactions in which an office holder (as defined below) has a personal interest and whether such transaction is extraordinary or material under the Companies Law); |
| ● | review and discuss the Company’s policies regarding information technology security and protection from cyber risks; |
| ● | examining the work plan of the internal auditor before its submission to our board of directors and proposing amendments thereto or, upon a decision of the board of directors, acting as the corporate body to approve such work plan; |
| ● | examining our internal controls and internal auditor’s performance, including whether the internal auditor has sufficient resources and tools at his disposal to fulfill his responsibilities; |
| ● | examining the scope of our independent auditor’s work and compensation and submitting a recommendation with respect thereto to our board of directors; and |
| ● | establishing procedures for the handling of employees’ complaints as to the management of our business and the protection to be provided to such employees. |
Compensation Committee
The Compensation Committee of the Board consists of James Culverwell, Dennis Langer and Michael Sember, with Dr. Langer chairing the committee. The Board of Directors has determined that each member of the Compensation Committee is independent under Nasdaq listing standards.
Our Board adopted the Amended and Restated Charter of the Compensation Committee that sets forth the responsibilities of such committee under Nasdaq listing standards, as well as the requirements for such committee under the Companies Law, including the following:
| ● | recommending to our board of directors a policy regarding the terms of engagement of the company’s office holders, to which we refer as a “compensation policy”; |
| ● | recommending whether the compensation policy should continue in effect, if the then-current policy has a term of greater than three years (approval of either a new compensation policy or the continuation of an existing compensation policy must in any case occur every three years); |
65
| ● | recommending to the board of directors updates to the compensation policy from time to time; |
| ● | assessing implementation of the compensation policy; |
| ● | resolving whether to approve arrangements with respect to the terms of office and employment of office holders, which require the approval of the compensation committee pursuant to the Companies Law; |
| ● | exempting, under certain circumstances, a transaction with our Chief Executive Officer from the approval of our shareholders.; |
| ● | making other determinations that the Companies Law assigns to a compensation committee; |
| ● | reviewing and recommending for approval by the board of directors the overall compensation policies with respect to our Chief Executive Officer and other executive officers; |
| ● | reviewing and recommending for approval by the board of directors the corporate goals and objectives relevant to the compensation of our Chief Executive Officer and other executive officers; |
| ● | evaluating the performance of our Chief Executive Officer and other executive officers in light of such goals and objectives; |
| ● | reviewing and approving the granting of options and other incentive awards, including the exercise of authorities delegated by the board of directors regarding the grant of equity incentives under our equity compensation plans; |
| ● | reviewing, evaluating and making recommendations regarding the compensation and benefits for our non-employee directors; |
| ● | overseeing our compliance with SEC and Nasdaq rules related to shareholder approval of certain executive compensation matters and equity compensation plans; |
| ● | considering and implementing policies with respect to oversight, assessment and management of risks associated with our compensation polices; and |
| ● | reviewing and establishing appropriate insurance coverage for our office holders. |
Nominating and Governance Committee
Our Nominating and Governance Committee consists of Natalie Leong and Joseph Cooper, with Ms. Leong chairing the committee. The Board of Directors has determined that each member of the Nominating and Governance Committee is independent under Nasdaq listing standards.
Our Board adopted the Amended and Restated Charter of the Nominating and Governance Committee that sets forth the responsibilities of such committee under Nasdaq listing standards, as well as the requirements for such committee under the Companies Law, including the following:
| ● | evaluating our corporate leadership structure, and reviewing important issues and developments in corporate governance, and developing appropriate recommendations for the Board; and |
| ● | overseeing and assisting our board in reviewing and recommending nominees for election as directors and members of committees of our board. |
Internal Auditor
Under the Companies Law, the board of directors of a public company must appoint an internal auditor based on the recommendation of the audit committee. The role of the internal auditor is, among other things, to review the company’s compliance with applicable law and orderly business procedure. Under the Companies Law, the internal auditor cannot be an interested party, an office holder, or a relative of an interested party or an office holder. Nor may the internal auditor be the company’s independent auditor
66
or its representative. An “interested party” is defined in the Companies Law as (i) a holder of 5% or more of the issued share capital or voting power in a company, (ii) any person or entity who has the right to designate one or more directors or to designate the chief executive officer of the company, or (iii) any person who serves as a director or as chief executive officer of the company. The role of the internal auditor is to examine, among other things, our compliance with applicable law and orderly business procedures. The audit committee is required to oversee the activities of the internal auditor and to assess his or her work plan and performance. Our internal auditor is Mr. Edo Pollack, a Certified Public Accountant and partner-in-charge of the Israel office of Eisner Advisory Group LLC.
Insider Trading Policy
Fiduciary Duties of Directors, Executive Officers and Shareholders
The Companies Law codifies the fiduciary duties that office holders owe to a company. An office holder is defined in the Companies Law as a general manager, chief business manager, deputy general manager, vice general manager, any other person assuming the responsibilities of any of these positions regardless of such person’s title, a director, and any other manager directly subordinate to the general manager. Each person listed in the table under “Management” is an office holder under the Companies Law.
An office holder’s fiduciary duties consist of a duty of care and a duty of loyalty. The duty of care requires an office holder to act with the level of care with which a reasonable office holder in the same position would act under the same circumstances. The duty of loyalty requires that an office holder act in good faith and in the best interests of the company.
The duty of care includes a duty to use reasonable means to obtain:
| ● | information on the advisability of a given action brought for the office holder’s approval or performed by virtue of his or her position; and |
| ● | all other important information pertaining to any such action. |
The duty of loyalty includes a duty to:
| ● | refrain from any conflict of interest between the performance of the office holder’s duties to the company and his or her other duties or personal affairs; |
| ● | refrain from any activity that is competitive with the company; |
| ● | refrain from exploiting any business opportunity of the company to receive a personal gain for himself or herself or others; and |
| ● | disclose to the company any information or documents relating to the company’s affairs which the office holder received as a result of his or her position as an office holder. |
67
Shareholder duties
Pursuant to the Companies Law, a shareholder has a duty to act in good faith and in a customary manner toward the company and other shareholders and to refrain from abusing his or her power with respect to the company, including, among other things, in voting at a general meeting and at shareholder class meetings with respect to the following matters:
| ● | an amendment to the company’s articles of association; |
| ● | an increase of the company’s authorized share capital; |
| ● | a merger; or |
| ● | interested party transactions that require shareholder approval. |
In addition, a shareholder has a general duty to refrain from discriminating against other shareholders.
Certain shareholders also have a duty of fairness toward the company. These shareholders include any controlling shareholder, any shareholder who knows that it has the power to determine the outcome of a shareholder vote, and any shareholder who has the power to appoint or to prevent the appointment of an office holder of the company or exercise any other rights available to it under the company’s articles of association with respect to the company. The Companies Law does not define the substance of this duty of fairness, except to state that the remedies generally available upon a breach of contract will also apply in the event of a breach of the duty of fairness.
Disclosure of Personal Interests of an Office Holder and Approval of Certain Transactions
The Companies Law requires that an office holder promptly disclose to the board of directors any personal interest and all related material information known to such office holder concerning any existing or proposed transaction with the company. A personal interest includes an interest of any person in an act or transaction of a company, including a personal interest of one’s relative or of a corporate body in which such person or a relative of such person is a 5% or greater shareholder, director, or general manager or in which such person has the right to appoint at least one director or the general manager, but excluding a personal interest stemming solely from one’s ownership of shares in the company. A personal interest includes the personal interest of a person for whom the office holder holds a voting proxy or the personal interest of the office holder with respect to the officer holder’s vote on behalf of a person for whom he or she holds a proxy even if such shareholder has no personal interest in the matter.
If it is determined that an office holder has a personal interest in a non-extraordinary transaction (meaning any transaction that is in the ordinary course of business, on market terms or that is not likely to have a material impact on the company’s profitability, assets or liabilities), approval by the board of directors is required for the transaction unless the company’s articles of association provide for a different method of approval. Any such transaction that is adverse to the company’s interests may not be approved by the board of directors.
Approval first by the company’s audit committee and subsequently by the board of directors is required for an extraordinary transaction (meaning any transaction that is not in the ordinary course of business, not on market terms or that is likely to have a material impact on the company’s profitability, assets or liabilities) in which an office holder has a personal interest.
A director and any other office holder who has a personal interest in a transaction which is considered at a meeting of the board of directors or the audit committee may generally (unless it is with respect to a transaction which is not an extraordinary transaction) not be present at such a meeting or vote on that matter unless a majority of the directors or members of the audit committee, as applicable, have a personal interest in the matter. If a majority of the members of the audit committee or the board of directors have a personal interest in the matter, then all of the directors may participate in deliberations of the audit committee or board of directors, as applicable, with respect to such transaction and vote on the approval thereof and, in such case, shareholder approval is also required.
Certain disclosure and approval requirements apply under Israeli law to certain transactions with controlling shareholders, certain transactions in which a controlling shareholder has a personal interest, and certain arrangements regarding the terms of service or employment of a controlling shareholder. For these purposes, a controlling shareholder is any shareholder that has the ability to direct the company’s actions, including any shareholder holding 25% or more of the voting rights if no other shareholder owns more than 50%
68
of the voting rights in the company. Two or more shareholders with a personal interest in the approval of the same transaction are deemed to be one shareholder.
Exculpation, insurance and indemnification of office holders
Under the Companies Law, a company may not exculpate an office holder from liability for a breach of the duty of loyalty. An Israeli company may exculpate an office holder in advance from liability to the company, in whole or in part, for damages caused to the company as a result of a breach of duty of care, but only if a provision authorizing such exculpation is included in its articles of association. Our articles of association include such a provision. An Israeli company may not exculpate a director from liability arising out of a prohibited dividend or distribution to shareholders.
An Israeli company may indemnify an office holder from the following liabilities and expenses incurred for acts performed as an office holder, either in advance of an event or following an event, provided a provision authorizing such indemnification is contained in its articles of association:
| ● | a financial liability imposed on him or her in favor of another person pursuant to a judgment, including a settlement or arbitrator’s award approved by a court. However, if an undertaking to indemnify an office holder with respect to such liability is provided in advance, then such an undertaking must be limited to events which, in the opinion of the board of directors, can be foreseen based on the company’s activities when the undertaking to indemnify is given, and to an amount or according to criteria determined by the board of directors as reasonable under the circumstances, and such undertaking shall detail the above mentioned events and amount or criteria; |
| ● | reasonable litigation expenses, including legal fees, incurred by the office holder (1) as a result of an investigation or proceeding instituted against him or her by an authority authorized to conduct such investigation or proceeding, provided that (i) no indictment was filed against such office holder as a result of such investigation or proceeding; and (ii) no financial liability, such as a criminal penalty, was imposed upon him or her as a substitute for the criminal proceeding as a result of such investigation or proceeding or, if such financial liability was imposed, it was imposed with respect to an offense that does not require proof of criminal intent; and (2) in connection with a monetary sanction; |
| ● | reasonable litigation expenses, including legal fees, incurred by the office holder or imposed by a court in proceedings instituted against him or her by the company, on its behalf or by a third-party or in connection with criminal proceedings in which the office holder was acquitted or as a result of a conviction for an offense that does not require proof of criminal intent; |
| ● | expenses, including reasonable litigation expenses and legal fees, incurred by an office holder in relation to an administrative proceeding instituted against such office holder, or certain compensation payments made to an injured party imposed on an office holder by an administrative proceeding, pursuant to certain provisions of the Israeli Securities Law; and |
| ● | expenses, including reasonable litigation expenses and legal fees, incurred by an office holder in relation to an administrative proceeding instituted against such office holder pursuant to certain provisions of the Israeli Economic Competition Law, 5758-1988. |
An Israeli company may insure an office holder against the following liabilities incurred for acts performed as an office holder if and to the extent provided in the company’s articles of association:
| ● | a breach of the duty of loyalty to the company, to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not prejudice the company; |
| ● | a breach of the duty of care to the company or to a third-party, including a breach arising out of the negligent conduct of the office holder; |
| ● | a financial liability imposed on the office holder in favor of a third-party; |
| ● | a financial liability imposed on the office holder in favor of a third-party harmed by a breach in an administrative proceeding, pursuant to certain provisions of the Israeli Securities Law; and |
69
| ● | expenses, including reasonable litigation expenses and legal fees, incurred by the office holder as a result of an administrative proceeding instituted against him or her, pursuant to certain provisions of the Israeli Securities Law. |
An Israeli company may not exempt, indemnify or insure an office holder against any of the following:
| ● | a breach of the duty of loyalty, except with respect to insurance coverage or indemnification, to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not prejudice the company; |
| ● | a breach of the duty of care committed intentionally or recklessly, excluding a breach arising out of the negligent conduct of the office holder; |
| ● | an act or omission committed with intent to derive illegal personal benefit; or |
| ● | a fine, monetary sanction, or forfeit levied against the office holder. |
Under the Companies Law, exculpation, indemnification, and insurance of office holders must be approved by the compensation committee and the board of directors (and, with respect to directors and the chief executive officer, by the shareholders). However, under regulations promulgated under the Companies Law, the insurance of office holders shall not require shareholder approval and may be approved by only the compensation committee if the engagement terms are determined in accordance with the company’s compensation policy, which was approved by the shareholders by the same special majority required to approve a compensation policy, provided that the insurance policy is on market terms and the insurance policy is not likely to materially impact the company’s profitability, assets, or obligations.
Our articles of association allow us to exculpate, indemnify, and insure our office holders to the maximum extent permitted by law. Our office holders are currently covered by a directors and officers’ liability insurance policy.
We have entered into agreements with each of our directors and executive officers exculpating them in advance, to the fullest extent permitted by law, from liability to us for damages caused to us as a result of a breach of duty of care, and undertaking to indemnify them to the fullest extent permitted by law. This indemnification is limited to events determined as foreseeable by the board of directors based on our activities and to an amount or according to criteria determined by the board of directors as reasonable under the circumstances.
In the opinion of the SEC, indemnification of directors and office holders for liabilities arising under the Securities Act, however, is against public policy and therefore unenforceable.
70
Item 11. Executive Compensation
Summary Compensation Table
The following table sets forth information concerning the compensation awarded to, earned by, or paid to our Chief Executive Officer, Chief Operating Officer and Chief Financial Officer (collectively referred to as “named executive officers”) during the years ended December 31, 2024 and 2023.
|
| Option |
| All Other |
| |||||||
Salary | Bonus(1) | Awards(2) | Compensation(3) | Total(4) | ||||||||
Name and Principal Position |
| Year |
| ($) |
| ($) |
| ($) |
| ($) |
| ($) |
Dr. Michael Myers | 2024 | 662,475 | — | 353,013 | 60,075 | 1,075,563 | ||||||
Chief Executive Officer | 2023 | 602,250 | 301,125 | 292,263 | 59,550 | 1,255,188 | ||||||
Denise Carter | 2024 | 529,980 | — | 353,017 | 63,625 | 946,622 | ||||||
Chief Operating Officer | 2023 | 481,800 | 240,900 | 292,266 | 56,000 | 1,070,966 | ||||||
Gordon Dunn | 2024 | 433,620 | — | 223,012 | — | 656,632 | ||||||
Chief Financial Officer | 2023 | 394,200 | 197,100 | 184,635 | — | 775,935 |
| (1) | For bonuses earned during the year ended December 31, 2023, represents a discretionary cash bonus granted in recognition of the applicable officer’s promotion of our long-term goals, strategy and operating plan, the need to have appropriate incentives for our officers, and contribution to the achievement of our objectives in accordance with the applicable officer’s respective corporate role during the year ended December 31, 2023. Dr. Myers’ and Ms. Carter’s bonuses were approved and/or ratified by the Board and the Compensation Committee, consistent with the Company’s Compensation Policy and within the limitations of the CEO Compensation Program (as described below) and the COO Compensation Program (as described below). The CEO Compensation Program and the COO Compensation Program were approved at our 2024 Annual Meeting held on December 5, 2024 (the “2024 Annual Meeting”). The amount of bonuses earned during the year ended December 31, 2024 is not calculable through the date of this Annual Report, and such amounts will be disclosed in a Current Report on Form 8-K after we obtain applicable approvals of our Board and the Compensation Committee. |
| (2) | Represents the grant date fair value of option awards granted to each of our named executive officers on October 26, 2023 and December 9, 2024, respectively, calculated in accordance with FASB ASC Topic 718. The 2023 options have an exercise price of $5.75 per ADS and vest in in three annual installments of 20% and a fourth annual installment of 40% beginning on October 26, 2024. The 2024 options have an exercise price of $0.78 per ADS and vest in in three annual installments of 20% and a fourth annual installment of 40% beginning on December 9, 2025. The option values were calculated using a Black-Scholes Model for pricing options. See Note 6 to the Consolidated Financial Statements included in this Annual Report for all relevant valuation assumptions used to determine the grant date fair value of these options. |
| (3) | Represents amounts paid as office and automobile allowance to Dr. Myers and Ms. Carter under their respective employment agreements, as well as the employer matching contribution to the executive’s 401(k) plan contributions under our Section 401(k) retirement plan (the “Section 401(k) Plan”), broken down as follows: |
Office | Car | 401(k) | ||||||||
Allowance | Allowance | Contributions | Total | |||||||
|
| ($) |
| ($) |
| ($) |
| ($) | ||
Michael Myers | 2024 | 30,000 | 18,000 | 12,075 | 60,075 | |||||
2023 | 30,000 | 18,000 | 11,550 | 59,550 | ||||||
Denise Carter | 2024 | 30,000 | 18,000 | 15,625 | 63,625 | |||||
2023 | 30,000 | 18,000 | 8,000 | 56,000 | ||||||
Gordon Dunn | 2024 | — | — | — | — | |||||
2023 | — | — | — | — |
Employment Agreements
We entered into written employment agreements with our named executive officers that contain customary provisions, including non-compete and confidentiality provisions.
71
Dr. Myers. Pursuant to his Executive Employment Agreement with Quoin Inc., dated March 9, 2018, which was amended as of November 9, 2021 (as amended, the “Myers Agreement”), Dr. Myers is entitled to an annual base salary of $550,000, which accrued monthly until paid by Quoin Inc. Dr. Myers may also receive, subject to employment by us on the applicable date of bonus payout, an annual target discretionary bonus of not less than 45% of his annual base salary, payable at the discretion of the board of directors after approval of our compensation committee, subject to shareholder approval by a Special Majority for Compensation Matters. Pursuant to the Myers Agreement, Dr. Myers is also eligible to receive healthcare benefits as may be provided from time to time by us to our employees generally, and to receive paid time off annually in accordance with our policies in effect from time to time. Additionally, the Myers Agreement provides Dr. Myers with a monthly office allowance of $2,500 and a monthly automobile allowance of $1,500. At the annual general meeting of shareholders held on October 26, 2023, shareholders approved an amendment to Dr. Myers’ employment agreement to increase Dr. Myer’s annual base salary by 9.5%, retroactive to January 1, 2023, to $602,250. After the 2024 Annual Meeting, on December 9, 2024, the Compensation Committee and the Board took the following actions which were consistent with the Company’s Compensation Policy and within the limitations of the CEO Compensation Program: (i) approved and ratified Dr. Myers’ 2024 annual base salary at $662,475 (retroactive to January 1, 2024), (ii) approved and ratified a discretionary cash bonus for Dr. Myers for fiscal 2023 services of $301,125; and (iii) granted Dr. Myers an option to purchase 536,603 ADSs under Quoin’s Amended and Restated Equity Incentive Plan, with an exercise price equal to $0.78 per ADS, the fair market value on the date of grant.
Ms. Carter. Pursuant to her Executive Employment Agreement with Quoin Inc., dated March 9, 2018, which was amended as of November 9, 2021 (as amended, the “Carter Agreement”), Ms. Carter is entitled to an annual base salary of $440,000, which accrued monthly until paid by Quoin Inc. Ms. Carter may also receive, subject to employment by us on the applicable date of bonus payout, an annual target discretionary bonus of not less than 45% of her annual base salary, payable at the discretion of the board of directors after approval of our compensation committee, subject to shareholder approval by a Special Majority for Compensation Matters. Pursuant to the Carter Agreement, Ms. Carter is also eligible to receive healthcare benefits as may be provided from time to time by us to our employees generally, and to receive paid time off annually in accordance with Quoin’s policies in effect from time to time. Additionally, the Carter Agreement provides Ms. Carter with a monthly office allowance of $2,500 and a monthly automobile allowance of $1,500. At the annual general meeting of shareholders held on October 26, 2023, shareholders approved an amendment to Ms. Carter’s employment agreement to increase to Ms. Carter’s annual base salary by 9.5%, retroactive to January 1, 2023, to $481,800. After the 2024 Annual Meeting, on December 9, 2024, the Compensation Committee and the Board took the following actions which were consistent with the Company’s Compensation Policy and within the limitations of the COO Compensation Program: (i) approved and ratified Ms. Carter’s 2024 annual base salary at $529,980 (retroactive to January 1, 2024), (ii) approved and ratified a discretionary cash bonus for Ms. Carter for fiscal 2023 services of $240,900; and (iii) granted Ms. Carter an option to purchase 536,609 ADSs under Quoin’s Amended and Restated Equity Incentive Plan, with an exercise price equal to $0.78 per ADS, the fair market value on the date of grant.
Mr. Dunn. Pursuant to his Service Agreement with Quoin Inc., dated November 1, 2021 (as amended, the “Dunn Agreement”), Mr. Dunn is entitled to an annual base salary of $360,000. In addition, Mr. Dunn received a signing bonus equal to one-twelfth of his annual base salary, and is entitled to receive subject to employment by us on the applicable date of bonus payout, an annual target discretionary bonus of not less than 45% of his annual base salary, payable at the discretion of the Board, which was prorated for 2021. Under the Dunn Agreement, we granted an option to Mr. Dunn to purchase our ordinary shares, with a $1.25 million grant date value. Mr. Dunn is also eligible to receive healthcare benefits as may be provided from time to time by us to our employees generally and paid time off annually in accordance with our policies in effect from time to time. Effective October 26, 2023, Mr. Dunn’s annual base salary was amended to provide for an increase to his annual base salary by 9.5%, retroactive to January 1, 2023, to $394,200. On December 9, 2024, the Compensation Committee and the Board took the following actions which were consistent with the Company’s Compensation Policy: (i) approved and ratified the setting of the 2024 annual base salary for Gordon Dunn, our Chief Financial Officer, at $433,620 (retroactive to January 1, 2024), (ii) approved and ratified a discretionary cash bonus for Mr. Dunn for fiscal 2023 services of $197,100; and (iii) granted Mr. Dunn an option to purchase 338,994 ADSs under Quoin’s Amended and Restated Equity Incentive Plan, with an exercise price equal to $0.78 per ADS, the fair market value on the date of grant.
Compensation Program for Dr. Michael Myers
At the 2024 Annual Meeting, the Company’s shareholders approved a compensation program for the Company’s Chief Executive Officer and Chairman of the Board, Dr. Michael Myers. The program sets forth the following compensation limitations applicable to Dr. Myers which the Compensation Committee and the Board can utilize in setting Dr. Myers’ compensation, beginning with the compensation to be paid in fiscal 2024, without the need to obtain further shareholder approval:
(i)an annual increase of base salary of up to 15% of Dr. Myers’ then effective base salary;
72
(ii)an annual cash bonus of up to 50% of Dr. Myers’ annual base salary during the fiscal year for which the annual cash bonus is paid (for example, Dr. Myers’ bonus to be paid in fiscal 2025 for fiscal 2024 services would be based upon a percentage, up to 50%, of Dr. Myers’ annual base salary in fiscal 2024); and
(iii)an annual equity grant in any form permitted under the Company’s equity incentive plan in effect from time to time with an annual value (determined in accordance with the Black-Scholes formula or another widely accepted and suitable formula for calculating the value of equity awards) of up to 500% of the maximum total fixed component (base salary and benefits) to which Dr. Myers is entitled in the grant year.
(together the “CEO Compensation Program”).
In setting future compensation for Dr. Myers consistent with the terms of the CEO Compensation Program, the Compensation Committee and the Board will continue to annually review market competitive compensation as a reference, individual performance, the need to have appropriate incentives for our officers, and Dr. Myers’ experience and expected contributions.
Compensation Program for Denise Carter
Also at the 2024 Annual Meeting, the Company’s shareholders approved a compensation program for the Company’s Chief Operating Officer and a member of the Board, Denise Carter. The program sets forth the following compensation limitations applicable to Ms. Carter which the Compensation Committee and the Board can utilize in setting Ms. Carter’s compensation, beginning with the compensation to be paid in fiscal 2024, without the need to obtain further shareholder approval:
(i)an annual increase of base salary of up to 15% of Ms. Carter’s then effective base salary;
(ii)an annual cash bonus of up to 50% of Ms. Carter’s annual base salary during the fiscal year for which the annual cash bonus is paid (for example, Ms. Carter’s bonus to be paid in fiscal 2025 for fiscal 2024 services would be based upon a percentage, up to 50%, of Ms. Carter’s annual base salary in fiscal 2024); and
(iii)an annual equity grant in any form permitted under the Company’s equity incentive plan in effect from time to time with an annual value (determined in accordance with the Black-Scholes formula or another widely accepted and suitable formula for calculating the value of equity awards) of up to 500% of the maximum total fixed component (base salary and benefits) to which Ms. Carter is entitled in the grant year.
(together the “COO Compensation Program”).
In setting future compensation for Ms. Carter consistent with the terms of the CEO Compensation Program, the Compensation Committee and the Board will continue to annually review market competitive compensation as a reference, individual performance, the need to have appropriate incentives for our officers, and Ms. Carter’s experience and expected contributions.
Health and Welfare Benefits
Our named executive officers are eligible to participate in the same employee benefit plans, and on the same terms and conditions, as all other full-time, salaried U.S. employees. These benefits include medical, dental, and vision insurance, an employee assistance program, health and dependent care flexible spending accounts, basic life insurance, accidental death and dismemberment insurance, short-term and long-term disability insurance, and commuter benefits.
We also maintain the “Section 401(k) Plan that provides eligible employees, including our named executive officers, with an opportunity to save for retirement on a tax-advantaged basis. Eligible employees are able to participate in the Section 401(k) Plan as of the first day of the month following the date they meet the plan’s eligibility requirements. Participants are able to defer up to 100% of their eligible compensation subject to applicable annual limits under the Internal Revenue Code (the “Code”). All participants’ interests in their deferrals are 100% vested when contributed. Currently, we match up to 100% of a participant’s first 1% of his or her eligible contributions to the Section 401(k) Plan, and we match up to 50% of the next 5% of his or her eligible contributions.
73
Outstanding Equity Awards at December 31, 2024
The following table sets forth information with respect to outstanding equity awards for each named executive officer as of December 31, 2024.
|
| Number of |
| Number of |
|
|
|
| ||
Securities | Securities | |||||||||
Underlying | Underlying | Option | ||||||||
Unexercised | Unexercised | Exercise | Option | |||||||
Option Grant | Options (#) | Options (#) | Price | Expiration | ||||||
Name | Date | Exercisable | Unexercisable(1) | ($)(2) | Date | |||||
Dr. Michael Myers |
| 4/12/2022 | 3,572 |
| 3,571 |
| 210 |
| 04/12/2032 | |
10/26/2023 | 16,193 | 64,772 | 5.75 | 10/26/2033 | ||||||
12/9/2024 | — | 536,603 | 0.78 | 12/9/2034 | ||||||
Denise Carter |
| 4/12/2022 | 3,572 |
| 3,571 |
| 210 |
| 04/12/2032 | |
10/26/2023 | 16,193 | 64,773 | 5.75 | 10/26/2033 | ||||||
12/9/2024 | — | 536,609 | 0.78 | 12/9/2034 | ||||||
Gordon Dunn | 4/12/2022 | 2,976 | 2,977 |
| 210 |
| 04/12/2032 | |||
10/26/2023 | 10,230 | 40,919 | 5.75 | 10/26/2033 | ||||||
| 12/9/2024 | — |
| 338,994 | 0.78 | 12/9/2034 |
| (1) | Represents the number of ADSs issuable upon the exercise of options. The 2022 options vest in four equal annual installments beginning on April 12, 2023. The 2023 options vest in three annual installments of 20% and a fourth annual installment of 40% beginning on October 26, 2024. The 2024 options vest in three annual installments of 20% and a fourth annual installment of 40% beginning on December 9, 2025. |
| (2) | Represents the exercise price per ADS. |
Amended and Restated Equity Incentive Plan
At our Annual General Meeting held on April 12, 2022, our shareholders approved our Amended and Restated Equity Incentive Plan (the “Plan”), which amended and restated our 2014 Global Incentive Option Scheme. The number of shares reserved for issuance under the Plan is equal to 15% of our outstanding ordinary shares on a fully-diluted basis. The purpose of the Plan is to attract, retain and motivate our employees (including prospective employees), non-employee directors and consultants. The Board has the power to administer the Plan, either directly or upon the recommendation of the Compensation Committee of the Board, in accordance with applicable law and our Articles of Association. Options granted under the Plan are subject to applicable vesting schedules and generally expire ten years from the grant date
Company Policies and Practices Related to the Grant of Certain Equity Awards Close in Time to the Release of Material Nonpublic Information
Clawback Policy
The Board adopted a clawback policy which requires the clawback of erroneously awarded incentive-based compensation of past or current executive officers awarded during the three full fiscal years preceding the date on which the issuer is required to prepare an accounting restatement due to the material noncompliance of the Company with any financial reporting requirement under the federal securities laws. There is no fault or misconduct required to trigger a clawback.
The Compensation Committee shall determine, in its sole discretion, the timing and method for promptly recouping such erroneously awarded compensation, which may include without limitation: (a) seeking reimbursement of all or part of any cash or equity-based award, (b) cancelling prior cash or equity-based awards, whether vested or unvested or paid or unpaid, (c) cancelling or
74
offsetting against any planned future cash or equity-based awards, (d) forfeiture of deferred compensation, subject to compliance with Section 409A of the Internal Revenue Code and the regulations promulgated thereunder, and (e) any other method authorized by applicable law or contract. Subject to compliance with any applicable law, the Compensation Committee may affect recovery under this policy from any amount otherwise payable to the executive officer, including amounts payable to such individual under any otherwise applicable Company plan or program, including base salary, bonuses or commissions and compensation previously deferred by the executive officer.
Potential Payments Upon Termination or in Connection With a Change of Control
Employment Agreements
Pursuant to each of the Myers Agreement and the Carter Agreement, Dr. Myers and Ms. Carter, respectively, are entitled to the following benefits upon termination of their employment:
| ● | Termination for any reason: Upon the termination of such executive’s employment for any reason, such executive will receive (i) his or her Base Salary (as defined in the Myers Agreement or the Carter Agreement, as applicable) through the Exit Date (as defined in the Myers Agreement or the Carter Agreement, as applicable), (ii) any Bonuses (as defined in the Myers Agreement or the Carter Agreement, as applicable) to which he or she is entitled and has already earned for the prior fiscal year, and (iii) any other accrued or vested benefits or reimbursements through the Exit Date to which such executive is entitled to contractually or by operation of law. |
| ● | Termination upon death or Disability: In the event of the executive’s termination due to his or her death or Disability (as defined in the Myers Agreement or the Carter Agreement, as applicable), then, in addition to the payments set forth above, the executive will receive his or her pro rata portion of the Bonus such executive would have been entitled to receive for the fiscal year in which the Exit Date occurs, based upon the percentage of the fiscal year that elapsed through the Exit Date. Additionally, in the event of termination due to Disability, the executive will receive, for a period of 24 months following the Exit Date, such executive monthly COBRA premium. |
| ● | Termination without Cause or for Good Reason: In addition to the payments set forth in the first bullet above, if Dr. Myers or Ms. Carter is terminated by the Company without Cause (as defined in the Myers Agreement or the Carter Agreement, as applicable), or Dr. Myers or Ms. Carter terminates his or her employment for Good Reason (as defined in the Myers Agreement or the Carter Agreement, as applicable), he or she will be entitled to receive (i) his or her Base Salary for 2 years from the Exit Date and 2 times the current years’ Bonus, and (ii) continuation of such executive’s medical benefits for 2 years from the Exit Date (unless the executive becomes employed elsewhere during such 2 year period and is eligible to receive comparable medical benefits). |
As a condition precedent to receiving any of the foregoing benefits, Dr. Myers and/or Ms. Carter, as applicable, must first sign a Release (as defined in the Myers Agreement or the Carter Agreement, as applicable).
Mr. Dunn, pursuant to the Dunn Agreement, is also entitled to the following benefits upon termination of his employment:
| ● | Garden Leave: During any period of notice to terminate Mr. Dunn’s employment, Mr. Dunn will continue to be entitled to his basic salary and contractual benefits in the usual course. |
| ● | Payment in lieu of notice: Upon the termination of Mr. Dunn’s employment at any time, Mr. Dunn will receive payment equal to his basic salary as of the termination date which he would have been entitled to receive under the Dunn Agreement during the notice period referred to in the bullet below, less income tax and national insurance contributions. Payment in lieu of notice will not include (i) any bonus or commission payments that might otherwise have been paid to Mr. Dunn during the period for which such payment in lieu of notice is made, (ii) benefits Mr. Dunn would have been entitled to during such time, and (iii) holiday entitlement that would have accrued during such time. |
| ● | Termination: Subject to successful completion of the probationary employment period as set forth in the Dunn Agreement, and except in connection with certain “for cause” events, as set forth in Section 20.2 of the Dunn Agreement, the Company may terminate Mr. Dunn’s employment by giving at least 12 months’ prior written notice, and is obligated to continue paying Mr. Dunn his basic salary and other benefits during such notice period. |
75
The foregoing descriptions of the Myers Agreement, the Carter Agreement and the Dunn Agreement do not purport to be complete and are qualified in their entirety by reference to the complete text of the Myers Agreement, the Carter Agreement and the Dunn Agreement, copies of which are included as exhibits to this Annual Report.
Option Awards
Under the Plan, upon termination of employment for any reason, other than in the event of death or disability or for ”Cause” (as defined in the Plan), all unvested options will expire and all vested options at time of termination will generally be exercisable for 90 days following termination, subject to the terms of the Plan and the governing option agreement. If we terminate a grantee for Cause, the grantee’s right to exercise all vested and unvested the options granted to the grantee will expire immediately. Upon termination of employment due to death or disability, all the vested options at the time of termination will be exercisable for 12 months after date of termination, subject to the terms of the Plan and the governing option agreement.
Compensation Policy under the Companies Law
In general, under the Companies Law, a public company must have a compensation policy approved by the board of directors after receiving and considering the recommendations of the compensation committee. In addition, our compensation policy must be approved at least once every three years, first, by our board of directors, upon the recommendation of our compensation committee, and second, by a simple majority of the ordinary shares present, in person or by proxy, and voting (excluding abstentions) at a general meeting of shareholders, provided that either:
| ● | such majority includes at least a majority of the shares held by shareholders who are not controlling shareholders and shareholders who do not have a personal interest in such compensation policy; or |
| ● | the total number of shares of non-controlling shareholders and shareholders who do not have a personal interest in the compensation policy and who vote against the policy does not exceed two percent (2%) of the aggregate voting rights in the Company. |
Under special circumstances, the board of directors may approve the compensation policy despite the objection of the shareholders on the condition that the compensation committee and then the board of directors decide, on the basis of detailed grounds and after discussing again the compensation policy, that approval of the compensation policy, despite the objection of shareholders, is for the benefit of the company.
If a company that initially offers its securities to the public, like us, adopts a compensation policy in advance of its initial public offering, and describes it in its prospectus for such offering, then such compensation policy shall be deemed a validly adopted policy in accordance with the Companies Law requirements described above. Furthermore, if the compensation policy is established in accordance with the aforementioned relief, then it will remain in effect for a term of five years from the date such company becomes a public company.
The compensation policy must be based on certain considerations, include certain provisions and reference certain matters as set forth in the Companies Law. The compensation policy must serve as the basis for decisions concerning the financial terms of employment or engagement of office holders, including exculpation, insurance, indemnification or any monetary payment or obligation of payment in respect of employment or engagement. The compensation policy must be determined and later reevaluated according to certain factors, including: the advancement of the company’s objectives, business plan and long-term strategy; the creation of appropriate incentives for office holders, while considering, among other things, the company’s risk management policy; the size and the nature of the company’s operations; and with respect to variable compensation, the contribution of the office holder towards the achievement of the company’s long-term goals and the maximization of its profits, all with a long-term objective and according to the position of the office holder. The compensation policy must furthermore consider the following additional factors:
76
| ● | the ratio between the cost of the terms of employment of an office holder and the cost of the employment of other employees of the company, including employees employed through contractors who provide services to the company, in particular the ratio between such cost to the average and median salary of such employees of the company, as well as the impact of disparities between them on the work relationships in the company; |
| ● | if the terms of employment include variable components — the possibility of reducing variable components at the discretion of the board of directors and the possibility of setting a limit on the value of non-cash variable equity-based components; and |
| ● | if the terms of employment include severance compensation — the term of employment or office of the office holder, the terms of the office holder’s compensation during such period, the company’s performance during such period, the office holder’s individual contribution to the achievement of the company goals and the maximization of its profits and the circumstances under which he or she is leaving the company. |
The compensation policy must also include, among other things:
| ● | with the exception of office holders who report to the chief executive officer, a means of determining the variable components on the basis of long-term performance and measurable criteria; provided that the company may determine that an immaterial part of the variable components of the compensation package of an office holder shall be awarded based on non-measurable criteria, or if such amount is not higher than three months’ salary per annum, taking into account such office holder’s contribution to the company; |
| ● | the ratio between variable and fixed components, as well as the limit of the values of variable components at the time of their payment, or in the case of equity-based compensation, at the time of grant; |
| ● | a condition under which the office holder will return to the company, according to conditions to be set forth in the compensation policy, any amounts paid as part of the office holder’s terms of employment, if such amounts were paid based on information later to be discovered to be wrong, and such information was restated in the company’s financial statements; |
| ● | the minimum holding or vesting period of variable equity-based components to be set in the terms of office or employment, as applicable, while taking into consideration long-term incentives; and |
Our compensation policy is designed to promote retention and motivation of directors and executive officers, incentivize superior individual excellence, align the interests of our directors and executive officers with our long-term performance and provide a risk management tool. To that end, a portion of our executive officer compensation package is targeted to reflect our short and long-term goals, as well as the executive officer’s individual performance. On the other hand, our compensation policy includes measures designed to reduce the executive officer’s incentives to take excessive risks that may harm us in the long-term, such as limits on the value of cash bonuses and equity-based compensation, limitations on the ratio between the variable and the total compensation of an executive officer and minimum vesting periods for equity-based compensation.
Our compensation policy also addresses our executive officers’ individual characteristics (such as their respective position, education, scope of responsibilities and contribution to the attainment of our goals) as the basis for compensation variation among our executive officers and considers the internal ratios between compensation of our executive officers and directors and other employees. Pursuant to our compensation policy, the compensation that may be granted to an executive officer may include: base salary, annual bonuses and other cash bonuses (such as a signing bonus and special bonuses with respect to significant events, such as a significant partnership, collaboration agreement or the generation of positive clinical trial results or regulatory approval of one of the Company’s products), equity-based compensation and termination of service grants.
An annual cash bonus may be awarded to executive officers upon the attainment of pre-set periodic objectives and individual targets. The annual cash bonus that may be granted to our executive officers is based primarily on measurable short- and long-term criteria. A non-material part of variable compensation for executive officers may be based on qualitative or non-measurable criteria which focus on the executive officer’s contribution to the Company, subject to a maximum amount linked to the executive officer’s base salary.
77
The equity-based compensation under our compensation policy for our executive officers is designed in a manner consistent with the underlying objectives in determining the base salary and the annual cash bonus, with its main objectives being to enhance the alignment between the executive officers’ interests with our long-term interests and those of our shareholders and to strengthen the retention and the motivation of executive officers in the long term. Our compensation policy provides for equity compensation in any form permitted under our equity incentive plan then in place. The equity-based compensation shall be granted from time to time and be individually determined and awarded according to the performance, educational background, prior business experience, qualifications, role and the personal responsibilities of the executive officer.
In addition, our compensation policy contains compensation recovery provisions which allow us under certain conditions to recover bonuses paid in excess, enables our compensation committee and board of directors to approve an immaterial change in the terms of employment of an executive officer and allow us to exculpate, indemnify and insure our executive officers and directors to the maximum extent permitted by Israeli law subject to certain limitations set forth therein.
Our compensation policy also provides for compensation to the members of our board of directors in accordance with market compensation trends, provided however that in the case of an external director, such compensation will be paid in accordance with the amounts provided in the Companies Regulations (Rules Regarding the Compensation and Expenses of an External Director) of 2000, as amended by the Companies Regulations (Relief for Public Companies Traded in Stock Exchange Outside of Israel) of 2000, as such regulations may be amended from time to time.
Our compensation policy was approved by our compensation committee, our board of directors and shareholders and became effective on April 12, 2022.
Non-Employee Director Compensation
Under our non-employee directors’ compensation program, as amended, non-employee directors are entitled to receive the following cash compensation for their services:
| ● | each non-employee director receives an annual base retainer of $82,500; |
| ● | each committee chairperson receives an additional retainer of $15,000 for his or her service as a chairperson; and |
| ● | each member of a standing committee receives an additional retainer of $5,000 for such service on a standing committee. |
In addition to cash compensation, our non-employee directors are also entitled to equity awards under our director compensation policy. Each non-employee director is entitled to receive an annual award of options with a value of no less than $20,000 and no more than $60,000, with such value being determined annually at the discretion of the Compensation Committee and the Board. In addition, each non-employee director who joins the Board is granted an inaugural award of options valued at $165,000. The following table sets forth information concerning the compensation awarded to, earned by or paid to non-employee directors for the year ended December 31, 2024.
| Fees Earned or |
| Option |
|
| |
Paid in Cash | Awards(1) | Total | ||||
Name | ($) | ($) | ($) | |||
Joseph Cooper |
| 92,500 | 33,363 | 125,863 | ||
James Culverwell |
| 102,500 | 33,363 | 135,863 | ||
Dr. Dennis H. Langer |
| 97,500 | 33,363 | 130,863 | ||
Natalie Leong |
| 102,500 | 33,363 | 135,863 | ||
Michael Sember |
| 87,500 | 33,363 | 120,863 |
| (1) | Represents the grant date fair value of option awards granted to each of our non-employee directors on December 9, 2024, calculated in accordance with FASB ASC Topic 718. These options have an exercise price of $0.78 per ADS and vests in four annual installments with 20% vesting on each of December 9, 2025, 2026 and 2027 and 40% vesting on December 9, 2028. The option values were calculated using a Black-Scholes Model for pricing options. See Note 6 to Consolidated Financial Statements included in this Annual Report for all relevant valuation assumptions used to determine the grant date fair value of these options. As of December 31, 2024, the aggregate number of outstanding options held by each of our non-employee directors was 59,438 ADSs. |
78
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth information relating to the beneficial ownership of our ordinary shares (including ordinary shares represented by ADSs) as of March 10, 2025 by:
| ● | each person, or group of affiliated persons, known by us to own beneficially 5% or more of our outstanding ordinary shares; |
| ● | each of our directors and named executive officers; and |
| ● | all of our directors and officers as a group. |
Beneficial ownership is determined in accordance with the rules of the SEC and generally means sole or shared power to vote or direct the voting or to dispose or direct the disposition of any security. Unless otherwise indicated in the footnotes to this table, we believe that each of the persons named in this table has sole voting and investment power with respect to the securities indicated as being beneficially owned.
Except as indicated by footnote, the beneficial ownership information is based upon 20,585,830 ordinary shares outstanding as of March 10, 2025. A security that may be acquired by a person within 60 days of March 10, 2025, pursuant to the exercise of options or warrants are deemed to be outstanding for purpose of computing the percentage ownership of such person, but are not deemed to be outstanding for purposes of computing the percentage ownership of ordinary shares beneficially owned by any other person shown in the table. Each ADS represents one ordinary share.
Unless indicated otherwise below, the address of our directors and executive officers is c/o Quoin Pharmaceuticals Ltd., 42127 Pleasant Forest Court, Ashburn, VA 20148-7349.
| Amount and Nature of Beneficial |
| Percentage |
| |
Name and Address of Beneficial Owner | Ownership | of Class | |||
Directors and Named Executive Officers: |
|
|
|
| |
Dr. Michael Myers(1) |
| 1,049,523 |
| 4.99 | % |
Denise Carter(2) |
| 1,049,532 |
| 4.99 | % |
Joseph Cooper(3) |
| 2,602 |
| * | |
James Culverwell(4) |
| 302,919 |
| 1.46 | % |
Dr. Dennis Langer(5) |
| 2,655 |
| * | |
Natalie Leong(6) |
| 2,602 |
| * | |
Michael Sember(7) |
| 2,602 |
| * | |
Gordon Dunn(8) |
| 410,214 |
| 1.97 | % |
All directors and officers as a group (8 persons) (9) |
| 2,822,651 |
| 13.46 | % |
* Less than 1%
| (1) | Consists of (i) 602,808 ADSs held directly, (ii) 21,550 ADSs issuable the upon the exercise of options which may be exercised within 60 days of March 10, 2025, (iii) 425,165 ADSs issuable upon the exercise of 425,165 December 2024 Warrants acquired in the December 2024 Offering which may be exercised within 60 days of March 10, 2025 (does not include 685,947 ADSs issuable upon the exercise of 685,947 December 2024 Warrants due to the 4.99% beneficial ownership limitation in such Warrants). |
| (2) | Consists of (i) 602,648 ADSs held directly, (ii) 21,550 ADSs issuable upon the exercise of options which may be exercised within 60 days of March 10, 2025, and (iii) 425,334 ADSs issuable upon the exercise of 425,334 December 2024 Warrants acquired in the December 2024 Offering which may be exercised within 60 days of March 10, 2025 (does not include 685,778 ADSs issuable upon the exercise of 685,778 December 2024 Warrants due to the 4.99% beneficial ownership limitation in such Warrants). |
| (3) | Represents 2,602 ADSs issuable the upon exercise of options which may be exercised within 60 days of March 10, 2025. |
| (4) | Consists of (i) 100,317 ADSs held directly, (ii) 2,602 ADSs issuable upon the exercise of options which may be exercised within 60 days of March 10, 2025, and (iii) 200,000 ADSs issuable upon the exercise of 200,000 December 2024 Warrants acquired in the December 2024 Offering which may be exercised within 60 days of March 10, 2025. |
79
| (5) | Consists of (i) 53 ADSs held directly and (ii) 2,602 ADSs issuable upon the exercise of options which may be exercised within 60 days of March 10, 2025. |
| (6) | Represents 2,602 ADSs issuable upon the exercise of options which may be exercised within 60 days of March 10, 2025. |
| (7) | Represents 2,602 ADSs issuable upon the exercise of options which may be exercised within 60 days of March 10, 2025. |
| (8) | Represents (i) 151,077 ADSs held directly, (ii) 14,695 ADSs issuable upon the exercise of options which may be exercised within 60 days of March 10, 2025, and (iii) 244,442 ADSs issuable upon the exercise of 244,442 December 2024 Warrants acquired in the December 2024 Offering. |
| (9) | Consists of (i) 1,456,903 ADSs held directly, (ii) 70,807 ADSs issuable upon the exercise of options which may be exercised within 60 days of March 10, 2025, and (iii) 1,294,941 ADSs issuable upon the exercise of 1,294,941 December 2024 acquired in the December 2024 Offering (such number does not include 1,371,725 ADSs issuable upon the exercise of 1,371,725 December 2024 Warrants due to the 4.99% beneficial ownership limitation in such Warrants). |
Equity Compensation Plan Table
The following table summarizes our equity compensation plan information as of December 31, 2024.
|
|
| Number of securities | ||||
remaining available for | |||||||
Number of securities | Weighted-average | future issuance under equity | |||||
to be issued upon exercise | exercise price of | compensation plans | |||||
of outstanding options, | outstanding options, | (excluding securities reflected | |||||
Plan category | warrants and rights(1) | warrants and rights(2) | in column (a)(1) | ||||
| (a) |
| (b) |
| (c) | ||
Equity compensation plans approved by security holders |
| 1,943,787 | $ | 4.29 |
| 7,253,490 | |
Equity compensation plans not approved by security holders |
| — |
| — |
| — | |
Total |
| 1,943,787 | $ | 4.29 |
| 7,253,490 | |
| (1) | Represents the number of ADSs issuable upon the exercise of options. |
| (2) | Represents the exercise price per ADS. |
Item 13. Certain Relationships and Related Transactions, and Director Independence
Director Independence
Under the corporate governance standards of Nasdaq, a majority of our directors must meet the independence requirements specified in those rules. The Board determined that Joseph Cooper, James Culverwell, Dr. Dennis Langer, Natalie Leong, and Michael Sember, qualify as independent directors, as such term is defined under Nasdaq listing rules.
Certain Relationships and Related Transactions
Due to the limited funding of Quoin Inc. prior to the consummation of the Merger, the compensation, including salary, office and car allowances and other benefits, due to Dr. Myers and Ms. Carter under their respective employment agreements, as well as reimbursement of expenses and other amounts paid by Dr. Myers and Ms. Carter to third parties on behalf of Quoin Inc., were not paid by Quoin Inc. to Dr. Myers and Ms. Carter, and were accrued as indebtedness to Dr. Myers and Ms. Carter. Following the closing of the Merger, Quoin Inc. began making payments of $25,000 per month to each of Dr. Myers and Ms. Carter to repay the above-described non-interest-bearing indebtedness. We repaid $300,000 and $300,000 of such indebtedness to Dr. Myers, and $300,000 and $300,000 to Ms. Carter, during the years ended December 31, 2023 and 2024, respectively. As of December 31, 2024, approximately $1,659,000 and $1,265,000 of such indebtedness was outstanding to Dr. Myers and Ms. Carter, respectively.
On October 2 2020, Quoin Inc. issued promissory notes (the “2020 Notes”) to five noteholders, including our directors, Messrs. Langer and Culverwell (collectively, “2020 Noteholders”). The 2020 Notes were mandatorily convertible into 432 ADSs, subject to adjustment and were converted in 2021. The ADSs issued to the 2020 Noteholders did not include accrued interest. Two of the five 2020 Noteholders received their amount due during the year ended December 31, 2022 and the Company’s estimate of the liability to the remaining three 2020 Noteholders (including Messrs Langer and Culverwell) was estimated to be $1,146,000 as of December 31, 2024 and December 31, 2023.
80
On December 23, 2024, we completed the December 2024 Offering of our ordinary shares represented by ADSs, Series F Warrants to purchase ordinary shares represented by ADSs, Series G Warrants to purchase ordinary shares represented by ADSs and pre-funded warrants to purchase ordinary shares represented by ADSs. The Company received aggregate gross proceeds from the December 2024 Offering of approximately $6.8 million, before deducting placement agent fees and other offering expenses. Dr. Myers, Ms. Carter, Mr. Dunn and Mr. Culverwell purchased an aggregate of 1,333,333 of our ADSs and accompanying Series F Warrants and Series G Warrants to purchase an aggregate of 2,666,666 of our ADSs, for a total purchase price of approximately $600,000, at the public offering price and on the same terms as the other purchasers in the December 2024 Offering.
Item 14. Principal Accountant Fees and Services
Marcum LLP (“Marcum”) has served as the Company’s independent registered public accounting firm since September 1, 2022.
The following table sets forth the aggregate accounting fees paid by us to Marcum for all services, including audit services, for the years ended December 31, 2024 and 2023, as applicable.
| Year Ended |
| Year Ended | |||
December 31, 2024 | December 31, 2023 | |||||
Type of Fees (a) (in thousands) |
|
|
|
| ||
Audit Fees | $ | 350 | $ | 244 | ||
Audit-Related Fees | — |
| — | |||
Tax Fees | — | — | ||||
All Other Fees | — |
| — | |||
Total | $ | 350 | $ | 244 | ||
| (a) | The aggregate fees included in Audit Fees are fees billed for the fiscal years. |
Audit Fees. Audit fees refer to the aggregate fees, including expenses, for the audit of our annual financial statements and review of financial statements included in our quarterly reports and other services that are normally provided in connection with statutory and regulatory filings or engagements.
Audit-Related Fees. Audit-Related fees refer to the aggregate fees, including expenses, for assurance and related services that are reasonably related to the performance of the audit or review of our financial statements not reported under “Audit Fees” above.
Tax Fees. Our independent registered public accounting firm did not provide any tax services during the periods.
All Other Fees. Our independent registered public accounting firm did not provide any “other services” during the periods.
Pre-Approval Policy
Our audit committee has a pre-approval policy for the engagement of our independent registered public accounting firm to perform audit and non-audit services. Pursuant to this policy, which is designed to assure that such engagements do not impair the independence of our auditors, the audit committee pre-approves annually a catalog of specific audit and non-audit services in the categories of audit services, audit-related services and tax services, if any, that may be performed by our independent registered public accounting firm. Unless the specific service has been previously pre-approved with respect to that year, the audit committee must approve the permitted service before the independent registered public accounting firm is engaged to perform it.
81
PART IV
Item 15. Exhibit and Financial Statement Schedules
(a)(1) Financial Statements
As part of this Annual Report, the consolidated financial statements are listed in the accompanying index to financial statements on page F-1.
(a)(2) Financial Statement Schedules.
All financial statement schedules have been omitted because they are not applicable, not required or the information required is shown in the financial statements or the notes thereto.
(a)(3) Exhibits.
The following is a list of exhibits filed as part of this Annual Report.
Exhibit No. |
| Exhibit Description |
2.1 | ||
2.2 | ||
2.3 | ||
2.4 | ||
2.5 | ||
2.6 | ||
2.7 | ||
2.8 | ||
2.9 | ||
2.10 | ||
2.11 | ||
3.1* | Amended and Restated Articles of Association of Quoin Pharmaceuticals Ltd., as amended | |
4.1 | ||
4.2 | Specimen American Depositary Receipt (included in Exhibit 2.1). | |
4.3 |
82
4.4 | ||
4.5 | ||
4.6 | ||
4.7 | ||
4.8 | ||
4.9 | ||
4.10 | ||
4.11 | ||
4.12 | ||
4.13 | ||
4.14 | ||
4.15* | ||
4.16 | ||
4.17 | ||
4.18 | ||
4.19 | ||
4.20 | ||
4.21 | ||
4.22 | ||
4.23 | ||
10.1# | ||
10.2# | ||
10.3# | ||
10.4# | ||
10.5# | ||
10.6# | ||
10.7 | ||
10.8 | ||
10.9 | ||
10.10 | ||
10.11 |
83
10.12 | ||
10.13 | ||
10.14 | ||
10.15 | ||
10.16 | ||
10.17 | ||
10.18 | ||
10.19 | ||
10.20 | ||
10.21 | ||
10.22 | ||
10.23 | ||
10.24 | ||
10.25 | ||
10.26 | ||
10.27 | ||
10.28 | ||
10.29 | ||
10.30 | ||
10.31 | ||
10.32 | ||
10.33 | ||
10.34# | ||
10.35# |
84
10.36 | ||
10.37 | ||
10.38 | ||
10.39 | ||
10.40 | ||
10.41 | ||
10.42 | ||
10.43 | ||
10.44* | ||
10.45* | ||
10.46* | ||
14.1 | ||
19.1* | ||
21.1 | ||
23.1* | ||
31.1* | ||
31.2* | ||
32.1* | Certification of Chief Executive Officer pursuant to 18 U.S.C. § 1350. | |
32.2* | Certification of Chief Financial Officer pursuant to 18 U.S.C. § 1350. | |
97.1 | ||
101* | Information formatted in Inline Extensible Business Reporting Language (XBRL): (i) Consolidated Balance Sheets, (ii) Consolidated Statements of Operations, (iii) Consolidated Statements of Shareholders’ Equity, (iv) Consolidated Statements of Cash Flows, and (v) Notes to Consolidated Financial Statements. | |
104* | Cover Page Interactive Data File (Embedded within the Inline XBRL document and included in Exhibit 101) |
* Filed herewith
# Indicates management contract or compensatory plan or arrangement.
Item 16. Form 10-K Summary
None.
85
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Date: March 13, 2025 | QUOIN PHARMACEUTICALS LTD. | |
By: | /s/ Dr. Michael Myers | |
Name: | Dr. Michael Myers | |
Title: | Chief Executive Officer | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant in the capacities and on the dates indicated.
Signature |
| Title |
| Date |
/s/ Dr. Michael Myers | Chairman and Chief Executive Officer | March 13, 2025 | ||
Dr. Michael Myers | (Principal Executive Officer) | |||
/s/ Gordon Dunn | Chief Financial Officer | |||
Gordon Dunn | (Principal Financial Officer and Principal Accounting Officer) | March 13, 2025 | ||
/s/ Denise Carter | ||||
Denise Carter | Director and Chief Operating Officer | March 13, 2025 | ||
/s/ Joseph Cooper | ||||
Joseph Cooper | Director | March 13, 2025 | ||
/s/ James Culverwell | ||||
James Culverwell | Director | March 13, 2025 | ||
/s/ Dennis Langer | ||||
Dennis Langer | Director | March 13, 2025 | ||
/s/ Natalie Leong | ||||
Natalie Leong | Director | March 13, 2025 | ||
/s/ Michael Sember | ||||
Michael Sember | Director | March 13, 2025 |
86
QUOIN PHARMACEUTICALS LTD.
Contents
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors of
Quoin Pharmaceuticals Ltd.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Quoin Pharmaceuticals Ltd. (the “Company”) as of December 31, 2024 and 2023, the related consolidated statements of operations, shareholders’ equity and cash flows for each of the two years in the period ended December 31, 2024, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2024 and 2023, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2024, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
Critical audit matters are matters arising from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. We determined that there are no critical audit matters.
/s/ Marcum LLP
Marcum LLP
We have served as the Company’s auditor since 2020
March 13, 2025
F-2
Consolidated Balance Sheets
December 31, | December 31, | |||||
| 2024 |
| 2023 | |||
ASSETS |
|
|
|
| ||
Current assets: |
|
|
|
| ||
Cash and cash equivalents | $ | | $ | | ||
Investments | | | ||||
Prepaid expenses and other current assets |
| |
| | ||
Total current assets |
| |
| | ||
Prepaid expenses - long term |
| |
| | ||
Intangible assets, net |
| |
| | ||
Total assets | $ | | $ | | ||
LIABILITIES AND SHAREHOLDERS’ EQUITY |
|
|
|
| ||
Current liabilities: |
|
|
|
| ||
Accounts payable | $ | | $ | | ||
Accrued expenses |
| |
| | ||
Accrued interest and financing expense |
| |
| | ||
| |
| | |||
Total current liabilities | | | ||||
| | |||||
Total liabilities | $ | | $ | | ||
Commitments and contingencies |
|
|
|
| ||
Shareholders’ equity: |
|
|
|
| ||
Ordinary shares, | $ | | $ | | ||
Additional paid in capital |
| |
| | ||
Accumulated deficit |
| ( |
| ( | ||
Total shareholders’ equity |
| |
| | ||
Total liabilities and shareholders’ equity | $ | | $ | | ||
The accompanying footnotes are an integral part of these consolidated financial statements
F-3
Consolidated Statements of Operations
Years Ended December 31, | ||||||
| 2024 |
| 2023 | |||
Operating expenses |
|
| ||||
General and administrative | $ | | $ | | ||
Research and development |
| |
| | ||
Total operating expenses |
| | | |||
|
| |||||
Other (income) and expenses |
|
|
|
| ||
Unrealized (gain) loss | ( | | ||||
Realized and accrued interest income | ( | ( | ||||
Total other income |
| ( |
| ( | ||
Net loss | $ | ( | $ | ( | ||
Loss per ADS |
|
|
|
| ||
Loss per ADS | ||||||
Basic | $ | ( | $ | ( | ||
Fully-diluted | $ | ( | $ | ( | ||
Weighted average number of ADS’s outstanding |
|
|
|
| ||
Basic |
| |
| | ||
Fully-diluted | | | ||||
The accompanying footnotes are an integral part of these consolidated financial statements
F-4
Consolidated Statements of Shareholders’ Equity
Years Ended December 31, 2024 and 2023
|
|
| Additional | ||||||||||||||||
Ordinary | No Par | Treasury | Paid in | Accumulated | |||||||||||||||
| Shares |
| ADS’s |
| Value |
| Stock |
| Capital |
| Deficit |
| Total | ||||||
Balance at December 31, 2022 | | | $ | — | $ | ( | $ | | $ | ( | $ | | |||||||
Net loss |
| — | — | — |
| — |
| — |
| ( |
| ( | |||||||
Stock based compensation |
| — | — | — |
| — |
| |
| — |
| | |||||||
Retirement of Treasury Stock | — | — | — | | ( | — | — | ||||||||||||
Issuance of ADS and Pre-Funded Warrants, net |
| | | — |
| — |
| |
| — |
| | |||||||
Balance at December 31, 2023 | | | $ | — | $ | — | $ | | $ | ( | $ | | |||||||
Net loss | — | — | — | — | — | ( | ( | ||||||||||||
Stock based compensation | — | — | — | — | | — | | ||||||||||||
Issuance of ADS and Pre-Funded Warrants - March 7, 2024, net | | | — | — | | — | | ||||||||||||
Issuance of ADS and Pre-Funded Warrants - December 23, 2024, net | | | — | — | | — | | ||||||||||||
Balance at December 31, 2024 | | | $ | — | $ | — | $ | | $ | ( | $ | | |||||||
The accompanying footnotes are an integral part of these consolidated financial statements
F-5
Consolidated Statements of Cash Flows
Years Ended December 31, | ||||||
| 2024 |
| 2023 | |||
Cash flows used in operating activities: |
|
| ||||
Net loss |
| $ | ( | $ | ( | |
Stock based compensation |
| |
| | ||
Amortization of intangibles | | | ||||
Asset impairment | — | | ||||
Unrealized gain and accrued interest on investments | ( | ( | ||||
Changes in assets and liabilities: |
|
|
|
| ||
Decrease in accounts payable and accrued expenses |
| |
| | ||
(Increase) decrease in prepaid expenses and other assets |
| ( |
| | ||
Net cash used in operating activities |
| $ | ( | $ | ( | |
Cash flows provided (used in) investing activities: |
|
|
|
| ||
Purchase of investments | $ | ( | $ | ( | ||
Proceeds from maturity of investments | | | ||||
Net cash provided (used in) investing activities |
| $ | ( | $ | | |
Cash flows provided by financing activities: |
|
|
|
| ||
Payments of deferred financing costs |
| $ | — | $ | ( | |
Payment of amounts due to officers |
| ( |
| ( | ||
Proceeds from sale of equity securities, net |
| |
| | ||
Net cash provided by financing activities |
| $ | | $ | | |
Net change in cash and cash equivalents: |
| |
| ( | ||
Cash and cash equivalents - beginning of year |
| |
| | ||
Cash and cash equivalents - end of year |
| $ | | $ | | |
Supplemental information - Non cash items: |
|
| ||||
Offering expenses associated with warrant modification | $ | | $ | | ||
Accrued offering expenses included in additional paid in capital | $ | | $ | — | ||
The accompanying footnotes are an integral part of these consolidated financial statements
F-6
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
NOTE 1 – ORGANIZATION AND BUSINESS
Quoin Pharmaceuticals Ltd. (“Quoin Ltd.,” or the “Company”), formerly known as Cellect Biotechnology Ltd. (“Cellect”), is the holding company for Quoin Pharmaceuticals, Inc., a Delaware corporation (“Quoin Inc.”). Quoin Inc. was incorporated in Delaware on March 5, 2018. On October 28, 2021, Cellect completed the business combination with Quoin Inc., with Quoin Inc. surviving as a wholly-owned subsidiary of Cellect (the “Merger”). Immediately after completion of the Merger, Cellect changed its name to “Quoin Pharmaceuticals Ltd.”
The Company is a late-stage clinical specialty pharmaceutical company focused on the development and commercialization of therapeutic products that treat rare and orphan diseases for which there are currently very limited or no approved treatments or cures. The Company’s initial focus is on the development of products, using the Company’s proprietary owned and in-licensed drug delivery technologies, that could help address rare genetic diseases. The Company’s lead product, QRX003, is under clinical development as a potential treatment for Netherton Syndrome (“NS”), a rare hereditary genetic disease. QRX003 is currently being tested in three regulatory clinical studies under an open Investigational New Drug (“IND”) application with the Food and Drug Administration (“FDA”). The Company has opened five clinical sites in the United States (“US”) and intend to open a sixth clinical site at Northwestern University. The Company is expanding its trials internationally into the Middle East, the United Kingdom and additional countries in Europe, including Spain and Germany. QRX003 is currently being tested in a pediatric NS patient at the Children’s Hospital in Dublin, Ireland and the Company intends to expand this study to include additional children with NS in Spain, the United Kingdom and potentially other countries. QRX003 is also being developed as a potential treatment for Peeling Skin Syndrome with the first subject being treated in New Zealand. In addition, the Company entered into two separate Research Agreements with the Queensland University of Technology (“QUT”), under which the Company has obtained an option for global licenses to QRX007 for the potential treatment of NS and QRX008 for the potential treatment of scleroderma, as well as a Research Agreement with the University College Cork (“UCC”) for the development of novel topical formulations of rapamycin (sirolimus) as potential treatments for a number of rare and orphan diseases. The Company is initiating the development of novel topical formulations of rapamycin using its in-licensed technology as potential treatments for microcystic lymphatic malformations, venous malformations and angifibromas. Other development products in the Company’s pipeline include QRX004 as a potential treatment for Recessive Dystrophic Epidermolysis Bullosa (“RDEB”). To date, no products have been commercialized and no revenue has been generated.
NOTE 2 - LIQUIDITY RISKS AND OTHER UNCERTAINTIES
The Company has incurred net losses every year since inception and has an accumulated deficit of approximately $
Additional financing will still be required to complete the research and development of the Company’s therapeutic targets and its other operating requirements until it achieves commercial profitability, if ever. Such financing may not be available at acceptable terms, if at all. If the Company is unable to obtain additional funding when it becomes necessary, the development of its product candidates will be impacted and the Company would likely be forced to delay, reduce, or terminate some or all of its development programs, all of which could have a material adverse effect on the Company’s business, results of operations and financial condition.
Other risks and uncertainties:
The Company is subject to risks common to development stage biopharmaceutical companies including, but not limited to, new technological innovations, dependence on key personnel, protection of proprietary technology, compliance with government regulations, product liability, pre-clinical and clinical trial outcome risks, regulatory approval risks, uncertainty of market acceptance and additional financing requirements.
The Company’s products require approval or clearance from the FDA prior to commencing commercial sales in the United States. There can be no assurance that the Company’s products will receive all of the required approvals or clearances. Approvals or clearances are also required in foreign jurisdictions in which the Company may license or sell its products.
F-7
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
There can be no assurance that the Company’s products, if approved, will be accepted in the marketplace, nor can there be any assurance that any future products can be developed or manufactured at an acceptable cost and with appropriate performance characteristics, or that such products will be successfully marketed.
The Company is also dependent on several third party suppliers, in some cases a single source supplier including the contract research organization managing both of the Company’s current clinical studies, the supplier of the active pharmaceutical ingredient (API), as well as the contract manufacturer of the drug product for clinical development.
On April 29, 2024, the Company received a deficiency letter from the Listing Qualifications Department of Nasdaq notifying the Company that for the preceding 31 consecutive business days (March 14, 2024 through April 26, 2024), the Company’s ADSs did not maintain a minimum closing bid price of $1.00 (“Minimum Bid Price Requirement”) per ADS as required by Nasdaq Listing Rule 5550(a)(2). In accordance with Nasdaq Listing Rule 5810(c)(3)(A), the Company had a compliance period of 180 calendar days, or until October 28, 2024, to regain compliance with Nasdaq Listing Rule 5550(a)(2). On October 16, 2024, the Company submitted a letter to Nasdaq requesting an additional 180-day grace period to regain compliance with the Minimum Bid Price Requirement. On October 29, 2024, the Company received a letter from the Listing Qualifications Department of Nasdaq granting the Company an additional 180 calendar day grace period, or until April 28, 2025, to regain compliance. The Staff’s determination in granting the Company the extension was based on the Company meeting the continued listing requirement for market value of publicly held shares and all other applicable requirements for initial listing on the Nasdaq Capital Market with the exception of the Minimum Bid Price Requirement, and the Company’s written notice of its intention to cure the deficiency during the second compliance period by effecting a reverse split, if necessary. Compliance may be achieved without further action if the closing bid price of the Company’s ADS is at or above $
If the Company cannot regain compliance with the Minimum Bid Price Requirement or if the Company otherwise fails to meet any of Nasdaq’s listing standards, the Company’s ADSs will be subject to delisting. If that were to occur, the Company’s ADSs would be subject to rules that impose additional sales practice requirements on broker-dealers who sell the Company’s securities. The additional burdens imposed upon broker-dealers by these requirements could discourage broker-dealers from effecting transactions in the Company’s ADSs. This would adversely affect the ability of investors to trade the Company’s ADSs and would adversely affect the value of the Company’s ADSs. Delisting from Nasdaq would cause the Company to pursue eligibility for trading of the Company’s ADSs on other markets or exchanges, or on an over-the-counter market. In such case, the Company’s stockholders’ ability to trade or obtain quotations of the market value of the Company’s ADSs would be severely limited because of lower trading volumes and transaction delays. These factors could contribute to lower prices and larger spreads in the bid and ask prices of these securities. There can be no assurance that the Company’s ADSs, if delisted from the Nasdaq, would be listed on a national securities exchange, a national quotation service or the over-the-counter markets. Delisting from the Nasdaq could also result in negative publicity, adversely affect the market liquidity of the Company’s ADSs, decrease securities analysts’ coverage of the Company or diminish investor, supplier and employee confidence.
F-8
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
NOTE 3 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation:
The accompanying consolidated financial statements have been prepared in accordance with generally accepted accounting principles in the United States (“U.S. GAAP”), which have been consistently applied. All intercompany accounts and transactions have been eliminated in consolidation.
Use of estimates:
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts in the financial statements and accompanying notes. Actual results could materially differ from those estimates. Management considers many factors in developing the estimates and assumptions that are used in the preparation of these financial statements including: expected business and operational changes, sensitivity and volatility associated with the assumptions used in developing estimates, and whether historical trends are expected to be representative of future trends. The estimation process often may yield a range of potentially reasonable estimates of the ultimate future outcomes and management must select an amount that falls within that range of reasonable estimates. Estimates are used in the following areas, among others: settlement of debt or other obligations, stock-based compensation, research and development expense recognition, intangible asset estimated useful lives and impairment assessments, allowances of deferred tax assets, and cash flow assumptions regarding going concern considerations.
Cash and cash equivalents:
The Company considers all highly liquid investments and short-term debt instruments with original maturities of three months or less to be cash equivalents. The Company, from time to time during the periods presented, has had bank account balances in excess of federally insured limits where substantially all cash is held in the United States. The Company has not experienced losses in such accounts. The Company believes that it is not subject to unusual credit risk beyond the normal credit risk associated with commercial banking relationships.
Warrants:
The Company classifies as equity any contracts that (i) require physical settlement or net-share settlement or (ii) provide the Company with a choice of net-cash settlement or settlement in its own shares (physical settlement or net-share settlement) provided that such contracts are indexed to the Company’s own stock. The Company classifies as assets or liabilities any contracts that (i) require net-cash settlement (including a requirement to net cash settle the contract if an event occurs and if that event is outside the Company’s control) or (ii) give the counterparty a choice of net-cash settlement or settlement in shares (physical settlement or net-share settlement).
The Company assesses classification of its warrants and other free-standing derivatives at each reporting date to determine whether a change in classification between assets, liabilities and equity is required. The Company evaluated the warrants to assess their proper classification using the applicable criteria enumerated under U.S. GAAP and determined that such warrants meet the criteria for equity classification in the accompanying consolidated balance sheets as of December 31, 2024 and December 31, 2023, respectively.
Investments:
Investments as of December 31, 2024 and 2023 consist of U.S. Treasury Bills and Notes, which are classified as trading securities, totaling $
F-9
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
Long-lived assets:
Long-lived assets are comprised of acquired technology and licensed rights to use technology, which are considered platform technology with alternative future uses beyond the current products in development. Such intangible assets are being amortized on a straight-line basis over their expected useful life of
The Company assesses the impairment for long-lived assets whenever events or circumstances indicate the carrying value may not be recoverable. Factors the Company considers that could trigger an impairment review include the following:
| ● | Significant changes in the manner of the Company’s use of the acquired assets or the strategy for its overall business, |
| ● | Significant underperformance relative to expected historical or projected development milestones, |
| ● | Significant negative regulatory or economic trends, and |
| ● | Significant technological changes which could render the platform technology obsolete. |
The Company recognizes impairment when the sum of the expected undiscounted future cash flows is less than the carrying amount of the asset. Impairment losses, if any, are measured as the excess of the carrying amount of the asset over its estimated fair value. During the year ended December 31, 2024, there were
Operating Segment:
The Company operates in
In addition to the significant expense categories included within consolidated net loss presented on the Company’s Consolidated Statements of Operations, see below for disaggregated amounts that comprise research and development expenses:
Year Ended December 31, | ||||||
| 2024 |
| 2023 | |||
External clinical development expenses | $ | | $ | | ||
Personnel related and stock-based compensation |
| |
| | ||
Other research and development expenses |
| |
| | ||
Total research and development expenses | $ | | $ | | ||
Research and development:
Research and development costs are expensed as incurred. Research and development expenses include personnel costs associated with research and development activities, including third-party contractors to perform research, conduct clinical trials and manufacture drug supplies and materials. The Company accrues for costs incurred by external service providers, including contract research organizations and clinical investigators, based on its estimates of service performed and costs incurred. These estimates include the level of services performed by third parties, patient enrollment in clinical trials when applicable, administrative costs incurred by third parties, and other indicators of the services completed. Based on the timing of amounts invoiced by service providers, the Company may also record payments made to those providers as prepaid expenses that will be recognized as expenses in future periods as the related services are rendered.
F-10
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
Income taxes:
The Company accounts for its income taxes using the asset and liability method. Accordingly, deferred tax assets and liabilities are recognized for future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases and tax credit carry-forwards. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. The Company maintains a full valuation allowance on its existing deferred tax assets.
The Company also accounts for uncertain tax positions using the more-likely-than-not threshold for financial statement recognition and measurement of a tax position taken in the Company’s income tax returns. As of December 31, 2024 and 2023, the Company had
Stock based compensation:
The Company recognizes compensation costs resulting from the issuance of stock-based awards to employees, non-employees and directors as an expense in the consolidated statements of operations over the requisite service period based on a measurement of fair value for each stock-based award. The fair value of each option grant is estimated as of the date of grant using the Black-Scholes option-pricing model, net of actual forfeitures. The fair value is amortized as compensation cost on a straight-line basis over the requisite service period of the awards, which is generally the vesting period.
Since the Company has a limited history of trading as a public company, the Company’s expected stock volatility is based on a weighting of its historical volatility along with a group of a publicly traded set of peer companies. The Company utilizes the simplified method to estimate the expected term. The risk-free interest rate was determined by reference to the U.S. Treasury yield curve in effect at the time of grant of the award for time periods approximately equal to the expected term of the award. The expected dividend yield was assumed to be zero as the Company has not paid dividends since its inception and does not anticipate paying dividends in the foreseeable future.
Fair value of financial instruments:
The Company considers its cash and cash equivalents, investments, accounts payable, accrued expenses to meet the definition of financial instruments. The carrying amounts of these financial instruments approximated their fair values due to the short maturities.
The Company measures fair value as required by ASC Topic 820, Fair Value Measurements and Disclosures (“ASC Topic 820”). ASC Topic 820 defines fair value, establishes a framework and gives guidance regarding the methods used for measuring fair value, and expands disclosures about fair value measurements. ASC Topic 820 clarifies that fair value is an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants.
Earnings (loss) per share:
The Company reports loss per share in accordance with ASC 260-10, Earnings Per Share, which provides for calculation of “basic” and “diluted” earnings per share. Basic earnings per share includes no dilution and is computed by dividing net income or loss available to shareholders by the weighted average shares outstanding for the period. Diluted earnings per share reflect the potential dilution of securities that could share in the earnings of an entity. The calculation of diluted net earnings (loss) per share gives effect to ordinary shares equivalents; however, potential shares are excluded if their effect is anti-dilutive.
For the year ended December 31, 2024, the number of shares excluded from the diluted net earnings (loss) per share included outstanding warrants to purchase
F-11
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
in the denominator would be anti-dilutive. For the year ended December 31, 2024 basic and diluted net earnings (loss) per share included
Recent Accounting Pronouncements:
In November 2023, the FASB issued ASU 2023-07, Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures, requiring public entities to disclose information about their reportable segments’ significant expenses and other segment items on an interim and annual basis. Public entities with a single reportable segment are required to apply the disclosure requirements in ASU 2023-07, as well as all existing segment disclosures and reconciliation requirements in ASC 280 on an interim and annual basis. The Company adopted ASU 2023-07 during the year ended December 31, 2024. Refer to segment reporting above in (Note 2).
In December 2023, the FASB issued ASU No. 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures. The standard is intended to enhance the transparency and decision usefulness of income tax disclosures primarily through changes to the rate reconciliation and income taxes paid information. The Company adopted ASU No. 2023-09 effective January 1, 2025, while the new standard does require further disaggregation of the income tax footnote, the Company currently does not expect the adoption of the new standard to have a material effect on its consolidated financial statements.
In November 2024, the FASB issued ASU 2024-03, Income Statement-Reporting Comprehensive Income-Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses, requiring public entities to disclose additional information about specific expense categories in the notes to the financial statements on an interim and annual basis. ASU 2024-03 is effective for fiscal years beginning after December 15, 2026, and for interim periods beginning after December 15, 2027, with early adoption permitted. The Company is currently evaluating the impact of adopting ASU 2024-03.
NOTE 4 – ACCRUED INTEREST AND FINANCING EXPENSE
On October 2, 2020, Quoin Inc. issued promissory notes (the “2020 Notes”) to certain investors (“2020 Noteholders”). The 2020 Notes were mandatorily convertible into
There was
NOTE 5 - FAIR VALUE OF FINANCIAL INSTRUMENTS
The Company applies fair value accounting for all assets and liabilities that are recognized or disclosed at fair value in the financial statements on a recurring basis. Fair value is defined as the price that would be received from selling an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. When determining the fair value measurements for assets and liabilities the Company considers the principal or most advantageous market in which it would transact and the market-based risk measurements or assumptions that market participants would use in pricing the asset or liability, such as risks inherent in valuation techniques, transfer restrictions and credit risk. For certain instruments, including cash and cash equivalents, accounts payable, and accrued expenses, it was estimated that the carrying amount approximated fair value because of the short maturities of these instruments.
Fair value is estimated using various valuation models, which utilize certain inputs and assumptions that market participants would use in pricing the asset or liability. The inputs and assumptions used in valuation models are classified in the fair value hierarchy as follows:
Level 1: Quoted prices (unadjusted) in active markets that are accessible at the measurement date for assets or liabilities. The fair value hierarchy gives the highest priority to Level 1 inputs.
Level 2: Quoted market prices for similar instruments in an active market; quoted prices for identical or similar assets and liabilities in markets that are not active; and model-derived valuations inputs of which are observable and can be corroborated by market data.
F-12
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
Level 3: Unobservable inputs and assumptions that are supported by little or no market activity and that are significant to the fair value of the asset and liability. The fair value hierarchy gives the lowest priority to Level 3 inputs.
In determining the appropriate hierarchy levels, the Company analyzes the assets and liabilities that are subject to fair value disclosure. Financial assets and liabilities are classified in their entirety based on the lowest level of input that is significant to their fair value measurement.
The following table presents the Company’s assets and liabilities that are measured at fair value on a recurring basis by fair value hierarchy at December 31, 2024 and 2023:
December 31, 2024 |
| Level 1 |
| Level 2 |
| Level 3 |
| Total | ||||
US Treasury Bills and Notes |
| $ | |
| $ | — | $ | — | $ | | ||
Total US Treasury Bills and Notes Asset |
| $ | |
| $ | — | $ | — | $ | | ||
December 31, 2023 |
| Level 1 |
| Level 2 |
| Level 3 |
| Total | ||||
US Treasury Bills and Notes | $ | | $ | — | $ | — | $ | | ||||
Total US Treasury Bills and Notes Asset | $ | | $ | — | $ | — | $ | | ||||
NOTE 6 – STOCK BASED COMPENSATION
In March 2022, the Board of Directors of the Company approved the Amended and Restated Equity Incentive Plan which was approved by the shareholders at the Company’s Annual General Meeting of Shareholders held on April 12, 2022 (the “Amended Plan”). The Amended Plan increased the number of ordinary shares reserved for issuance under such equity incentive plan to
The following table summarizes stock-based activities under the Amended Plan:
|
| Weighted |
| Weighted | |||
Average | Average | ||||||
ADS Underlying | Exercise | Contractual | |||||
Options | Price | Terms | |||||
Outstanding at December 31, 2022 |
| | $ | |
| ||
Granted | | | — | ||||
Outstanding at December 31, 2023 | | $ | | ||||
Granted |
| | |
| — | ||
Outstanding at December 31, 2024 |
| | $ | |
| ||
Exercisable options at December 31, 2024 |
| | $ | |
| ||
The intrinsic value of outstanding options at December 31, 2024 was $
F-13
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
Stock options granted during the years ended December 31, 2024 and 2023 were valued using the Black-Scholes option-pricing model with the following weighted average assumptions:
| December 31, | December 31, |
| ||||
2024 |
| 2023 |
| ||||
Expected volatility |
| | % | | % | ||
Risk-free interest rate |
| | % | | % | ||
Expected dividend yield |
| | % | | % | ||
Expected life of options in years |
| ||||||
Exercise Price | $ | | $ | | |||
Fair value of common stock | $ | | $ | | |||
Estimate fair value of option | $ | | $ | | |||
Stock based compensation expense was approximately $
At December 31, 2024, the total unrecognized compensation expense related to non-vested options was approximately $
NOTE 7 – PREPAID EXPENSES AND OTHER CURRENT ASSETS
Prepaid expenses and other current assets are as follows:
| December 31, | December 31, | ||||
2024 |
| 2023 | ||||
Prepaid R&D costs | $ | | $ | | ||
Prepaid insurance |
| |
| | ||
Prepaid expense |
| |
| | ||
Deferred offering costs (note 13) | — | | ||||
Total | $ | | $ | | ||
Less: Short-term portion | ( | ( | ||||
Long-term portion | $ | | $ | | ||
NOTE 8 – ACCRUED EXPENSES
Accrued expenses are as follows:
| December 31, | December 31, | ||||
2024 |
| 2023 | ||||
Research contract expenses (note 12) | $ | | $ | | ||
Payroll (note 11) | | | ||||
Payroll taxes (note 11) | | | ||||
Professional fees | | | ||||
Other expenses |
| |
| | ||
Total | $ | | $ | | ||
F-14
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
NOTE 9 –IN-LICENSED TECHNOLOGY
Polytherapeutics:
In March 2018, Quoin Inc. entered into a securities purchase agreement (the “Acquisition Agreement”), in which it agreed to acquire all of the equity interests in Polytherapeutics, Inc. (the “Seller” or “Polytherapeutics”) for $
Skinvisible:
In October 2019, Quoin Inc. entered into the Exclusive Licensing Agreement (as amended from time to time, the “License Agreement”) with Skinvisible Pharmaceuticals, Inc. (“Skinvisible”), under which Skinvisible granted the Company an exclusive royalty-bearing license relating to the production and manufacture of prescription drug products related to certain patents held by Skinvisible, including those related to QRX003 and QRX004. The Company made Skinvisible a one-time non-refundable, non-creditable license fee of $
NOTE 10 - INTANGIBLE ASSETS
Intangible assets are as follows:
| December 31, | December 31, | ||||
2024 |
| 2023 | ||||
Technology license – Skinvisible | $ | | $ | | ||
Total cost |
| |
| | ||
Accumulated amortization |
| ( |
| ( | ||
Net book value | $ | | $ | | ||
The Company recorded amortization expense of approximately $
NOTE 11 – RELATED PARTY TRANSACTIONS
Due to Officers/Founders:
Due to the limited funding of Quoin Inc. prior to the consummation of the Merger, the compensation, including salary, office and car allowances and other benefits, due to Dr. Myers and Ms. Carter under their respective employment agreements, as well as reimbursement of expenses and other amounts paid by Dr. Myers and Ms. Carter to third parties on behalf of Quoin Inc., were not paid by Quoin Inc. to Dr. Myers and Ms. Carter, and were accrued as indebtedness to Dr. Myers and Ms. Carter. Following the closing of the Merger, Quoin Inc. began making of $
F-15
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
Ms. Carter in the year ending December 31, 2024 and 2023, respectively. As of December 31, 2024, approximately $
Amounts due to officers at December 31, 2024 and 2023 consisted of the following:
| December 31, | December 31, | ||||
2024 |
| 2023 | ||||
$ | | $ | | |||
( | ( | |||||
$ | | $ | | |||
Insider Participation in December 2024 Offering:
See Note 13.
Interest Payable:
See Note 4 for interest payable on the 2020 Notes.
NOTE 12 – RESEARCH, CONSULTING AGREEMENTS AND COMMITMENTS
Research and consulting agreement
In November 2020, Quoin Inc. entered into a Master Service Agreement with Therapeutics Inc. for the management of the preclinical and clinical development of QRX003 for Netherton Syndrome. The initial term of the agreement was
In November 2021, the Company entered into a research agreement with Queensland University of Technology (QUT) for a pre-clinical research program for the development of a product to treat Netherton Syndrome of approximately $
On June 10, 2024, the Company signed a research agreement with The School of Pharmacy at University College Cork, Ireland (UCC). The scope of the agreement encompasses the development of novel topical formulations of rapamycin (sirolimus) as potential treatments for a number of rare and orphan diseases for which there are currently no approved therapies or cures. Under the terms of the agreement, based on the achievement of certain milestones, the Company will fund up to approximately €
F-16
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
on this research project commenced in December 2024 and the Company accrued $
Performance milestones and Royalties
See Note 9 for asset and in-licensed technology commitments.
NOTE 13 – SHAREHOLDERS’ EQUITY
As of December 31, 2024, the authorized share capital of the Company was
Each holder of an ordinary share is entitled to one vote per share held on all matters submitted to a vote of shareholders at each shareholders meeting. The board of directors shall determine and provide a record date for each shareholders meeting and all shareholders on such record date may vote. Unless stipulated differently in the Companies Law or in the articles of association, all shareholders’ resolutions shall be approved by a simple majority vote.
In November 2023 the company retired
Under Israeli law, the Company may declare and pay dividends only if, upon the determination of its board of directors, there is no reasonable concern that the distribution will prevent the Company from being able to meet the terms of its existing and foreseeable obligations as they become due. Under the Companies Law, the distribution amount is further limited to the greater of retained earnings or earnings generated over the
2023 Public Offering
On February 24, 2023 (the “2023 Closing Date”), the Company completed an offering (the “2023 Offering”) of
In connection with the 2023 Offering, the Company entered into an Amendment No. 1 to Warrant to Purchase Ordinary Shares Represented by American Depositary Shares, dated February 24, 2023 (collectively, the “2023 Warrant Amendments”), with each of the purchasers (the “2022 Purchasers”) who participated in both the Company’s August 2022 public offering (the “2022 Offering”) and the 2023 Offering. The 2023 Warrant Amendments amended certain terms of the ordinary warrants issued in the 2022 Offering to such 2022 Purchasers. Specifically, the 2023 Warrant Amendments reduced the exercise price of warrants to purchase
F-17
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
Alumni Equity Line and Purchase Agreement
On January 25, 2024, the Company entered into a purchase agreement (the “Alumni Purchase Agreement”) with Alumni Capital LP (“Alumni”). Pursuant to the Alumni Purchase Agreement, the Company has the right to sell to Alumni up to $
The Company does not have the right to commence any sales of ordinary shares represented by ADSs to Alumni under the Alumni Purchase Agreement until the date, which the Company refers to as the Commencement Date, that all of the conditions set forth in the Alumni Purchase Agreement have been satisfied, including that the registration statement the Company agreed to file with the SEC pursuant to the Alumni Purchase Agreement is declared effective by the SEC, and the Company’s shareholders have approved of the issuance of ADSs under the Alumni Purchase Agreement, which approval was obtained on April 5, 2024.
From and after the Commencement Date, the Company may, from time to time and at the Company’s sole discretion for a period of three months, which the Company at its sole discretion may increase by an additional three months (such period, including any extension, the “Commitment Period”), on any business day that the Company may select, direct Alumni to purchase ordinary shares represented by ADSs. The purchase price for the ordinary shares represented by ADSs the Company may sell to Alumni will be based upon formulas set forth in the Alumni Purchase Agreement based on the then current market price of the ADSs as computed under the Alumni Purchase Agreement and will depend on the type of purchase notice the Company submits to Alumni from time to time. There is no upper limit on the price per share that Alumni could be obligated to pay for the ADSs under the Alumni Purchase Agreement; provided, however at no time can the purchase price be below a floor price of $
As consideration for Alumni’s irrevocable commitment to purchase ADSs under the Alumni Purchase Agreement, the Company agreed to issue to Alumni, at the times set forth in the Alumni Purchase Agreement beginning with the trading day after the Commencement Date, a number of ADSs with a value at the time of issuance not to exceed $
In connection with each of the March 2024 Offering and the December 2024 Offering, the Company agreed not to sell any ADSs to Alumni under the Alumni Purchase Agreement for a period of days from the closing date of such Offering, see below. Per mutual agreement between the parties, the Company has not filed the required registration statement or sold any ADS to Alumni under the Alumni Purchase Agreement. The Company is restricted from selling ADSs under the Alumni Purchase Agreement for days from the closing of the December 2024 Offering (see below).
The Company expensed approximately $
2024 Public Offerings
On March 7, 2024, (the “March 2024 Closing Date”) the Company completed an offering (the “March 2024 Offering”) of the following securities (i)
F-18
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
and Series E Warrants have an exercise price of $
On March 7, 2024, the Company also entered into privately negotiated agreements with the holders of certain existing outstanding warrants to purchase up to
On December 23, 2024, the Company completed an offering (the “December 2024 Offering” and, together with the March 2024 Offering, the “2024 Offerings”) of the following securities (i)
In connection with the 2024 Offerings, the Company entered into Securities Purchase Agreements (the “2024 Purchase Agreements”) dated March 7, 2024 and December 23, 2024, respectively, with certain institutional investors signatory thereto, pursuant to which the Company agreed to issue and sell to such investors, certain of the ADSs, pre-funded warrants and ordinary warrants sold in the 2024 Offerings. Pursuant to the terms of each of the 2024 Purchase Agreements, the Company agreed, subject to certain exceptions, (i) to not enter into variable rate financings for a period of
Certain of the Company’s officers and directors purchased an aggregate of
On December 20, 2024, the Company also entered into privately negotiated agreements with the holders of certain existing outstanding warrants to purchase up to
F-19
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
Warrants
The following table summarizes warrant activities during the year ended December 31, 2023 and the year ended December 31, 2024:
Weighted | ||||||
Underlying | Average | |||||
| Warrants |
| Exercise Price | |||
Outstanding at December 31, 2022 | | $ | | * | ||
Granted Common Warrants | | | ||||
Granted Pre-Funded Warrants | | — | ||||
Exercised Pre-Funded Warrants | ( | — | ||||
Outstanding and exercisable at December 31, 2023 | | $ | | ** | ||
Granted Common Warrants | | | ** | |||
Granted Pre-Funded Warrants | | — | ||||
Exercised Pre-Funded Warrants | ( | — | ||||
Terminated | ( | $ | | |||
Outstanding and exercisable at December 31, 2024 | | $ | | |||
*Note that the exercise price of certain Ordinary Warrants issued in the Company’s 2022 Offering were reduced from $
**Note that the exercise price of certain Ordinary Warrants issued in the Company’s 2022 Offering and 2023 Offering were reduced from $
As of December 31, 2024, outstanding Ordinary Warrants expire in 2026, 2027, 2028 and 2029 and have an intrinsic value of approximately $
NOTE 14 – INCOME TAXES
Significant components of the Company’s deferred tax assets and liabilities at December 31, 2024 and December 31, 2023 are as follows:
(table in thousands) |
| 2024 |
| 2023 | ||
Net operating losses | $ | | $ | | ||
Accrued Expenses and Other | | | ||||
R&D Credit Carryforward | | | ||||
Stock Compensation | | | ||||
R&D Capitalization | | | ||||
Intangibles | | | ||||
Total gross deferred tax assets/(liabilities) | $ | | $ | | ||
Less valuation allowance | ( | ( | ||||
Net deferred tax assets/(liabilities) | $ | — | $ | — | ||
F-20
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
The income tax benefit for the years ended December 31, 2024 and December 31, 2023 differed from the amounts computed by applying the U.S. federal income tax rate of
(table in thousands) | 2024 | 2023 | ||||
Federal Statutory Rate |
| $ | ( |
| $ | ( |
Permanent Differences |
| |
| | ||
Research and Development |
| ( |
| ( | ||
State Income Tax |
| ( |
| | ||
State rate change | ( | — | ||||
Change in Valuation Allowance |
| |
| | ||
Deferred True Up |
| |
| ( | ||
Effective Tax | $ | — | $ | — | ||
A valuation allowance is required to reduce the deferred tax assets reported if, based on the weight of the evidence, it is more likely than not that some portion or all of the deferred tax assets will not be realized. After consideration of the available evidence, both positive and negative, the Company determined that valuation allowances of $
At December 31, 2024 and 2023, the Company had gross U.S. Federal income tax net operating loss (“NOL”) carryforward of approximately $
The Internal Revenue Code (the “IRC”) contains limitations on the use of net operating loss carryforwards after the occurrence of a substantial ownership change as defined by IRC Section 382. The Company has not performed a detailed analysis, however utilization of such net operating loss carryforwards will likely be significantly limited due to the shares issued in the Primary Financing and the Merger.
The income tax benefit for the years ended December 31, 2024 and 2023 differed from the amounts computed by applying the US federal income tax rate of
The Tax Cuts and Jobs Act of 2017 (TCJA) has modified the IRC 174 expenses related to research and development for the tax years beginning after December 31, 2021. Under the TCJA, the Company must now capitalize the expenditures related to research and development activities and amortize over five years for U.S. activities and 15 years for non-U.S. activities using a mid-year convention. Therefore, the capitalization of research and development costs in accordance with IRC 174 resulted in a gross deferred tax asset of $
F-21
QUOIN PHARMACEUTICALS LTD.
Notes to Consolidated Financial Statements
December 31, 2024 and 2023
NOTE 15 - CONTINGENCIES
From time to time, the Company may become involved in various legal matters arising in the ordinary course of business. Management is unaware of any matters requiring accrual for related losses in the financial statements.
NOTE 16 – LICENSE AGREEMENTS
As of both December 31, 2024 and December 31, 2023, the Company had
NOTE 17 - SUBSEQUENT EVENTS
Exercise of Warrants: In January and February 2025, certain investors in the December 2024 Offering exercised (i) the remaining outstanding
F-22
Exhibit 3.1
PUBLIC COMPANY
COMPANIES LAW, 5759–1999
A COMPANY LIMITED IN SHARES
AMENDED AND RESTATED
ARTICLES OF ASSOCIATION
OF
QUOIN PHARMACEUTICALS LTD
(PUBLIC CORPORATION 52-003648-4)
Index
Part One: Preamble | 1 | |
|
|
|
1. | Name of the Company | 1 |
|
|
|
2. | Objectives of the Company | 1 |
|
|
|
3. | Liability of the shareholders | 1 |
|
|
|
4. | The capital | 1 |
|
|
|
Part Two: General Provisions | 1 | |
|
|
|
5. | Definitions and interpretation | 1 |
|
|
|
6. | Change of Articles | 3 |
|
|
|
Part Three: Capital of the Company | 3 | |
|
|
|
7. | Ordinary shares | 3 |
|
|
|
8. | Redeemable securities | 4 |
|
|
|
9. | Capital of the Company, increase of capital and its cancellation | 4 |
|
|
|
10. | Issuance of securities | 5 |
|
|
|
Part Four: Shareholders | 5 | |
|
|
|
11. | Shareholder and share certificates | 5 |
|
|
|
12. | Calls for payment | 6 |
|
|
|
13. | Forfeiture | 7 |
| | |
Part Five: Transfer of shares in the register of shareholders | 9 | |
|
|
|
14. | Transfer of shares | 9 |
|
|
|
15. | Share transfer deed | 9 |
|
|
|
16. | Assignment of shares by law | 10 |
|
|
|
17. | Registration of transfer of shares | 10 |
|
|
|
Part Six: General Meetings | 11 | |
|
|
|
18. | Annual General Meetings | 11 |
|
|
|
19. | Special General Meetings | 11 |
|
|
|
20. | Agenda | 12 |
|
|
|
21. | Notice of a meeting | 12 |
|
|
|
22. | Quorum | 13 |
|
|
|
23. | Chairman of the general meeting | 14 |
|
|
|
24. | Voting in the general meeting | 14 |
|
|
|
25. | Vote count or secret ballot | 14 |
|
|
|
26. | Vote by proxy; vote of a corporation; partners | 15 |
|
|
|
27. | Voting instrument | 17 |
|
|
|
28. | Minutes | 17 |
|
|
|
29. | Meetings of a class | 17 |
|
|
|
29A. | Saving Provisions | 17 |
|
|
|
Part Seven: The Board of Directors | 17 | |
|
|
|
30. | Members of the Board | 17 |
|
|
|
31. | Restrictions on the appointment of Directors | 18 |
|
|
|
32. | External Directors | 19 |
|
|
|
33. | Revoked. | 19 |
|
|
|
34. | Alternate director | 19 |
|
|
|
35. | Dismissal of a Director | 20 |
| | |
36. | Authorities of the Board of Directors | 21 |
|
|
|
37. | Assumption of authorities of the Board | 23 |
|
|
|
38. | The rights of a Director | 23 |
|
|
|
39. | Chairman of the Board | 23 |
| | |
40. | Convening a meeting of the Board | 24 |
|
|
|
41. | Agenda | 25 |
|
|
|
42. | Notice of a meeting of the Board of Directors | 25 |
|
|
|
43. | Quorum | 25 |
|
|
|
44. | Voting on the Board | 26 |
|
|
|
45. | Minutes of a meeting of the Board | 26 |
|
|
|
46. | Defects in the convening of a meeting | 26 |
|
|
|
47. | Committees of the Board | 27 |
|
|
|
Part Eight: Audit Committee | 27 | |
|
|
|
48. | Appointment of an audit committee | 27 |
|
|
|
49. | Audit committee | 28 |
|
|
|
Part Nine: Exemption, indemnification and liability insurance | 28 | |
|
|
|
50. | Exemption and indemnification | 28 |
|
|
|
51. | Liability insurance | 30 |
|
|
|
Part Ten: General Manager (Chief Executive Officer) | 31 | |
|
|
|
52. | The Chief Executive Officer | 31 |
|
|
|
53. | Delegation and qualification of the CEO’s authorities | 31 |
|
|
|
Part Eleven: Management of the Company | 31 | |
|
|
|
54. | Registered office | 31 |
|
|
|
55. | Register of shareholders and register of material shareholders | 32 |
|
|
|
56. | Auditor | 32 |
|
|
|
57. | Expiration of the term of the Auditor | 32 |
|
|
|
58. | Remuneration of the Auditor | 33 |
|
|
|
59. | Authorities, duties and responsibility of the Auditor | 33 |
|
|
|
60. | Internal auditor | 34 |
|
|
|
Part Twelve: Financial statements and signature | 34 | |
|
|
|
61. | Financial statements | 34 |
|
|
|
62. | Stamp and signatory right | 34 |
|
|
|
Part Thirteen: Dividends and Bonus Shares | 34 | |
|
|
|
63. | Dividends and bonus shares | 34 |
| | |
Part Fourteen: Notices and Dissolution | 36 | |
|
|
|
64. | Notices | 36 |
|
|
|
65. | Dissolution | 37 |
| | |
Part One: Preamble
1. Name of the Company
In Hebrew: קויין פארמסוטיקל בע"מ
In English: Quoin Pharmaceuticals Ltd.
2. Objectives of the Company
(a)To engage in any lawful business.
(b) | The Company may donate from time-to-time reasonable sums for appropriate causes, even if the donations are not within the framework of the business considerations of the Company. |
3. Liability of the shareholders
The liability of a shareholder for the debts of the Company is limited to the payment of the unpaid portion which he undertook to pay for the share held by him in accordance with the terms of issuance of said share.
4. Registered Share Capital
The registered share capital of the Company is 100,000,000 (one hundred million) ordinary shares without any nominal value each (hereinafter: “Ordinary Shares”).
Part Two: General Provisions
5. Definitions and interpretation
(a) | Definitions |
“Applicable Law”: the Companies Law, the Securities Law, the Companies Ordinance or any other law of the State of Israel (and with regards to each of the above, including any rules or regulations promulgated thereunder), as well as any rules of a foreign stock exchange to the extent any of the foregoing apply to the Company, as such laws, rules and regulations may be amended, replaced or re-enacted from time to time, but excluding any of the foregoing which would derogate from a mandatory provision of Israeli law.
“the Articles” or “these Articles”: these Articles of Association of the Company, as they may be amended from time to time.
“Audit Activity”: has the meaning given to that term under Section 154 of the Companies Law.
“Auditor”: an accountant or accounting firm appointed for the audit of the Company's financial statements and other Audit Activity.
“the Board of Directors” or “the Board” means the Board of Directors of the Company. “Chief Executive Officer" or “CEO”: the Company's chief executive
1
officer holding the office of General Manager as described in the Companies Law, and/or a person so appointed under Section 119(a) of the Companies Law.
“Companies Law”: the Companies Law, 5759-1999, as it may be amended from time to time, including any regulations that are or may be promulgated thereunder.
“Companies Ordinance”: the Companies Ordinance [New Version], 5743-1983, as it may be amended from time to time, including any regulations that are or may be promulgated thereunder.
“the Company”: Quoin Pharmaceuticals Ltd.
“General Meeting” or “Meeting”: a general meeting of shareholders, including any adjournment thereof.
“the Office”: The registered office of the Company at any given time.
“Securities Law”: the Securities Law, 5728-1968, as it may be amended from time to time, including any regulations that are or may be promulgated thereunder.
(b) | Interpretation |
(1) | “Director”, “External Director”, “Internal Auditor” and “Officer” [“נושא משרה”] have the respective meanings given to those terms in Section 1 of the Companies Law unless and to the extent specifically defined otherwise herein and “Control” has the meaning given to that term in the Securities Law. |
(2) | Any capitalized term in these Articles shall have the meaning ascribed to that term by the Companies Law, unless otherwise defined herein or unless the context dictates otherwise. |
(3) | Reference to the “register of shareholders” means the register of shareholders of the Company kept pursuant to Sections 130-134 of the Companies Law, as further described in Article 55 of these Articles. |
(4) | References made in the singular shall include the plural and vice versa. Reference made in the masculine gender shall include the feminine (and vice versa) or no gender, and words that connote persons shall include also corporations, unless the context dictates another interpretation. The term “including” or any derivative thereof means without limiting the generality of the foregoing. |
(5) | Reference to a “person” (whether or not that term is capitalized) includes reference to any natural person, entity or body corporate, trust or syndicate, including a partnership, corporation, limited liability company, association, unincorporated organization, government or agency or political subdivision thereof; all of the above unless the context indicates otherwise. |
(6) | Unless and to the degree the context clearly indicates otherwise, reference in these Articles to any meeting shall include a meeting convened and held in any manner permitted by these Articles, including a General Meeting held in accordance with Article 19(g), all persons shall be deemed to be present at that meeting for all purposes of the Companies Law and these Articles, and words such as attend, presence, participate, attending, participating, attendance and participation shall be construed accordingly. |
(7) | Reference to “writing” or “written” shall mean any method of reproducing words in a legible and non-transitory form whether handwritten, printed, typewritten, photocopied, or other written manner, including by fax, email, or any other method of duplication or transmission by electronic means; and for |
2
any signature, including by means of electronic signature or the electronic transmission of a signature.
(8) | Reference in these Articles to a resolution (including a shareholders’ resolution), decision, approval, consent or other act by an organ of the Company means a resolution, approval, consent or act adopted by that organ in accordance with these Articles and the Companies Law. |
(9) | The headings of sections in these Articles are for the purpose of convenience only and shall not be used as an accessory to interpret or for the interpretation of these Articles. |
(10) | The specific provisions of these Articles shall supersede the provisions of the Companies Law and the Companies Ordinance, to the extent permitted under the Companies Law and the Companies Ordinance, as applicable. Wherever these Articles provide that the provisions hereof are subject to the provisions of any Applicable Law, such reference means (unless and to the extent the context requires otherwise) subject to those provisions of the Companies Law and/or Applicable Law applicable to the Company which cannot be derogated from. In the case of a contradiction between a mandatory provision of such law and any provision of these Articles - the provisions of the law shall prevail if and to the extent they may not be derogated from, without impairing the validity of any remaining provision of these Articles. |
(11) | These Articles have the legal force of a contract between the Company and its shareholders, and between the shareholders and themselves. |
6. Change of Articles
Subject to the provisions of the Companies Law the Company may amend these Articles by a resolution adopted by the General Meeting of the Company.
Part Three: Capital of the Company
7. Ordinary shares
(a) | All the ordinary shares shall have equal rights among them and each ordinary share shall confer upon its holder the following rights: |
(1) | The right to receive invitations or notices about all general meetings of the Company, to participate in the meetings and to vote in them on any matter that is raised in the meeting, where each ordinary share confers on its holder one vote on every vote on a resolution; |
(2) | The right to participate in any distribution that the Company makes to its shareholders, and to receive dividends and/or bonus shares, if they are distributed in accordance with the provisions of these Articles and the provisions of the Companies Law, proportionate to the number of the shares allocated and the rate that they are paid up by the shareholders, if they are not are not fully paid up; and |
(3) | The right to participate in the dissolution of the Company, in the distribution of the assets of the Company, that remain to be distributed, after the Company meets all of its obligations and payment of all its debts in any case, proportionate to the number of the shares allocated and the rate that such shares are paid up by the shareholders, if they are not fully paid up, and subject to the provisions of these Articles and without prejudicing existing rights of all the shareholders in the Company of any kind or class. |
3
(b) | The Company is entitled to pay a commission (including underwriting fees) to any person, in consideration for underwriting services, or the marketing or distribution of securities of the Company, whether reserved or unreserved, as determined by the Board. Payments, as stated in this Article 7(b), may be made in cash, shares or other securities of the Company, or any combination thereof. |
8. Redeemable securities
The Company is entitled, taking into account the provisions of the Companies Law, to issue redeemable shares and to redeem them. At the time of the redemption of the shares the Company will act in accordance with the provisions of the Companies Law.
9. Capital of the Company, increase of capital and its cancellation
(a) | The Company may have shares, bonds, or other securities, each with different rights. |
(b) | The Company will not issue bearer shares or stock that state that their holder is a holder of bearer stock. |
(c) | The Company is entitled from time to time by a shareholders’ resolution adopted in a General Meeting: |
(1) | To increase the registered share capital of the Company by classes of shares, as determined; |
(2) | To cancel registered share capital that has not yet been allocated, provided that there is no commitment by the Company, including a conditional commitment, to allocate the shares; |
(3) | To consolidate and redistribute its share capital into shares of a nominal value; and |
(4) | To convert, from time to time, part of the allocated shares into shares with other rights. |
(d) | Unless established otherwise in a resolution approving the change of share capital, the new shares shall be subject to the provisions of these Articles regarding calls for payment, forfeiture, transfer, delivery etc., applicable to the shares of the original share capital. |
(e) | Without derogating from the generality of the authority of the Board of Directors, with respect to any consolidation of shares and any other action which may result in fractional shares, the Board of Directors may settle any difficulty which may arise with regard thereto, as it deems fit, including, inter alia, by resorting to one or more of the following actions: |
(1) | allot, in contemplation of or subsequent to such consolidation or other action, such shares or fractional shares sufficient to preclude or remove fractional share holdings; |
(2) | to the extent as may be permitted under the Companies Law, redeem or purchase such shares or fractional shares sufficient to preclude or remove fractional shareholdings; and |
(3) | to the extent as may be permitted under the Companies Law, cause the transfer of fractional shares by certain shareholders of the Company to other shareholders thereof so as to most expediently preclude or remove any fractional shareholdings, and cause the transferees to pay the transferors the fair value of fractional shares so transferred, and the Board of Directors is hereby authorized to act as agent for the transferors and transferees with |
4
power of substitution for purposes of implementing the provisions of this sub-Article.
10. Issuance of securities
(a) | The Board may issue or allocate shares or other securities (including options, bonds, warrants or other instruments), that are convertible into or may be exercised for shares, until a limit of the registered share capital of the Company, under such the terms and condition as the Board may determine; for this purpose convertible securities or securities which may be exercised for shares shall be deemed as if they were converted or exercised on the date of their issuance. |
(b) | The authority of the Board as set forth in Article 10(a) may be delegated as enumerated in Articles 10(b)(1) or 10(b)(2) herein: |
(1) | To a committee of the Board – regarding an issuance or allocation of securities as part of a workers compensation plan or employment or remuneration agreements or wage agreements between the Company and its employees, or between the Company and the employees of an Affiliated Company to which its board agreed in advance, provided that the issuance or allocation is according to a plan that includes detailed criteria that has been approved by the Board; |
(2) | To a committee of the Board, to the CEO or a deputy acting in place of the CEO, or to another person who is recommended by the CEO or a deputy acting in his place - regarding an issuance or allocation of shares following an exercise or conversion of securities of the Company. |
(c) | The Board of Directors may decide to issue a series of bonds as part of its authority to borrow on behalf of the Company, within the limits set by said authority. |
(d) | The provision of Article 10(c) above does not negate or limit the authority of the CEO, a Board Committee or other person so authorized, to borrow on behalf of the Company, to issue individual bonds, promissory notes and bills of exchange, within the limits set by said authority. |
(e) | The Company shall not allocate a share the consideration of which, in full or in part, is not paid in cash, unless the consideration for the share is specified in a written document. |
(f) | If the Company decides to allocate shares with a nominal value insofar as there will be shares with a nominal value as part of the capital of the Company, for a lower amount than the nominal value, including bonus shares, it must change part of its profits into share capital (under such meaning in Section 302(b) of the Companies Law), from a premium on shares, or from any other source included in its equity capital, that are listed in its last financial statements, for a sum equal to the differential between the nominal value and such lower amount. |
Part Four: Shareholders
11. Shareholder and share certificates
(a) | A shareholder of the Company is any one of the following: |
(1) | A person for whose benefit a share is registered with a member of a stock exchange and said share is included among the shares registered in the register of shareholders in the name of the relevant nominee Company; and/or |
(2) | A person who is registered as a shareholder in the register of shareholders. |
5
(b) | Other than as stated in Article 11(a) above, a person or legal entity shall not be recognized by the Company as having any right to a share, and the Company shall not be bound or recognize any benefit in equity or trust relationships or chose in action, planned or partial, but only the right of a shareholder, to a complete share, and all of the above unless a competent court of the law orders otherwise. |
(c) | If two or more holders are registered as joint owners of a share: |
(1) | In respect of a vote, giving proxies, and notices, the shareholder who is registered first in the register of shareholders shall be considered as the sole shareholder, unless all the holders of the joint share give written notice to the Company that another person should be referred as sole shareholder. |
(2) | Each of the holders may give a valid receipt in respect of all the joint holders for each dividend, other money or property that is received from the Company for the share or in respect thereto, and the Company is entitled to pay a dividend, the other money or the property for the share to one or more of the joint holders of the share, as it chooses to do. |
(d) | Subject to the provisions of Applicable Law, a shareholder who is a trustee shall be registered in the register of shareholders, as a shareholder, with a statement concerning his trusteeship status. Notwithstanding the foregoing, the Company will recognize the trustee as a shareholder, for all intents and purposes, and will not be required or obligated to recognize any other person, including the beneficiary, as holding any title in or right to a share. |
(e) | A shareholder registered in the register of shareholders is entitled to receive from the Company one share certificate testifying to his ownership of the share. |
A shareholder registered in the register of shareholders, shall be entitled to receive one share certificate for the shares registered in his name and fully paid up, or, if the Board approves (after payment of the amount that the Board establishes from time to time), a number of share certificates, for one or more of the shares. Each share certificate shall state the number of the shares for which it is issued.
(f) | Share certificates shall be issued with the stamp of the Company and with the signatures of two Directors of the Company or in any other manner determined by the Board of Directors. |
(g) | A share certificate in the name of two or more persons in the name of two or more persons, shall be delivered to the person whose name appears first in the shareholder register among the names of the joint holders. |
(h) | A new share certificate may be issued in place of a share certificate that was destroyed, lost or ruined, for the payment and under the terms regarding evidence, indemnification, guarantee against damages and/or issuance of an affidavit, as determined by the Board of Directors in its sole discretion from time to time. |
12. Calls for payment
(a) | A shareholder shall not be entitled to a dividend or participate in the allocation of bonus shares or exercise any right of a shareholder in the Company, unless he has paid up all the sums and calls for payment that he owes the Company until said time in respect of his shares in the Company. |
(b) | The Board of Directors may, from time to time, in its discretion, make calls for payment on shareholders for any sums that have not been paid up in respect of shares held by each of the shareholders, and for which pursuant to the terms of the allocation of the shares are not payable at a fixed time, and each shareholder shall pay the amount |
6
of the call made upon him, at the time and place designated by the Board of Directors. The Board of Directors may instruct that a call for payment be made in installments.
(c) | Notice of a call for payment shall be given and shall specify the amount of payment (no less than 14 days from the date of the notice) and the place for payment provided that prior to the time of payment for the call for payment, the Board of Directors may, by written notice to the shareholders, cancel the call or extend the time for payment or payment for any part thereof. |
(d) | Joint holders of a share shall be jointly and severally liable to pay all amounts and calls for payment in respect of such share held jointly. Without derogating from the aforesaid generality, a call for payment delivered to one of the holders shall be deemed as having been delivered to all the owners. |
(e) | If pursuant to the terms of the issuance of a share or otherwise, an amount is made payable at a fixed time or in installments at fixed times, whether on account of the share capital or by way of premium, such amount or installment shall be payable at such time as if it were payable by virtue of a call duly made by the Board of Directors for which notice was duly given, and all the provisions of these Articles in respect of calls for payment shall be applicable to such amount or installment. |
(f) | If a call for payment or an installment is not paid on the due date or prior to such time, then the person who at such time is the holder of the share for which the call for payment was made, or for which the installment is due, shall pay interest on such sum at the maximum amount set at such time by Bank Leumi :Le-Israel B.M. for unauthorized overdrafts, or at a lower rate that the Board will determine from time to time, from the date designated for its payment until the actual payment thereof, however the Board may waive the payment of interest, in whole or in part. |
The provisions of this Article do not detract or impair from the remedies and relief available to the Company by these Articles or by any Applicable Law or agreement.
(g) | The Board of Directors may decide to accept money from a shareholder who wishes to advance payments, in whole or in part, on account of shares which have not been fully paid up and with respect to which the time for their payment has not yet matured, and to pay interest on such sums for a period not to exceed the period between the date of payment and the date on which this sum was designated to be paid, at the rate agreed by the Board of Directors and the shareholder. |
13. Forfeiture
(a) | A shareholder who has not fully paid up a sum for which a call has been made by the designated date, may be furnished with a written notice by the Board of Directors demanding that he pay the unpaid sum with interest and any expenses which the Company incurs due to the default in payment on the designated date for payment. |
(b) | The notice shall specify another date for payment, which shall not be earlier than seven days after the notice, and it shall state that if the amount is not paid up by this date the share for which such notice is given may be forfeited. |
(c) | If the demands in the notice are not satisfied, the Board of Directors may, so long as the sum is not paid up, including the interest and expenses, decide to forfeit the share. The forfeiture shall also apply to any dividends announced with respect to the forfeited shares (insofar as they are eligible for dividends) which were not actually paid out prior to the forfeiture. |
(d) | A share that has been forfeited shall be deemed the property of the Company, and the Board of Directors may, taking into account the provisions of these Articles, sell or transfer it or reallocate it in another manner, under such terms and manner as it decides. A share so forfeited so long as it has not been sold, transferred or allocated again as |
7
stated, shall become a dormant share under such meaning in Section 308 of the Companies Law which shall not confer any rights at all so long as it is owned by the Company.
(e) | Insofar as nothing has been done with the forfeited share, the Board of Directors may cancel the forfeiture under the terms that it establishes. |
(f) | A shareholder whose shares have been forfeited: |
(1) | Shall cease being a shareholder in respect of the shares that were forfeited and upon the forfeiture all of his rights and obligations for the forfeited shares shall be revoked and any action and/or demand against the Company regarding the forfeited shares shall be cancelled, other than those rights and obligations which are excepted from this rule by these Articles and/or which are imposed on the former shareholder by law; however |
(2) | He shall continue to be obligated to pay the Company and will pay the Company, without delay, all the calls for payment, payment installments, interest and expenses owed on account of the forfeited shares or for them at the time of the forfeiture, together with interest on those sums from the date of the forfeiture until the date of actual payment, at the maximum rate permitted at that time by law, provided that if the shares that were forfeited are sold, transferred or reissued, the shareholder’s debt will be reduced by the sum actually received by the Company (after the expenses of the sale), from their sale, transfer or reissuance, as warranted. |
(g) | The provisions in these Articles regarding forfeiture shall apply to the default of payment of any sum that is to be paid on a designated date according to the terms of issuance of the share, whether on account of the share or in the form of a premium, as if it were a sum that was meant to be defrayed by virtue of a call for payment and a duly delivered notice. |
(h) | In the case of a sale after forfeiture, the Board of Directors may appoint a person to sign a transfer instrument of the share that was sold and to arrange (subject to the provisions of Applicable Law) so that the buyer will be registered in the register of shareholders as the owner of the shares that were sold or which will be received by him in any other manner. The recipient of the share that was sold, transferred, allocated or sent shall not be responsible for how the consideration for the sale is used, if received, his right to the share shall not be harmed due to a defect or a disqualification in the forfeiture, sale, allocation or transfer process, and after he is registered in the register (subject to the provisions of Applicable Law) or he receives the share into his possession in any other manner, no such claim shall be raised, and the validity of the sale or the transfer shall not be appealed. |
(i) | An affidavit duly made by a Director of the Company that a certain share of the Company has been duly forfeited on the date specified in the affidavit shall serve as conclusive proof of its content against any person who asserts a claim to the share. The affidavit with a Company receipt for the consideration, if given, for the share, in its sale or transfer, shall confer a right to the share on the transferee. |
(j) | The net proceeds of any sale following a forfeiture after the discharge of the sale expenses, shall be applied in discharging the debts and the fulfillment of the obligations of such shareholder (including the debts, obligations and agreements for which the date of discharge or maturity have not yet come due), and the balance (if any) shall be paid to him or to whoever is conferred a right to the shares following the death, bankruptcy or dissolution of the shareholder. |
(k) | The provisions of this Article shall not be construed as derogating from any other relief available to the Company against the debtor shareholder. |
8
Part Five: Transfer of shares in the register of shareholders
14. Transfer of shares
(a) | Subject to the provisions of Applicable Law, the Board of Directors may stop the registration in the register of transfers of shares for a specific period of time, that will not exceed 30 days per year, provided that it will not do so during the 14 days prior to the determining date for ownership of a share to establish eligibility to the rights for the share (such as the determining date for eligibility to vote in a general meeting or to receive a dividend or other distribution from the Company). |
(b) | Part of a share may not be transferred, but a share which is jointly held by a number of owners, each may transfer their right to the share. |
(c) | In the case of a transfer of shares, the transferee shareholder shall have all the rights that were attached to the transferred shares and all the obligations related to them according to these Articles, unless otherwise agreed in writing, between the transferor shareholder and the transferee shareholder. |
(d) | For the avoidance of doubt, shares registered in the name of a depository trust company or its nominee shall be transferrable in accordance with the policies and procedures of that depository trust company. |
15. Share transfer deed
(a) | The Company shall not be required to register a transfer of shares in the register of shareholders unless a transfer instrument is delivered to the Office in accordance with these Articles. A share transfer deed in the Company shall be signed by the transferor and the transferee, and the transferor will be deemed the owner of the share until the name of the transferee is registered in the register of shareholders in respect of the transferred share. |
(b) | The instrument of transfer of a share shall be in the following form or as near thereto as possible, in any usual or common form, or any other form reasonably satisfactory to the Board of Directors and/or the Company’s transfer agent: |
Share Transfer Deed
I, _________________ of ___________________ (hereinafter: “the Transferor”) do hereby transfer to _______________ of ___________________ (hereinafter: “the Transferee”), in consideration of the amount of [NIS][U.S.$] ________ (in words ______________________ [new shekels][United States dollars]) paid to me ________ shares of Quoin Pharmaceuticals Ltd. (Company no. 52-003648-4), and they shall belong to the transferee, the administrators of his estate and representatives, subject to the terms by which I/we held the same immediately before the execution of this deed; and I/we, the transferee(s), do hereby agree to accept the shares subject to these terms.
In witness whereof we set our hand this _____ day of the month of __________ in the year _________.
Signature of transferor __________ Signature of transferee __________
Witness to signature __________ Witness to signature __________
(c) | The Board may approve other methods of recognizing the transfer of shares, taking into account the manner of trading of the Company's shares. |
(d) | A transfer deed shall be submitted to the Office for registration, along with the share certificates that are being transferred (if there are certificates) and/or any other evidence required by the Board, in its discretion, regarding the proprietary right of the |
9
transferor or in respect of his right to transfer the shares. Transfer deeds that are registered shall remain with the Company but any transfer deed with respect to which the Board refuses to cause shares in question to be entered in the register of shareholders in the name of the transferee, shall be returned upon request, to the person who so delivered them, together with the share certificate (if delivered).
16. Assignment of shares by law
(a) | The Board of Directors may, at any time and subject to the provisions of relevant law, register as a shareholder a person who is entitled to a share by law, including an heir, trustee, executor of an estate, receiver, liquidator or trustee in bankruptcy following any event of death, bankruptcy, insolvency, liquidation or dissolution, including without limitation a "Trustee" as that term is defined in the Insolvency and Economic Rehabilitation Law, 5778-2018, after the Company is presented with a probate order, a succession order or any other sufficient evidence, as required or designated by any relevant law for such purpose or as the Board deems fit, demonstrating the right to the shares to the satisfaction of the Board or a person appointed or designated by the Board for such purpose. An eligible person who is so registered as a shareholder in the Company, is entitled, subject to the provisions of these Articles dealing with the transfer of shares and the provisions of relevant law, to transfer these shares to another person. Without derogating from the above, the Board may refuse to perform such registration or may delay it in a manner it would have been entitled to do, had the last registered owner himself transferred the share, prior to the transference of such right. |
(b) | Subject to the provisions of the Companies Law and these Articles: |
(1) | The executors of an estate of a shareholder who died, or in the absence of an executor of estate or administrator of an estate, persons who have a right by virtue of being heirs of the shareholder who died, shall be the only ones to be recognized by the Company as right holders to the share. A share registered in the name of two or more persons and one died, the Company shall recognize only the shareholders who are alive as the persons with rights to the share or benefits to it. Nonetheless the aforesaid shall not be construed as releasing the estate of the joint shareholder who died from all the obligations with respect to the shares. |
(2) | A person who is entitled to a share by law but has yet to be registered in the register of shareholders is not entitled: (1) to receive dividends, distributions or any other money and/or property paid for said share as if he was the registered owner of the share; and (2) by virtue of said share to benefit from all rights of a shareholder regarding notices about general meetings, to be present at them or to vote in them, or class meetings, as the case warrants, of the Company or to make use of any other right of shareholders. |
17. Registration of transfer of shares
(a) | Subject to the provisions of Applicable Law, the Company shall change the registration of ownership in the register of shareholders if each of the following is present: |
(1) | The Company is delivered a transfer deed of the share with the signatures of the transferor and the transferee as stated in Article 15 above, and the requirements of these Articles are satisfied; |
(2) | The Company is delivered a court order to amend the register; |
(3) | It is proven to the Company that the conditions in the law to assign the right have been satisfied; or |
(4) | Another condition is satisfied which according to these Articles is sufficient to that the change can be registered in the register of shareholders. |
10
(b) | The transferor of the shares shall be considered the shareholder until the registration of the share transfer in the register of shareholders in the name of the transferee in respect of the transferred share. |
(c) | The Company will keep all the registration in the register of shareholders as stated in this Article 17. The Company may destroy share transfer instruments and share certificates that were cancelled after the expiration of 7 years from the date of registration of the revision in the register of shareholders, where there will be an absolute presumption that the destroyed documents as stated above were binding and valid and that the transfers, the revocations and the registrations, as warranted, were lawfully made. |
Part Six: General Meetings
18. Annual General Meetings
(a) | The Company shall convene a General Meeting of shareholders of the Company on an annual basis (the “Annual Meeting”), and no later than 15 months after the previous Annual Meeting. |
(b) | The agenda at the Annual Meeting shall include matters required by the Companies Law or other Applicable Law to be deliberated upon at an annual general meeting or such other matters as shall be determined by the Board of Directors (including any matter that is scheduled for the agenda as set forth in Article 20 herein). |
19. Special General Meetings
(a) | At any time, the Board of Directors may convene a General Meeting of the Company’s shareholders other than the Annual Meeting (a “Special General Meeting”) by means of a Board resolution. |
(b) | The Board of Directors shall convene a Special General Meeting upon the demand of each of the following: |
(1) | Two Directors, or a quarter of the Directors then serving; |
(2) | One or more shareholders of the Company holding at least the required percentage under the Companies Law. |
(c) | A Board of Directors that is required to convene a Special General Meeting shall convene such Meeting in accordance with the provisions of Section 63(c) of the Companies Law. |
(d) | If the Board of Directors fails to convene a Special General Meeting in accordance with Article 19(c) above, the person demanding such meeting, and if shareholders, those representing more than half of the voting rights of the demanding shareholders, may, subject to the provisions of Section 64(a) of the Companies Law, convene such Special General Meeting of their own accord ; subject to Section 64(b) of the Companies Law, reasonable expenses incurred in holding such Meeting will be borne by the Company. |
(e) | Subject to Applicable Law, no flaw or defect in the convening of a General Meeting or in the management thereof shall invalidate any resolution adopted by a General Meeting, nor render defective any discussion taking place therein. |
(f) | A General Meetings of the Company shall be convened at a location (whether outside or in Israel) established in the notice of the Meeting, including, subject to Applicable Law, satellite locations as described in Article 19(g). |
11
(g) | Notwithstanding the specification of a meeting place in a notice convening a General Meeting, subject to Applicable Law, the Board or the Chairman of a General Meeting may (but shall not be obliged to): (1) make arrangements so that any persons entitled to attend a General Meeting may do so by simultaneous attendance and participation at the principal place of the Meeting and/or any number of satellite meeting place(s) anywhere in the world that the Board may establish; all shareholders present in person, by proxy or by written ballot at such satellite meeting places shall be counted in the quorum for and entitled to vote at the General Meeting in question; and that Meeting shall be duly constituted and its proceedings valid; provided that the Chairman of the General Meeting is satisfied that adequate facilities are available throughout the General Meeting to ensure that shareholders attending at all meeting places are able to: (i) participate in the business for which the Meeting has been convened, (ii) hear all persons who speak (whether by the use of microphones, loudspeakers audio-visual communications equipment or otherwise) in the principal meeting place and any satellite meeting place(s), and (iii) be heard by all other persons so present in the same way; and/or (2) take such action, give such direction or put in place such arrangements as they consider appropriate under the circumstances, including any form of communication by electronic means, to accommodate the health, safety or security of the persons attending or entitled to attend such Meeting and promote the orderly conduct of the business of the Meeting, including with regard to any health, safety or security regulations or recommendations issued by any government regulator or advisory authority applicable to the place of the Meeting and/or any satellite locations thereof, including with regard to Covid-19 or any other current or potential pandemic, endemic, epidemic or other public health concern. Nothing in these Articles shall be interpreted to preclude the holding and conducting of a General Meeting in such manner that persons who are not physically present together at the same place or places participate by electronic means. For this purpose, “electronic means” includes, without limitation, website addresses and conference call systems, and any device, system, procedure, method or other facility whatsoever providing an electronic means of attendance at and/or participation in a General Meeting, or any other means as may be determined by the Board from time to time. |
(h) | Any decision of the Chairman of a General Meeting on matters of procedure or conduct of the Meeting, or matters arising incidentally from the business of a General Meeting, and any determination by the Chairman of a General Meeting as to whether a matter is of such a nature, shall, subject to Applicable Law, be final. |
20. Agenda
(a) | The agenda of a General Meeting shall be set by the Board of Directors, and shall include topics for which a Special General Meeting was demanded to be convened pursuant to Article 19, above as well as any subject that is required as set forth in Article 20(b) herein. |
(b) | The General Meeting shall adopt resolutions on subjects that are specified on the agenda only. Notwithstanding the above, it is understood that the General Meeting, may, inter alia, adopt resolutions related to other subjects that were not included on the original agenda of the General Meeting with respect to matters which: |
(1) | the Companies Law permits to be raised, even if they are not included on the original agenda of the general meeting; and - |
(2) | in light of the circumstances under which the General Meeting is convened, the Chairman of the General Meeting believes is proper and correct to be discussed; or |
(3) | is raised by a shareholder and approved by the Board of Directors in accordance with Section 66(b) of the Companies Law. |
12
21. Notice of a meeting
(a) | Prior notice of a General Meeting shall be given at such time and in such manner as prescribed by Applicable Law and shall include sufficient detail as required by Applicable Law. |
(b) | Unless and to the extent required by Applicable Law, the Company is not required to provide a notice or invitation to a General Meeting to any shareholder or class of shareholders of the Company, whether or not such shareholders appear in the register of shareholders. |
(c) | A shareholder who is interested in voting in a General Meeting shall prove to the Company his ownership of shares in accordance with the Companies Law. |
(d) | Shareholders who are eligible to participate and vote in a General Meeting are those who are holders of shares at the time of the record date, as determined by the Board of Directors in accordance with Article 21(e) below. |
(e) | Notwithstanding anything to the contrary, and to allow the Company to determine the shareholders entitled to: (i) receive notice of, participate in, or vote at, any General Meeting or any adjournment thereof; (ii) express consent to, or dissent from, any corporate action in writing without a meeting; (iii) receive payment of any dividend or other distribution or allotment of any rights; (iv) exercise any rights in respect of, or to take or be the subject to, any other action; or (v) any other matter which the Board may deem relevant for this purpose; the Board may fix a record date before the date of such meeting or other action, which date shall not be more than the maximum number of days permitted under the Companies Law, nor less than the minimum number of days required under the Companies Law. A determination of shareholders of record entitled to notice of, participate in or vote at a Meeting shall apply to any adjournment of the Meeting. |
(f) | A General Meeting with a quorum present may decide to adjourn the Meeting, or the discussion or adoption of a particular resolution on a topic that is on the agenda, to another time or place that it determines; at the adjourned meeting no subject shall be discussed other than a subject that was on the agenda and which was not previously resolved. |
If a General Meeting is adjourned for a period of over twenty-one (21) days, notices and invitations shall be delivered for the adjourned Meeting as set forth in this Article 21.
If a General Meeting is adjourned for a period that is less than twenty-one (21) days, the Company will give notice of the date and time of the adjourned Meeting as soon as possible, but no later than seventy two (72) hours prior to the time of the adjourned Meeting.
22. Quorum
(a) | Proceedings in the General Meeting shall not commence until a quorum is present at the start of the proceedings. |
(b) | The provisions of these Articles relating to General Meetings shall, mutatis mutandis, apply to any separate General Meeting of the holders of the shares of a particular class, it being clarified that the requisite quorum at any such separate General Meeting shall be two or more Shareholders (not in default in payment of any sum referred to in Article 13 hereof) present in person or by proxy and holding not less than thirty-three and one-third percent (331∕3%) of the issued shares of such class, provided, however, that if (i) such separate General Meeting of the holders of the particular class of Shares was initiated by and convened pursuant to a resolution adopted by the Board of Directors and (ii) at the time of such meeting the Company is qualified to use the forms of a “foreign private issuer” under US securities laws, then the requisite quorum at any |
13
such separate General Meeting shall be two or more Shareholders (not in default in payment of any sum referred to in Article 13 hereof) present in person or by proxy and holding not less than twenty-five percent (25%) of the issued shares of such class. For the purpose of determining the quorum present at such General Meeting, a proxy may be deemed to be two (2) or more Shareholders pursuant to the number of Shareholders represented by the proxy holder.
(c) | If a quorum is not present after one half hour from the time the General Meeting was designated to start, the Meeting shall be adjourned for one week, to the same day, same time and place or to a later date if specified in the invitation to the General Meeting or to another day and/or place as will be determined by the Board of Directors in a notice to shareholders who are eligible to vote. |
(d) | If a quorum is not present at the adjourned meeting as set forth in Article 22(c) above, after a half hour from the time designated for its start, the meeting shall take place with any number of participants, even if the General Meeting was convened at the demand of shareholders as set forth Article 19 above. |
(e) | Regarding any quorum or vote in a General Meeting, or regarding the person serving or designated to serve as Chairman of a General Meeting, words such as “presence”, “present” or “attendance” (including in the negative, such as “absence”) refer to the presence of such person himself, including by teleconference, electronic communications or other means established in accordance with Article 19(g) above, and further including through a voting instrument as set forth in Article 27 herein , or by proxy or representative as set forth in Article 26 herein. |
23. Chairman of the general meeting
(a) | The Chairman of the Board of Directors, or any other Officer appointed for such purpose, from time to time, by the Board, shall convene and serve as Chairman of each General Meeting. |
(b) | If the person designated in Article 23(a) is absent from the meeting within 15 minutes from the time designated for the meeting, or if he refuses to sit as Chairman of the General Meeting, an Officer of the Company then present shall convene and serve as Chairman of the Meeting; if no such person is willing to serve as Chairman, the General Meeting may elect one of the shareholders or shareholder proxies present, to serve as Chairman of the Meeting. |
(c) | The Chairman of the General Meeting shall conduct the General Meeting. |
24. Voting in the general meeting
(a) | Subject to the provisions of the Companies Law and unless established otherwise in these Articles, a shareholders’ resolution shall be considered adopted upon an affirmative vote by a regular majority of the votes of shareholders who are present at the meeting and voting on the resolution (for avoidance of doubt, without abstentions taken into account for the purposes of voting). |
A shareholder shall not be entitled to vote in the General Meeting prior to paying all of the sums and calls for payment owed from him at such time to the Company for his shares in the Company.
(b) | The Chairman of the General Meeting shall not have an additional or conclusive vote. |
(c) | A declaration by the Chairman of the General Meeting that a resolution was unanimously adopted, rejected, or adopted by a specific majority, shall constitute presumptive proof of its accuracy. |
14
25. Vote count or secret ballot
(a) | Any resolution put to a vote in a General Meeting shall be decided by counting votes, unless at least one shareholder who is present and who holds at least five percent (5%) of the voting rights in the Company, demands a secret ballot. |
(b) | If a demand is made for a secret ballot, the vote will take place in a manner, time and place that the Chairman of the General Meeting shall instruct, whether immediately or after a recess or adjournment or in another manner in the Chairman’s discretion, and the results of the secret ballot shall be considered a resolution of the General Meeting in which the secret ballot was demanded. Those demanding a secret ballot may cancel the demand at any time prior to the secret ballot. |
A secret ballot regarding the selection of a Chairman or adjournment of a General Meeting shall take place without delay.
(c) | A demand for a secret ballot shall not prevent the continuation of the General Meeting and discussion on any issue other than the one with respect to which the secret ballot was demanded. |
26. Vote by proxy; vote of a corporation; partners
(a) | A shareholder may vote personally or by proxy, through an instrument appointing the proxy as set forth below, or in the case of a corporation - by a representative through an instrument of appointment as set forth below. Likewise a shareholder may vote by a voting instrument, as set forth in Article 27 herein. A representative or proxy does not need to be a shareholder of the Company. |
(b) | A corporation that is a shareholder of the Company may, by a resolution of its board of directors, or by act of any person or managing body competent under the bylaws of that corporation, give an instrument of appointment to a representative and empower such person whom it finds suitable to be its representative at every meeting of the Company. |
A representative of the corporation as stated above shall be entitled to exercise on behalf of the corporation that he represents those powers that the corporation itself could have used if it were a shareholder of the Company who is not a corporation.
(c) | The instrument appointing a proxy shall be signed by the principal or his agent who is so authorized by a duly written instrument, and if the principal is a corporation - by the signature of the person authorized to issue an instrument of appointment for the corporation as set forth in Article 26(b) above or by the signature of an authorized signatory of the corporation. An instrument of appointment of a representative or proxy in effect for a non-specified period, shall expire following 12 months from the date of the last signature on it. |
(d) | The instrument to appoint a proxy or a copy certified by an attorney or certified in another manner to the satisfaction of the Company, and confirmation of the ownership of a share, shall be deposited in the office or in another location as the Board will establish from time to time in a general manner or for a specific case, no less than twenty-four (24) hours prior to the date designated for the Meeting or the adjourned Meeting for which the instrument of proxy is written, or on a date established by the Board in its discretion, provided that it is received by the Company prior to the time set for such General Meeting or adjourned Meeting . If it is not so deposited, the instrument shall not be valid for said General Meeting or an adjourned General Meeting. |
(e) | Any document appointing a proxy for a particular Meeting, or for a specific time frame, shall be written in the format below or in such other form as may be approved by the Board or a Director or Officer serving as the Chairman of the Meeting, including an instrument effected through the internet or other electronic medium, and including a |
15
form which provides for a continuing proxy until the occurrence of a date or event specified in that proxy:
I _______________ bearer of identity card no/company no./public company no. __________ of _______________________ a shareholder in Quoin Pharmaceuticals Ltd. (public company no. 52-003648-4) hereby appoint Mr./Ms. _____________________________________________ of ______________________________________ as my proxy (and in the case of a corporation - as my representative) to vote as my proxy in my name and on my behalf in a general (annual/special) meeting of the Company to be held on the ____ day of the month of __________ in the year __________ and in any adjourned meeting.
In witness whereof I hereby set my hand on this ____ day of the month of __________ in the year __________
Signature: __________
(f) | If the statement of appointment does not specify the number of shares for which it is given or it specifies a number of shares that is higher than the number of shares registered in the name of the shareholder (in the register or title certificate), the instrument of appointment shall be considered as if it were given for all of the shares registered in the name of the shareholder. If the instrument of appointment is given for a number of shares that is lower than the number of shares registered in the name of the shareholder, the shareholder shall be considered as abstaining from being present at the vote of the remainder of the shares which are registered in the name of the shareholder, and the instrument of appointment will be valid only for the number of the shares listed in it. |
(g) | A vote in accordance with an instrument of appointment shall be lawful even if the instrument has a defect that is not immediately apparent and/or if prior to said vote the principal died or became legally incompetent and/or the instrument of appointment was revoked or the power of attorney by which the instrument was signed was revoked and/or the share with respect to which the instrument was given was transferred, unless a written notice was received in the Office and/or by the chairman of the General Meeting prior to the meeting of the defect, the death, disqualification, revocation or transfer. |
(h) | Without derogating from the aforesaid, a shareholder holding more than one share shall be entitled to appoint more than one proxy or representative, subject to the following provisions: |
(1) | Each instrument of appointment will specify the class of the shares and the number of shares for which it is given. |
(2) | If the total number of shares of any class listed in the instrument of appointment is greater than the number of shares of said class registered in the name of said shareholder, the entire instrument of appointment will be null with respect to the shares of said class that was given by the shareholder. |
(3) | A shareholder or proxy or representative for the vote, may vote by virtue of some of the shares that are in his possession or for which he is serving as a proxy or representative, and he may vote by virtue of these shares in one manner and by virtue of some shares in another manner. |
(i) | In a vote by joint holders of a share an instrument of appointment to a proxy shall be signed by the person who is authorized to vote as set forth in Article 11(c)(1) above. |
(j) | A shareholder who is incompetent may vote through his lawful guardian or another person appointed by a court, and they may vote for him through proxies or instruments of appointment as stated in these Articles. |
16
27. Voting instrument
(a) | A shareholder may vote in the General Meeting and in meetings of a class of shares through a voting instrument in which the shareholder will specify the manner of his vote, on resolutions on topics that the law permits voting on them through a voting instrument, and for any other subject with the Board of Directors decides that a vote in the General Meeting on a specific subject may also be adopted by way of a voting instrument. |
(b) | A voting instrument in which a shareholder indicates the manner of his vote and which he completes as required, which reaches the Company by the final time established for such in the invitation to the General Meeting, shall be considered as a presence in the General Meeting for purposes of a quorum as set forth in Article 22 above and for the purpose of counting the votes. |
(c) | A voting instrument that is received by the Company as set forth in Article 27(b) above, for a specific matter for which a vote was not taken in the General Meeting, shall be considered as abstaining on the vote in that General Meeting on the resolution for an adjourned Meeting pursuant to the provisions of Section 74 of the Companies Law, and it will be counted in the adjourned Meeting that will be held pursuant to the provisions of Sections 74 or 79 of the Companies Law. |
28. Minutes
(a) | The Company shall keep minutes of the proceedings (protocols) in the General Meeting, and shall keep them in the Office, for a period of at least seven years from the date of the General Meeting. |
(b) | A declaration by the Chairman of the General Meeting that a resolution has been carried unanimously, or carried by a particular majority, or lost, and an entry to that effect in the minutes (protocols) of the Company, shall be conclusive evidence of the fact without proof of the number or proportion of the votes recorded in favor of or against such resolution. |
29. Meetings of a class
The provisions of Articles 18-28 above shall apply, mutatis mutandis, on a meeting of shareholders of a class of shares, insofar as the Company must hold them.
29A. Saving Provisions
Notwithstanding anything else to the contrary in these Articles, should the Companies Law or other Applicable Law afford or allow any greater degree of discretion, flexibility or dispensation to the Company, the Board of Directors, the Chairman of the Board of Directors, the Chairman of the General Meeting and/or a Board resolution convening a General Meeting regarding any matter described in Articles 18-29 (as the case may be), whether such discretion, flexibility or dispensation apply by virtue of any law, regulation or any derogation therefrom, then to the greatest extent afforded by Applicable Law, the Board of Directors may elect to utilize or make available such discretion, flexibility or dispensation.
Part Seven: The Board of Directors
30. Members of the Board
(a) | The number of Directors in the Company, shall be determined from time to time by a resolution of the annual general meeting, provided that the number of Directors (including External Directors, if any are appointed) shall not be less than five (5) Directors and no more than eight (8) Directors. |
17
(b) | The Directors (other than External Directors, if any are appointed), until their maximum number as set forth in subsection (a) above, shall be elected by a resolution of the General Meeting, and each Director shall function in his capacity until his office is vacated or another Director is chosen in his stead. A member of the Board whose term of office has ended may be reelected. |
(c) | The Board may, in its discretion but subject to Applicable Law, determine from time to time any number of Directors who must possess accounting and/or financial expertise. |
(d) | The office of a Director shall begin from the date of his appointment, or a later date if the resolution of his appointment so provides. |
(e) | The Board of Directors is entitled at any time and from time to time to appoint any person as a Director, provided that the number of Directors does not exceed at any time the maximum number as specified above. A Director who is so appointed, shall serve insofar as his office is not vacated in accordance with the provisions of Article 35 herein. |
(f) | The Company will maintain in the Office a register of Directors and their alternates, if they have alternates pursuant to the provisions of Article 34 herein, which will be open for inspection by any person. |
(g) | Subject to the provisions of the Companies Law, all the activities and resolutions of the Board, a committee of the Board or a Director who is acting by virtue of his office, as well as any act that is taken according to their instructions, shall be valid, even if it is discovered afterwards that there was a defect in the appointment of a Director/Directors or if all or one of them were unfit from serving as Directors, as if each of them was appointed lawfully and as if they all had the necessary qualifications to be a member of the Board or committee. |
31. Restrictions on the appointment of Directors
(a) | A candidate for Director must disclose to the Company if he was convicted of an offense as described below, and five years have not yet passed since the conviction was issued or - in respect of sub Article (3) herein - the period that was established by the court according to that sub Article: |
(1) | Offenses according to Sections 290 to 297, 392, 415, 418 to 420 and 422 to 428, of the Penal Law, 5737-1977, and according to Sections 52c, 52d, 53(a) and 54 of the Securities Law; |
(2) | A conviction in a foreign court for the offenses of bribery, fraud, corporate administrative offenses or insider trading; or |
(3) | A conviction for another offense which the court holds that due to its nature, severity or circumstances, he is not fit to serve as a director in a public company, for the period that the court determines which shall not exceed five years from the date of the judgment. |
(b) | A candidate for Director in the Company will disclose if the administrative enforcement board imposed on him any enforcement measures that prevent him from serving as a director of a public company or a private company which is a bonds company, and the period established by the administrative enforcement board in its decision has not yet passed. |
(c) | A person convicted by a judgment of an offense enumerated in Article 31(a) above shall not be appointed as a Director, unless the period stated in said Article expired (unless a court establishes otherwise as stated in Section 226(b) of the Companies Law), and a person shall not be appointed as Director if the administrative enforcement |
18
board imposed on him enforcement measures prohibiting him from serving as a Director in a company, for a period determined by such board.
(d) | A Director shall not be appointed if he is a minor, legally incompetent, or declared bankrupt so long as he has not been absolved. |
(e) | A candidate for Director who is one of the above in sub-Article (d) shall disclose this to the Company. |
32. External Directors
(a) | External Directors, if and to the extent any are appointed, will satisfy the conditions for External Directors as set forth in the Companies Law and may be appointed only by a General Meeting in accordance with the relevant provisions of the Companies Law. |
(b) | If and to the extent External Directors are appointed in accordance with Article 32(a) above, at least one such External Director shall serve in each committee that is entitled to exercise any of the authorities of the Board. |
(c) | The terms of office of an External Director, if any are appointed in accordance with Article 32(a), shall be three years, which may be extended to a maximum of two additional terms of three years each. |
(d) | An External Director, if any is serving as such after being duly appointed in accordance with Article 32(a), shall not be removed, and his term of office shall not be truncated, other than according to the provisions of the Companies Law relevant to the removal of an External Director. |
(e) | In the event the Board of Directors elects, in accordance with Israel’s Companies Regulations (Relief for Companies Traded on a Stock Exchange Outside of Israel), 5760-2000, or any successor regulations, to opt out from the requirement of the Companies Law to appoint External Directors, then, for so long as such election of the Board of Directors remains in effect, all references to External Directors in these Articles shall be disregarded. |
33. Revoked.
34. Alternate director
(a) | Subject to the provisions of the Companies Law, each Director may appoint another person capable of being appointed Director as an alternate director and may revoke that person’s appointment at any time. |
(b) | An appointment of an alternate director and the revocation of his appointment shall be done by written notice to the Company by the appointing Director or in another manner as decided by the Board of Directors. The appointment will enter into effect upon receipt of the notice by the Board of Directors or the Chairman of the Board, or a later date if stated in the notice. |
(c) | An alternate director shall be directly liable to the Company, and shall have the same duties, responsibilities, and obligations to the Company as a Director. |
(d) | The appointment of an alternate director shall not negate the liability of the Director for whom he is serving as alternate. The liability of the appointing Director shall continue apply notwithstanding the alternate directors assumption of any duty or authority, taking into account the circumstances of the situation, including the appointment of the alternate director and the terms of his office. |
19
(e) | The alternate director shall have all the authorities belonging to the Director for whom is serving as the alternate. |
(f) | An alternate director shall not be entitled to participate and vote in a meeting of the Board in which the Director who appointed him participates. |
(g) | An alternate director who is a currently serving Director may not be appointed an alternate to another member of the Board of Directors, but may be appointed as an alternate director for a Director in his capacity of membership in a Board committee, provided that such candidate for alternate director is not a member of that committee and provided further that if he is an alternate director for an External Director (if and to the extent an External Director is appointed in accordance with Article 32(a)), that candidate must himself be a duly appointed External Director with Accounting and Financial Expertise or with Professional Ability as those terms are defined under the Companies Law, in accordance with the qualifications of the External Director for whom he is serving as an alternate. |
(h) | The office of an alternate director or an attorney shall be vacated: |
(1) | Automatically if the office of the Director, for whom he is serving as the alternate, is vacated for any reason; |
(2) | If the alternate director experiences any of the instances enumerated in Article 35 herein or if for another reason established in the Companies Law he is not fit to serve as a Director or an alternate director; or |
(3) | His appointment as an alternate director is cancelled by the person who so appointed him. |
35. Dismissal of a Director
(a) | The office of Director shall be automatically vacated upon the occurrence of each of the following instances: |
(1) | Upon his death; |
(2) | He is found to be legally or mentally incompetent or mentally ill. |
(3) | He is declared to be bankrupt; |
(4) | If he resigns by written notice to the Company as stated in Article 35(b) herein; |
(5) | If he is dismissed by a resolution of the General Meeting as set forth in Article 35(c) herein or is dismissed as stated in Article 35(d) herein; |
(6) | On the date of the issuance of the notice of a conviction for an offense as set forth in Article 35(e) herein; |
(7) | According to a decision by a court pursuant to the provisions of Section 233 of the Companies Law; |
(8) | On the date of the notice about the imposition of enforcement measures by an administrative enforcement board prohibiting him to serve as director of a public company or in the Company, as set forth in Section 232(a) of the Companies Law; or |
(9) | A condition needed pursuant to the Companies Law no longer exists in regard to the Director in order for him to serve as Director or a cause for the expiration of his term as Director exists. |
20
(b) | A Director or an alternate director may resign by delivery of written notice to the Board of Directors, the Chairman of the Board or the Company and his resignation shall enter into effect on the date the notice is delivered, unless another date is specified in the notice. A Director or alternate director shall state the reasons for his resignation. |
A notice received of the resignation of a Director or an alternate director, shall be brought before the Board and the minutes of the first meeting convened after the resignation, shall record the fact of the resignation and the reasons given for it.
(c) | The General Meeting may at any time dismiss a Director, by a resolution, provided that the Director is given a reasonable opportunity to bring his position before the General Meeting. |
(d) | If the Company becomes aware that a Director or an alternate director was appointed contrary to the provisions of Article 31(d) above (namely, Section 227(a) of the Companies Law) or contrary to the provisions of Article 31(c) above (namely, Sections 226(a) and (al) and 226A of the Companies Law), or that the Director violated the provisions of Article 31(a) above (namely, Section 225 of the Companies Law), Article 31(e) above (namely, Section 227(b) of the Companies Law), or the provisions of Article 35(e) herein (namely, Section 232 of the Companies Law), the Board must decide in the meeting of the Board convened right after it becomes aware of such, to end the service of said Director, if it finds that the stated conditions are present, and from the date of the resolution, the service shall expire. |
(e) | A Director who is convicted of an offense as stated in Article 31(a) above shall notify the Company of such and his service will end on the date of the delivery of the notice, and he may not be re-appointed as a Director, unless the period in which the Director may not serve has expired, as stated in Article 31(c) above (namely, Sections 226(a) and 226(a1) of the Companies Law). If the administrative enforcement board decides to impose on a person enforcement measures which prohibit him to serve as director in any public company or the Company, he will notify the Company and his term will expire on the date of the delivery of the notice, and he may not be reappointed as a Director, unless the period of the prohibition has expired as set forth in Article 31(c) above (namely, Section 226(a) of the Companies Law). |
(f) | A Director (including an External Director, if appointed as such) who no longer meets a requirements pursuant to the Companies Law in order to serve as a Director (including those of an External Director, if appointed as such) or if a reason for his service as Director to expire exists, will notify of such immediately to the Company, and his service shall expire on the date of the delivery of said notice. |
(g) | A Director who violates the duty of disclosure pursuant to Article 31(a) above (namely, Section 225 of the Companies Law), Article 31(f) above (namely, Sections 227(a) and 245(a) of the Companies Law), Article 31(e) above (namely, Section 227(b) of the Companies Law), or Article 35(e) above (namely, Sections 232 and 232A of the Companies Law), shall be considered as someone who violated his fiduciary duty to the Company. |
36. Authorities of the Board of Directors
(a) | The Board shall delineate the policy of the Company and supervise the performance of the CEO and his activities, including the authorities listed in Section 92(a) of the Companies Law. |
(b) | The authorities of the Board of Directors pursuant to Article 36(a) above may not be delegated to the CEO other than as set forth in Article 10(b) above. |
(c) | Without derogating from, restricting or reducing any specific or general authority conferred on the Board of Directors pursuant to Article 36(a) above or any other |
21
authority conferred on it by these Articles or by Applicable Law, the Board of Directors shall have the following additional authorities:
(1) | To appoint a person or persons (incorporated or otherwise), to receive and hold in trust for the Company any property belonging to the Company or in which the Company has an interest, or for any other purpose, and to do or perform any activity, act or things needed with respect to any such trust, and to act to pay the salaries of the trustee or trustees; |
(2) | To establish the authorized signatories of the Company for bills of exchange, promissory notes, receipts, endorsements, checks, dividend certificates, releases, contracts and other documents of any kind; |
(3) | To appoint, and in its discretion, to remove or suspend a CEO, manager, secretary, clerk, employee or agent, whether if they are employed on a permanent or interim basis or for special services, as the Board of Directors sees fit from time to time, and to define their authorities and obligations and to set their salaries and wages and to demand guarantees, in the cases and in the amounts that the Board deems fit; |
(4) | To establish local management for the management of any of the businesses of the Company in a specific place in Israel or abroad, and to appoint any persons to be local managers and to determine their wages or to dismiss any of these people from their service, and from time to time and at any time delegate to any person who is so appointed any powers or authorities or discretion that is conferred at such time on the Board, and to authorize the members at that time in any local management, to fill any vacancy in it and to act notwithstanding the vacancies; |
Any such appointment or delegation may be done under the same terms and subject to the same conditions that the Board deems proper in accordance with the Companies Law, and the Board may at any time cancel any appointment or delegation or change them. The Board may authorize the persons to whom powers, authorities or discretion were delegated and which are conferred on them at such time, to delegate them, all of some, with a secondary delegation;
(5) | Subject to the provisions of Applicable Law, to appoint by power of attorney any person or persons to be the attorney or attorneys of the Company for the purposes and with the powers, authorities and discretion (which shall not exceed those given or conferred for use by the Board according to these Articles or by law) for a period of time and subject to the same terms as the Board deems proper from time to time, and any such appointment may be given (if the Board sees fit to do so) to any local manager, or any company or its members, its directors, agents or managers of any company or firm or a person who is established by any company or firm. Any such power of attorney may contain in it authorities for the protection or convenience of persons who come into contact with these attorneys as the Board deems fit; |
(6) | To open, manage, defend, compromise, or neglect any legal proceedings on behalf of or against the Company or against its officials or related in another manner to its affairs and to compromise or extend the time for payment or defrayal of any debt owed or actions or demands by the Company or against it; |
(7) | To deliver for arbitration any action or demand of the Company or against it; |
(8) | To appoint on behalf of the Company an attorney or attorneys in Israel or abroad to represent the Company before any court, legal and quasi legal bodies, government offices or bodies, municipal or otherwise in Israel or abroad and to confer on such attorney the authorities that the Board feels |
22
proper to give, including the authority to delegate his authorities, in whole or in part, to another or others;
(9) | Subject to the provisions of the Companies Law (including Section 113 of thereof) and these Articles, to delegate to any person, firm, Company or group of persons as stated, the powers, authorities and discretion conferred on the Board of Directors; |
(10) | The Board is entitled to exercise any authority of the Company which was not conferred by law or these Articles to another organ of the Company. |
37. Assumption of authorities of the Board
(a) | The General Meeting may assume authorities given to the Board for a specific matter, or for a specific time frame, that does not exceed the time required under the circumstances. The assumption of authorities shall be done upon the adoption of a shareholders’ resolution to such effect in the General Meeting. |
(b) | If the Board cannot exercise its authorities and the exercise of any of its authorities is essential for the proper management of the Company, the General Meeting may exercise it in its stead, so long as the Board is prevented from doing so, provided that the General Meeting establishes that in fact the Board cannot do so and that the exercise of the authority is essential as stated. |
(c) | If the General Meeting assumes authorities conferred by law on the Board, the shareholders shall have the rights, duties and liability applicable to the Directors for the matter of the exercise of those authorities, mutatis mutandis, and, the provisions of chapters three, four and five of the Sixth Part of the Companies Law shall apply to them, taking into account their holdings in the Company, their participation in the Meeting and the manner of their vote. |
38. The rights of a Director
Subject to Applicable Law and the issuance of the required approvals, a Director shall not be disqualified, because of his office, from holding another office in the Company or in any other company in which the Company is a shareholder, or in which it has another benefit or from entering into a contract with the Company, whether as a vendor or buyer or in another manner.
39. Chairman of the Board
(a) | The Board of Directors will choose, dismiss, with a normal majority of votes, one of the members of the Board to serve as Chairman of the Board, and the provisions in Articles 39(b)-(f) below will apply to him. |
(b) | The term of service of the Chairman of the Board shall be until a resolution of the Board of the termination of his service and appointment of another Chairman in his stead. However, it is understood, that an outgoing Chairman may be re-appointed as Chairman. |
(c) | If the service of a Director is vacated for one of the instances listed in these Articles and said Director is the Chairman of the Board, his appointment as Chairman shall automatically expire, and another Chairman shall be chosen in his stead. |
(d) | The Chairman of the Board shall set the agenda as set forth in Article 41 herein and will preside over the meetings of the Board. |
(e) | If the Chairman of the Board of Directors is absent from a meeting 15 minutes from the designated time for the meeting or if he is unwilling to preside over the meeting, the Board of Directors shall elect one of its members to preside over the meeting and sign the minutes of the meeting. The Chairman of the Board in such instance shall not |
23
have an extra or casting vote in any vote by the Board of Directors in the event of a tie vote.
(f) | The Chairman of the Board may serve as the CEO of the Company or exercise his authorities for periods that do not exceed three years each from the date of the resolution, subject to and in accordance with the provisions of Section 121(c) of the Companies Law. |
40. Convening a meeting of the Board
(a) | The Board of Directors will convene for meetings pursuant to the needs of the Company and at least once every three months. |
(b) | The Board will be convened according to one of the following methods: |
(1) | The Chairman is entitled to convene a meeting at any time. |
(2) | In the following instances, the Chairman of the Board will convene the Board without delay: |
1. | A notice or report by the CEO to the Chairman of the Board about any irregular matter that is material to the Company that requires an act by the Board; and |
2. | Notice by the Auditor of the Company to the Chairman of the Board that he became aware during its Audit Activity of material deficiencies in the accounting audit of the Company. |
(3) | The Chairman of the Board will convene the Board, at the demand of: (i) any two Directors at any time, (ii) a single Director at any time, if at such time the number of Directors in office is five (5) or less, (iii) any Director who at any time becomes aware of a matter of the Company in which there may be an apparent violation of law or a material flaw in proper corporate governance, in which case that Director shall inform the Chairman or the Board of such, and the Chairman or the Board once so informed shall act without delay to convene a meeting of the Board. |
(c) | If a meeting of the Board is not convened within seven days from the date of the notice or report by the CEO or the Auditor as stated in Article 40(b)(2) above or from the date of the demand as set forth in Article 40(b)(2) above, each of those listed above, may convene a meeting of the Board to discuss the subject specified in the demand, notice or report, as the case warrants, within at least two business days prior to the date of the meeting. |
(d) | The Board (including any committee of the Board) may hold meetings through the use of any communications devices or other electronic devices, provided that all the Directors participating can hear each other simultaneously and all members of the Board or the Board committee participating by such means shall be considered “present” and have “participated” at such meeting, including with regards to establishing a quorum and voting. |
(e) | The Board may adopt resolutions even without an actual meeting (such as in writing, fax or email), provided that all the directors who are entitled to participate in the meeting and vote on the matter brought for a resolution agree to do so. |
(f) | Resolutions adopted in the manner specified in subsection (e), shall be formalized in a protocol, including the resolution not to convene a meeting, and the protocol shall be signed by the Chairman of the Board. |
24
(g) | Subject to the provisions of Applicable Law, the Company may enter into any contract or otherwise transact any business with any Officer in which contract or business such Officer has a personal interest, directly or indirectly and may enter into any contract or otherwise transact any business with any third party in which contract or business an Officer has a personal interest, directly or indirectly. |
41. Agenda
The agenda of Board meetings shall be set by the Chairman of the Board and shall include:
(a) | Subjects set by the Chairman of the Board; |
(b) | Subjects that were set as set forth in Article 40 above; |
(c) | Any subject regarding which a Director, or the CEO submits a written request to the Chairman of the Board, a reasonable time prior to the meeting; and |
(d) | A subject in connection with a substantial flaw detected by the Auditor in the course of an Audit Activity, as described by the Auditor in writing to the Chairman of the Board, a reasonable time prior to the meeting. |
42. Notice of a meeting of the Board of Directors
(a) | Notice of a meeting of the Board shall be delivered to all the Directors at least seventy-two (72) hours prior to the date designated for the meeting, unless all the Directors gave prior written consent to convene the meeting within a shorter time frame, or in urgent cases - and with the consent of a majority of the Directors - even without such notice. |
(b) | Notice pursuant to Article 42(a) above shall be delivered to the address of the Director (including e-mail address) that was previously delivered to the Company by the Director in writing and which shall state the date of the meeting and the location, and a reasonable description of all the subjects on the agenda. It is understood that the dispatch of such notice covers the liability of the Company and the Director is solely responsible to update the Company about a change of his address for the purpose of the sending of such notices. A change of address of the Director shall be done by him in writing provided that it is delivered a reasonable time prior to the date designated for a meeting of the Board of Directors. |
43. Quorum
(a) | A quorum for discussion in meetings of the Board shall be the presence of half of the Directors who serve at the time of the meeting, on their own or through alternates. The quorum shall be established at the start of each meeting of the Board and shall constitute a quorum for the entire duration of the meeting, for all the resolutions that are on the agenda, even in the case or cases where a quorum is not present during the continuation of the meeting. |
(b) | If a half hour passes from the time designated for the start of the meeting of the Board and a quorum is not present, the meeting shall be adjourned for twenty four (24) hours exactly (after the original time designated for the meeting) or to another time set by the Chairman of the Board (but in any case no earlier than twenty four (24) hours). The quorum at an adjourned meeting shall be the presence of at least two Directors, who are serving at the time of the meeting, on their own or through an alternate. If the Board cannot act due to the absence of a quorum at the adjourned meeting, the General Meeting may exercise the authorities of the Board for the purpose/s for which the meeting of the Board was convened and the provisions of Article 37 above will apply. |
(c) | Each duly convened meeting of the Board of Directors, in which a quorum is present, shall have all the authorities, powers and discretions given at such time in accordance |
25
with the provisions of these Articles to the Board of Directors or those exercised by it in general.
(d) | If a specific member is not appointed to the Board or if the office of a Director is vacated, the remaining Directors may operate for all matters, so long as their number is not less than the minimum fixed in Article 30(a) above. If the number is less than the minimum, they may not exercise their authorities according to these Articles, except to convene a General Meeting with an agenda to appoint additional Directors or to establish a lower minimum of Directors or to appoint additional Directors themselves. The General Meeting may decide not to approve acts of the Directors when their number falls below the minimum number and to exercise on its own the authorities of the Board, until the number of Directors again reaches the minimum as set forth in Article 30(a) above. |
44. Voting on the Board
(a) | Each Director shall have one vote in each vote on a resolution. |
(b) | Resolutions of the Board shall be adopted by a regular majority of those present participating in the vote. |
(c) | If the votes are tied in a Board meeting, the proposed resolution shall be considered as rejected. |
(d) | A Director (or alternate director) is entitled to vote by physical presence or by means of communication or other electronic means where the Directors who are participating can hear or see each other simultaneously. |
45. Minutes of a meeting of the Board
(a) | The Company will keep minutes of the proceedings in meetings of the Board and its committees and will keep them and the resolutions adopted in writing in lieu of actual meetings of the Board, in the Office for a period of seven years from the date of the meeting or adoption of the resolution, as the case warrants. |
(b) | Minutes approved and signed by the Director who presided over the meeting, shall serve as prima facie proof of its contents. |
(c) | An announcement by the Chairman of the Board, that a resolution was adopted unanimously or by a specific majority, or was rejected and a notation recorded in this matter in the minutes of the meeting of the Board, shall serve as conclusive evidence of the authenticity of its contents, and it is not necessary to prove how many votes there were or how many were for or against the resolution. |
46. Defects in the convening of a meeting
(a) | A resolution adopted in a meeting of the Board that was convened without the prior conditions satisfied for its convening (hereinafter - “Defect in the Convening”) may be revoked at the demand of each of the following: |
(1) | A Director who was present at the meeting, provided that he demanded that a resolution for which the defect was present not be adopted, prior to the adoption of the resolution; or |
(2) | A Director who was entitled to be invited to a meeting but was not present, within a reasonable time after he was informed about the adoption of the resolution and no later than the first Board meeting that was held after he was informed of the resolution; |
26
It is understood that if there was a Defect in the Convening of the meeting related to the notice about the location of the meeting or its time, a Director who came to the meeting may not, notwithstanding such defect, demand the revocation of the resolution.
(b) | The provisions of Article 46(a) above shall not impair from the validity of an act done for the Company which was retroactively approved by the Board or if the party with whom the act was done did not know or could not have known about the irregularity or lack of authorization. |
47. Committees of the Board
(a) | Subject to the provisions of Section 112 of the Companies Law which prohibits the delegation of authorities and the provisions of these Articles (including Article 44(d) above), the Board may establish committees of the Board, provided that only members of the Board are appointed to such committees (hereinafter: “Board Committee”) and delegate all or some of its authorities to a Board Committee. The Board may from time to time cancel the delegation of said authority, |
Each Board Committee that is so established must, when exercising its authorities, comply with all the regulations that are established by the Board of Directors.
(b) | A Board Committee will report to the Board on a regular basis about its decisions or recommendations. Decisions or recommendations of a Board Committee which requires the approval of the Board, will be brought to the attention of the Directors a reasonable time prior to the deliberations on the Board. |
(c) | The meetings of a Board Committee and its management shall be in accordance with the provisions of procedures and management of meetings of the Board, as set forth in the provisions of these Articles, mutatis mutandis, so long as they are appropriate and if they do not replace the instructions that are given by the Board in accordance with this Article. |
(d) | A committee of the Board with a mandate to provide counsel or recommendations to the Board may include among its membership a person who is not a member of the Board who shall act on such committee and be invited to and participate in its meetings solely in an advisory and/or consulting capacity. |
(e) | A resolution that is adopted or an act that is done by a Board Committee, according to an authority that was delegated to it from the authorities of the Board, shall be the same as a resolution adopted or an act that was done by the Board. However, the Board may revoke any decision of a Board Committee that it appointed, but such cancellation shall not harm the validity of a decision of a committee where the Company acted in accordance therewith in relation to another person who was not aware of the revocation. |
Part Eight: Audit Committee
48. Appointment of an audit committee
(a) | The Board of the Company shall appoint among its members an audit committee. The number of members of the audit committee shall be determined by the Board, from time to time provided that it shall not be less than three members, and further provided that all External Directors, if and to the extent any are appointed in accordance with the relevant provisions of the Companies Law, will be members of the committee. The Chairman of the Board and any Director who is employed by a person exercising Control over the Company (“a Controlling Shareholder”) or by a corporation under the control of a Controlling Shareholder, or a Director who provides services, on a regular basis, to a Controlling Shareholder or to a corporation under the Control of a Controlling Shareholder, as well as a Director whose main income is dependent upon |
27
the Controlling Shareholder, shall not be members of the audit committee. Likewise, a Controlling Shareholder or a Relative thereof shall not be members of the audit committee.
(b) | The audit committee shall choose one of its members, who will be an External Director, if and to the extent there are any External Directors serving on the Board, to serve as Chairman of the audit committee, by a resolution adopted by a regular majority of the audit committee present at such meeting. |
(c) | The term of office of the Chairman of the audit committee shall be until a resolution of the audit committee about the termination of his term and the appointment of a Chairman for the audit committee in his stead. However, it is understood that a Chairman of the audit committee who ended his term of service may be reappointed. |
49. Audit committee
(a) | The audit committee shall carry out those functions described in Section 117 of the Companies Law. |
(b) | The Internal Auditor of the Company shall receive notices of meetings of the audit committee and may participate in such meetings. The Internal Auditor may ask the Chairman of the audit committee to convene the committee to discuss a subject that he describes in his request, and the Chairman of the audit committee will convene the meeting within a reasonable time from the request, if he sees a reason to do so. |
(c) | A notice of a meeting of the audit committee, in which a subject related to the audit of the financial statements is raised, shall be delivered to the Internal Auditor who is entitled to participate in it. |
(d) | Subject to the provisions of the Companies Law, the procedures of the meetings and activities of the audit committee and its management shall be in accordance with the provisions of the procedures and management of meetings of the Board of Directors, as described in these Articles, mutatis mutandis, insofar as they are appropriate and insofar as they do not replace instructions given by the Board pursuant to this Article. |
Part Nine: Exemption, indemnification and liability insurance
50. Exemption and indemnification
(a) | Exculpation: The Company may exempt in advance an Officer from his liability, in whole or in part, for damage due to a breach of the duty of care to the Company, other than a breach of the duty of care in a Distribution. |
(b) | Indemnification. The Company may indemnify an Officer, in whole or in part, for any or all of the liabilities or expenses as described in paragraphs (1)-(6) below, imposed on him pursuant to an act or omission committed in such Officer's capacity as an Officer of the Company: |
(1) | A financial liability imposed on him in favor of another person by a court judgment, including a judgment affirming a settlement or an arbitration award that is ratified by a court; |
(2) | Reasonable litigation costs, including legal fees, expended by the Officer following an investigation or proceeding that was conducted against him by an authority authorized to carry out such investigation or proceeding, provided that such investigation or proceeding concluded either: (i) without any indictment filed against him and without any financial liability imposed on him in lieu of a criminal proceeding; or (ii) without any indictment filed against him but with a financial liability imposed in lieu of a criminal |
28
proceeding regarding an offense that does not require proof of criminal intent; or (iii) with a non-criminal financial sanction;
In this context, the terms “conclusion of a proceeding without filing an indictment in a matter in which a criminal investigation was opened” and – “a financial liability in lieu of a criminal proceeding” or similar terms of different wording, shall be interpreted in accordance with the specific meanings ascribed to those terms by Section 260(a)(la) and Section 260(a)(1) of the Companies Law, respectively;
(3) | Reasonable litigation costs, including legal fees that the Officer incurred or which he was charged to pay by a court, in a proceeding filed against him by the Company or on its behalf or by another person, or in a criminal indictment for which he was acquitted, or an indictment for which he was convicted of a crime that does not require proof of criminal intent; |
(4) | Costs, including reasonable litigation costs including attorneys' fees, expended by the Officer in connection with an administrative enforcement proceeding that conducted in his regard. |
For this purpose “an administrative enforcement proceeding” – means a proceeding pursuant to Parts 8(3) (Imposition of Financial Sanctions by the Securities Authority), 8(4) (Imposition of Administrative Enforcement Measures by the Administrative Enforcement Committee), or 9(1) (Arrangement to Prevent the Commencement of Proceedings or to Halt Proceedings that is Predicated on Conditions) of the Securities Law and a proceeding according to Section D of Chapter Four in Part Nine of the Companies Law and subject to any other relevant Applicable Law, any similar proceeding to these, by whatever name it is called.
(5) | Payment to a person injured by a violation as stated in Section 52BBB(a)(l)(a) of the Securities Law. |
(6) | Any liability or expense resulting from any other act or omission committed in his capacity as an Officer of the Company, for which indemnification can be made under Applicable Law. |
(7) | Without derogating from the generality of the foregoing, expenses paid by the Officer in connection with a proceeding conducted under the Economic Competition Law, 5748-1988, including reasonable legal expenses. |
(c) | The Company may provide an undertaking to indemnify an Officer, subject to the relevant provisions of Applicable Law (an “Indemnity Undertaking”). An Indemnity Undertaking can be given regarding either or both of the following circumstances: |
(1) | as described in Article 50(b)(1) above, provided that the Indemnity Undertaking is limited to: (i) events which the Board deems foreseeable in light of the Company's actual activities at the time of giving the Indemnity Undertaking, and (ii) an amount or criteria which the Board has determined to be reasonable under the circumstances, and further provided that the Indemnity Undertaking mentions: (x) those events which the Board regards as foreseeable in light of the Company's actual activities at the time the Indemnity Undertaking is given, and (y) the amount or criteria which the Board has determined to be reasonable under the circumstances; or |
(2) | as described in Articles 50(b)(2)-(6) above. |
(d) | The Company may indemnify any Officer after the fact, in whole in part, as the Company may, by way of shareholders’ resolution, decide. |
29
(e) | The Company may make an undertaking to any Company employee, including an Officer who is not a Director of the Company, who serves or has served as the Companies' representative or by the Company's request as director of another company in which the Company holds shares, directly or indirectly (a “Director of Another Company”), to indemnify him for any liability or expense enumerated in Article 50(b) above, that may be incurred pursuant to an act or omission committed by him in his capacity as the Director of Another Company, provided that such undertaking is limited to the types of events which the Board deems foreseeable at the time of giving such indemnity, and to an amount which the Board has determined to be reasonable under the circumstances. |
(f) | Without derogating from the generality of Article 50(e) above, the Company may indemnify a Director of Another Company after the fact, for any liability or expense listed in Article 50(b) above, that was incurred pursuant to an act or omission committed by him in his capacity as the Director of Another Company. |
51. Liability insurance
(a) | The Company may enter into a contract for liability insurance for an Officer for reasonable legal expenses and/or a liability imposed on that Officer for an act or omission taken by virtue of his being an Officer of the Company, any subsidiary of the Company or any Affiliate of the Company, for any or all of the following: |
(1) | A breach of the duty of care towards the Company or another person; |
(2) | Breach of a fiduciary duty towards the Company provided that the Officer acted in good faith and had reasonable grounds to assume that the such act or omission would not harm the Company’s interests; |
(3) | A monetary obligation that is imposed on him in favor of another person; |
(4) | A payment to a person injured by a violation of Section 52BBB(a)(1)(a) of the Securities Law, or any expense incurred by the Officer regarding an administrative enforcement proceeding (as defined in Article 50(b) above); |
(5) | Any expense incurred by the Officer in connection with a proceeding in his regard under the Economic Competition Law 5748-1988, including reasonable legal costs including attorneys' fees; |
(6) | Any other matter or liability which may be insured by law, and to the extent such law may require the inclusion of a provision of these Articles specifically permitting such insurance in these Articles, such provision is deemed to be included and incorporated herein by way of reference; |
(b) | In any case where the insurance contract as aforementioned in Article 51(a) above will also cover the Company, the Officer shall take precedence over the Company in receiving insurance compensation. |
(c) | Nothing in Article 50 above or in this Article 51 shall be interpreted so as to limit the Company in any matter of exculpation, indemnity or insurance: |
(1) | in connection with a person who is not an Officer of the Company or Director of Another Company, including, without derogating the generality of the foregoing, any employee or consultant; or |
(2) | in connection with an Officer of the Company or Director of Another Company, insofar as such exculpation, indemnification is not proscribed by law. |
30
(d) | The provisions of Articles above and this Article 51 shall apply to an alternate director, mutatis mutandis. |
Part Ten: General Manager (Chief Executive Officer)
52. The Chief Executive Officer
(a) | A CEO of the Company will be appointed and dismissed in accordance with a resolution adopted by the Board of Directors of the Company, and the Board may appoint more than one CEO, for a fixed period of time or without any time limitation, and it may from time to time dismiss or release him or them from their office and appoint another or others in his or their stead. |
(b) | Subject to the provisions of an employment agreement between the CEO and the Company and subject to Applicable Law, the CEO is responsible for the ongoing management of the affairs of the Company as part of the policy set by the Board and subject to its instructions and he shall be supervised by and report to the Board. |
(c) | Subject to the provisions of the agreement between the CEO and the Company, the CEO will have all the authorities of management and implementation that were not conferred by law or these Articles to another organ of the Company. |
(d) | Subject to the provisions of Applicable Law, the Board of Directors may from time to time confer upon or delegate to the CEO, any of the authorities conferred upon the Board by virtue of Article 36(c) above, as it deems fit to manage the ordinary business of the Company and it may confer authorities for a period of time, and for certain purposes and needs for those times and under such conditions and restrictions as it deems fit. Nothing in this Article 52(d) or in Article 36(c) shall be interpreted to derogate from an authority which the CEO would otherwise have possessed. |
(e) | The CEO must notify the Chairman of the Board of Directors about any irregular matter that is material to the Company; if the Company does not have a Chairman of the Board or if the Chairman of the Board is prevented from serving in such capacity, the CEO will notify all the Directors. |
(f) | Any Officer of the Company, other than Directors and the CEO or [chief operating officer] (namely, a chief business manager, CFO, deputy to the CEO or vice-CEO, in-house legal advisor, any person fulfilling such role in the Company even if his title is different, or any other manager or officer subject directly to the CEO) may be appointed and dismissed by the CEO, subject to the provisions of the Companies Law dealing with the approval of the terms of service and employment of an Officer. |
53. Delegation and qualification of the CEO’s authorities
The CEO may, with the Board's approval, delegate some of his authorities to another person who reports directly to the CEO. The Board of Directors may instruct the CEO how to act regarding any specific matter; if the CEO does not fulfil such instructions and/or if the CEO is prevented, for any reasons, from exercising his authorities, in whole or in part, the Board of Directors may exercise those authorities in his stead.
Part Eleven: Management of the Company
54. Registered office
(a) | The Company will maintain an Office in Israel, to which any notice to the Company can be sent. Without derogating from the provisions of any law, the Company will keep in its Office documents as set forth in Section 124 of the Companies Law. |
31
(b) | Delivery of a document to the Company shall be to the Office as it is registered with the Companies Registrar, and shall be considered delivered upon the delivery of such document to such Office. |
(c) | A person who is entitled to inspect documents, is entitled to receive a copy of them, for a fee that the Board or the CEO establishes. |
55. Register of shareholders and register of material shareholders
(a) | The Company shall keep a register of shareholders and a register of material shareholders and will update the changes to them as soon as possible after it becomes aware of them. |
(b) | The register of shareholders and the material shareholder register shall be open for inspection by any person. |
(c) | The details enumerated in Section 130(a) of the Companies Law shall be recorded in the register of shareholders. |
(d) | The material shareholder register shall contain reports that the Company received pursuant to Applicable Law about the holdings of the material shareholders in Company shares. |
(e) | The Company will keep all the records that are recorded in the register of shareholders as set forth in Article 55(c) above. |
(f) | The register of shareholders will be prima facie proof of the contents recorded in it. |
(g) | In the case of a contradiction between the register of shareholders and a share certificate, the register of shareholders shall have more evidentiary value than that of the share certificate. |
56. Auditor
(a) | The Company will appoint an Auditor who will audit the annual financial statements of the Company and give his opinion about them (hereinafter: “Audit Activity”). |
(b) | An Auditor will be appointed at each Annual General Meeting and shall serve in his capacity until the end of the following Annual General Meeting; however, the General Meeting may appoint an Auditor who will serve in his position for a longer period of time, that shall not be longer than the end of the third Annual General Meeting after the one in which he is appointed. |
(c) | The Company may appoint a number of Auditors to carry out the Audit Activity together. |
(d) | If the office of the Auditor is vacated and the Company does not have another auditor, the Board will convene a special General Meeting, as soon as possible, with the agenda of appointing an Auditor. |
57. Expiration of the term of the Auditor
(a) | The General Meeting may terminate the service of the Auditor. |
If the agenda of the Company includes the termination of the service of the Auditor or the non-renewal of his service, the audit committee will bring its position before the General Meeting, after affording the Auditor a reasonable opportunity to bring his position before it.
32
(b) | If the Board becomes aware that there are dependent relationships pursuant to the provisions of Section 160 of the Companies Law, it will notify the Auditor without delay that he must act to cease such dependency immediately; if the dependency continues, the Board will convene a Special General Meeting within a reasonable time period, with the agenda to terminate the service of the Auditor. |
(c) | The General Meeting that is convened as set forth in Article 57(b) above, shall decide on the termination of the service of the Auditor; however, the General Meeting may, after the Auditor brings his position before it, decide not to accept the recommendation of the Board to end his service, if it finds that the Auditor has no dependency in the Company. |
(d) | The Board of Directors will give the Auditor a reasonable opportunity to bring his position before the General Meeting with the agenda of ending or not renewing his service, and for this purpose the Auditor will be invited to participate in the general meeting. |
(e) | If the Auditor resigns under circumstances in which the shareholders in the Company have an interest, the Auditor will notify the Board of same. |
(f) | Without derogating from the provisions of Applicable Law, the Board of Directors will notify the shareholders about the reasons for the resignation of the Auditor as it deems fit, and it may also give notice about its position in the matter. |
58. Remuneration of the Auditor
(a) | The remuneration of the Auditor for the Audit Activity and for additional services shall be set by the Board of Directors, in accordance with the extent of the work, the duration of his services and any additional relevant term related to his services. |
The Board will notify the General Meeting about the remuneration of the Auditor, and all matters related to his fees for additional services - also about the terms of contract with the Auditor, including payments and undertakings of the Company towards the Auditor.
For the purpose of this Article - an Auditor - includes a partner, employee or relative and including a corporation under his control.
(b) | The Company will not stipulate the payment of the fee of the Auditor on terms that limit the manner of his performance of the Audit Activity or which make a connection between the results of the audit and his fees. |
(c) | The Company or anyone on its behalf shall not indemnify, directly or indirectly, the Auditor, for an obligation imposed on him due to a breach of his professional responsibility in providing services that must be provided by an Auditor by Applicable Law, or following the violation of another duty imposed on him by law. |
59. Authorities, duties and responsibility of the Auditor
(a) | The Auditor may at any time inspect documents of the Company required by him to perform his job and receive explanations about them. |
(b) | The Auditor may participate in any General Meeting in which financial statements are submitted for which he conducted Audit Activity and any meeting of the Board which deliberates the approval of the financial statements, in meetings of the committee to inspect the financial statements and in meetings of the Board convened pursuant to Article 40(b)(2)2 above; the Board of Directors will notify the Auditor of the place and time of the General Meeting or the Board or committee meeting for the examination of the financial statements. |
33
(c) | If the Auditor becomes aware during his Audit Activity about material defects in the audit of the Company, he will notify the Chairman of the Board of such. |
60. Internal auditor
(a) | The Board of Directors of the Company will appoint an Internal Auditor; the Internal Auditor will be appointed in accordance with the recommendation of the audit committee. |
An Internal Auditor shall not be an interested party in the Company, an officer in the Company, a relative of any of the above, or of the Auditor or his representative.
(b) | The organizational supervisor over the Internal Auditor shall be the Chairman of the Board, or whoever the Board of the Company determines from time to time. |
(c) | The Internal Auditor will check, inter alia, the validity of the activities of the Company in respect of compliance with law and proper business conduct. |
(d) | The term of service of the Internal Auditor shall not be terminated without his consent and he shall not be suspended, unless the Board decides on such after obtaining the position of the audit committee, and after giving the Internal Auditor a reasonable opportunity to state his position before the Board and before the audit committee. |
For this purpose, the quorum for the opening of a meeting of the Board shall not be less than a majority of the Directors.
Part Twelve: Financial statements and signature
61. Financial statements
The Company will keep accounts, and likewise will keep financial statements pursuant to Applicable Law.
62. Stamp and signatory right
(a) | The Company may establish a stamp or rubber stamps for sealing documents. |
(b) | The Board will determine the person or persons (even if they are not directors) who are authorized to sign on behalf of the Company, and their signatures together with the stamp of the Company or its printed name shall bind the Company, provided that he or they acted and signed within their authority or authorities. |
Part Thirteen: Dividends and Bonus Shares
63. Dividends and bonus shares
(a) | A resolution by the Company to distribute dividends or allocate bonus shares may be adopted by the Board of Directors of the Company. The Board of the Company shall decide on the date for payment of such dividend. |
(b) | In addition, the Board may, prior to offering a dividend, allocate from the profits of the Company, amounts, as it deems fit, as a reserve fund or funds as they establish, in the sole discretion of the Board of Directors, for unforeseeable needs or to equalize dividends with special dividends to correct, to improve or to maintain any property of the Company, and for many other types of purposes, as the Board, according to their absolute discretion, believes is beneficial for the affairs of the Company, and it may invest these allocated sums in investments that they feel are proper (other than in shares of the Company), and from time to time manage these investments or change them and use all or some of them for the benefit of the Company, and it may divide the reserve fund into special funds, as it deems fit, and use the fund or any part of it for the |
34
Company’s business, without having to keep the monies separate from the rest of the assets of the Company.
(c) | A Board of Directors which announces the distribution of dividends may decide that this dividend be paid in full or in part by distribution of certain assets, including by the distribution of fully paid-up shares, bonds or a series of bonds of any other company, or in one or more of these methods. |
(d) | In order to validate a resolution of the Board (including according to Article 63(c) above), the Board may: |
(1) | Resolve any difficulty that may arise with respect to the distribution of a dividend and/or allocation of bonus shares as it deems fit; |
(2) | Issue partial certificates, including certificates for fractional shares or decide not to count fractions under a certain amount, or sell fractions and transfer their consideration to those eligible to receive them; |
(3) | To establish for the distribution of a dividend and/or allocation of bonus shares the value of any specific asset; |
(4) | To decide that payment in cash will be done for shareholders on the basis of the value that will be so established, or that parts the value of which are less than one shekel will not be taken into account in order to adjust the rights of all the parties; |
(5) | To deposit such monies or specific assets with trustees against securities, for persons eligible to receive dividends and/or bonus shares or to a fund that was converted into capital; |
(6) | If required, a proper contract will be drawn up and the Board may appoint a person to sign such contract on behalf of those eligible to receive dividends, bonus shares and/or fund converted into capital and such appointment will be valid; and/or |
(7) | To make any other arrangement (in respect of the distribution of dividends and/or allocation of bonus shares), as the Board of Directors deems fit according to its sole discretion. |
(e) | The Board of Directors may deduct and offset from any dividend, bonus or other monies that are due to be paid for shares held by a shareholder, whether or not he is the sole owner or holds the share jointly with others, all sums of money owed from him which he must defray on his own or jointly with any other person to the Company on account of calls for payment etc. |
(f) | A shareholder shall not be entitled to a dividend if he has not delivered by the date designated for such, a bank account into which the relevant sums are to be transferred. Further, a shareholder is not entitled to change the bank account number after a date reasonably set by the Board prior to the date for the actual distribution of the dividend by the Company. |
(g) | The Board may invest each dividend that is not claimed within one year from the announcement of its distribution or to use it in another manner for the benefit of the Company until it is claimed. The Company is not obligated to pay interest or index linkage for an unclaimed dividend. |
(h) | Shareholders entitled to a dividend, are shareholders as of the record date set, subject to Applicable Law, by the Board of Directors for such purpose. |
35
Part Fourteen: Notices and Dissolution
64. Notices
Subject to the provisions of Article 21 above (to wit notice of a meeting), the arrangement set forth in Article 21 above shall apply:
(a) | The Company is entitled to deliver notice to any shareholder by personal delivery, by fax, by email or by dispatch by mail in a letter, prepaid envelope or packaging intended for the shareholder, to the address as delivered to the Company at the time of the allocation of the shares or transfer of the shares, unless said shareholder gave written notice of a change of his address (hereinafter: “Registered Address”). |
(b) | A shareholder whose registered address is outside of Israel may, from time to time, give written notice to the Company about an address in Israel, and if so given, that address will be considered as his address for the delivery of notices - as stated above. |
(c) | All notices regarding shares, to which persons are jointly entitled, shall be delivered to the person who appears first in the register of shareholders, unless they deliver other instructions, and a notice sent as stated shall serve as sufficient notice to all these shareholders. |
(d) | Any notice sent to a shareholder to his registered address by post shall be considered as having been delivered three (3) business days from the day of dispatch of the letter or envelope or other packaging, when properly bearing the registered address of the recipient. A written certificate signed by the secretary or manager or other official of the Company that the letter, envelope or packaging containing the notice with the registered address was delivered to the post office as stated, shall serve as prima facie proof of the fact. Any notice sent by fax or email shall be considered as having been delivered one (1) day from the day it was sent, provided, with respect to fax, that confirmation of the dispatch of the fax is presented, and if hand delivered - at the time of delivery. |
(e) | Notwithstanding the foregoing, subject to Applicable Law, the Company may announce the delivery of a document or other notice to any other place determined by the Board and/or by any other means, including through the internet or by other electronic means. |
(f) | Subject to the provisions of Applicable Law, a shareholder, Director or other person entitled to receive notice in accordance with these Articles or by law, may waive the right to receive such notice, whether in advance or in retrospect, whether for a specific event or in general, and once such person does so this will be considered as if the notice is or has been duly delivered, and any proceeding or action for which such notice should have been given will be deemed valid and in force. |
(g) | A person who becomes eligible to a share by virtue of the law, a transfer or in any other manner, shall be copied on every notice for such share, that was duly delivered to the registered address of the shareholder (from whom the right to the share is derived) registered in the register of shareholders. |
(h) | Any notice or document sent by post to a shareholder or left at his registered address, then notwithstanding the fact that said shareholder died - and it does not matter if the Company knew of the death or not - shall be seen as having been duly delivered in respect of all the shares registered, whether if they were held by the same shareholder separately or jointly with other persons, until the other person will be registered in his place as the owner or the joint owner of the shares, and such delivery will be seen, for the purposes of these Articles, as sufficient delivery of the notice or the document to the personal representative, or all persons, if any, jointly interested in the same shares. Without derogating from the foregoing generality, a notice to a shareholder may be |
36
delivered to a person, including any trustee, receiver or liquidator, who has a right to receive a share owing to the death, bankruptcy, insolvency, or liquidation of a shareholder (including without limitation a "Trustee" as that term is defined in the Insolvency and Financial Rehabilitation Law, 5778-2018) upon the entry of that person in the register of shareholders in such capacity.
65. Dissolution
(a) | Without derogating from the authority of the liquidator or trustee pursuant to the Insolvency and Economic Rehabilitation Law, 5778-2018 and any other Applicable Law and subject to special conditions, benefits and restrictions attached to shares of the Company, shares of the Company shall have equal rights regarding the return of the capital and participation in the distribution of surplus assets of the Company whether the Company winds up voluntarily or whether in any other manner, after defrayal of all the obligations of the Company, its assets shall be distributed, among all the shareholders, proportionate to the nominal value of their shares without taking into account any premium paid on them. |
(b) | For the purpose of Article 65(a) above, a person who is entitled to shares but have not yet been allocated the shares, shall be considered as if the shares to which he is entitled were allocated to him prior to the dissolution, and that the amount paid on account of the nominal value of the shares has been paid up. In this case one who is entitled to the shares, is entitled to payment of an equal sum to the amount that he would have received in a dissolution if he would have held the shares of the Company on the eve of the adoption of the resolution of the dissolution, with a deduction of the price of the exercise that he would have had to pay if he would have exercised his right to the shares of the Company on the eve of the resolution of the dissolution. |
(c) | If the Company winds up and the property of the Company that is to be distributed among the members is not enough to return all the paid-up capital, these assets will be distributed inasmuch as possible in a proportionate manner to the paid-up capital, or which is considered paid up at the start of the dissolution, of the shares held by each of the members. |
37
Exhibit 4.15
DESCRIPTION OF THE REGISTRANT’S SECURITIES
REGISTERED PURSUANT TO SECTION 12 OF THE
SECURITIES EXCHANGE ACT OF 1934, AS AMENDED
As of the date of this Annual Report on Form 10-K, Quoin Pharmaceuticals, Ltd., an Israeli company (“we,” “our” and the “Company”) has one class of securities registered under Section 12 of the Securities Exchange Act of 1934, as amended: American Depositary Shares, each representing one (1) ordinary share, no par value per share. The following description of such securities is intended as a summary of the terms of such securities as currently in effect and is qualified in its entirety by the provisions of our amended and restated articles of association, a copy of which is filed as an exhibit to this Annual Report on Form 10-K and is incorporated by reference herein. This description contains all material information concerning such securities but does not purport to be complete. We encourage you to read our amended and restated articles of association and the applicable provisions of the Israeli Companies Law, 5759-1999 (the “Companies Law”), for additional information.
DESCRIPTION OF ORDINARY SHARES
Ordinary Shares
As of December 31, 2024, our authorized share capital consisted of 100,000,000 ordinary shares, no par value. As of December 31, 2024, there were 8,948,164 ordinary shares, no par value per share, outstanding and 8,948,164 ADSs outstanding (assuming all ordinary shares are represented by ADSs), with each ADS representing one (1) ordinary share. Our ordinary shares are not redeemable and do not have any preemptive rights.
Articles of Association
The following are summaries of material provisions of our articles of association and the Israeli Companies Law, as amended (the “Companies Law”), insofar as they relate to the material terms of our ordinary shares.
Purposes and Objects of the Company
Our purpose is set forth in Section 2 of our articles of association and includes every lawful purpose.
Registration Number
Our number with the Israeli Registrar of Companies is 520036484.
Voting Rights
Holders of our ordinary shares have one vote for each ordinary share held on all matters submitted to a vote of shareholders at a shareholders meeting. Shareholders may vote at shareholders meetings either in person, by proxy or by written ballot. Israeli law does not allow public companies to adopt shareholder resolutions by means of written consent in lieu of a shareholders meeting. The board of directors shall determine and provide a record date for each shareholders meeting and all shareholders at such record date may vote. Unless stipulated differently in the Companies Law or in the articles of association, all shareholders’ resolutions shall be approved by a simple majority vote. As a general rule, an amendment to our articles of association requires the prior approval of a simple majority of our shares represented and voting at a general meeting.
Transfer of Shares
Our ordinary shares that are fully paid for are issued in registered form and may be freely transferred under our articles of association, unless the transfer is restricted or prohibited by applicable law or the rules of a stock exchange on which the shares are traded. The ownership or voting of our ordinary shares by non-residents of Israel is not restricted in any way by our articles of association or Israeli law, except for ownership by nationals of some countries that are, or have been, in a state of war with Israel.
Amendment of Share Capital
Our articles of association enable us to increase or reduce our share capital. Any such changes are subject to the provisions of the Companies Law and must be approved by a resolution duly passed by our shareholders at an annual or special general meeting by voting on such change in the capital. In addition, transactions that have the effect of reducing capital, such as the declaration and payment of dividends in the absence of sufficient retained earnings and profits, or an issuance of shares for less than their nominal value (which would be applicable to our company should our articles be changed so as to permit the issue of shares having a nominal value, however our shares currently have no nominal value), require a resolution of our board of directors and court approval.
Dividends and liquidation rights
We may declare a dividend to be paid to the holders of our ordinary shares in proportion to their respective shareholdings. Under the Companies Law, dividend distributions are determined by the board of directors and do not require the approval of the shareholders of a company unless the company’s articles of association provide otherwise. Our amended and restated articles of association do not require shareholder approval of a dividend distribution and provide that dividend distributions may be determined by our board of directors.
Pursuant to the Companies Law, the distribution amount is limited to the greater of retained earnings or earnings generated over the previous two years, according to our then last reviewed or audited financial statements (less the amount of previously distributed dividends, if not reduced from the earnings), provided that the end of the period to which the financial statements relate is not more than six months prior to the date of the distribution. If we do not meet such criteria, then we may distribute dividends only with court approval; as a company listed on an exchange outside of Israel, however, court approval is not required if the proposed distribution is in the form of an equity repurchase, provided that we notify our creditors of the proposed equity repurchase and allow such creditors an opportunity to initiate a court proceeding to review the repurchase. If within 30 days such creditors do not file an objection, then we may proceed with the repurchase without obtaining court approval. In each case, we are only permitted to distribute a dividend if our board of directors and, if applicable, the court determines that there is no reasonable concern that payment of the dividend will prevent us from satisfying our existing and foreseeable obligations as they become due.
In the event of our liquidation, after satisfaction of liabilities to creditors, our assets will be distributed to the holders of our ordinary shares in proportion to their shareholdings. This right, as well as the right to receive dividends, may be affected by the grant of preferential dividend or distribution rights to the holders of a class of shares with preferential rights that may be authorized in the future.
Exchange Controls
There are currently no Israeli currency control restrictions on payments of dividends or other distributions with respect to our ordinary shares or the proceeds from the sale of the shares, except, under certain circumstances, for shareholders who are subjects of countries that are, or have been, in a state of war with Israel. Israeli residents have an obligation to file reports with the Bank of Israel regarding certain transactions. However, legislation remains in effect pursuant to which currency controls can be imposed by administrative action at any time.
Shareholders Meetings
Under Israeli law, we are required to hold an annual general meeting of our shareholders once every calendar year and in any event no later than 15 months after the date of the previous annual general meeting. All meetings other than the annual general meeting of shareholders are referred to as special meetings. Our board of directors may call special meetings whenever it sees fit, at such time and place, within or outside of Israel, as it may determine. In addition, the Companies Law and our articles of association provide that our board of directors is required to convene a special meeting upon the written request of (1) any two or more of our directors, (2) one quarter of the directors then in office; or (3) as a company listed on an exchange in the U.S., one or more shareholders holding, in the aggregate either (a) 10% or more of our issued and outstanding share capital and 1% of our outstanding voting rights, or (b) 10% or more of our outstanding voting rights. Under Israeli law, one or more shareholders holding at
least 1% of the voting rights at the general meeting of the shareholders may request that the board of directors include a matter in the agenda of a general meeting of the shareholders to be convened in the future, provided that it is appropriate to discuss such a matter at the general meeting. Notwithstanding the foregoing, as a company listed on an exchange outside of Israel, a matter relating to the appointment or removal of a director may only be requested by one or more shareholders holding at least 5% of the voting rights at the general meeting of the shareholders.
Subject to the provisions of the Companies Law and the regulations promulgated thereunder, shareholders entitled to participate and vote at general meetings of a company are the shareholders of record on a date to be decided by the board of directors which for us, as a company listed on an exchange outside Israel, may be between four and sixty days prior to the date of the meeting.
The Companies Law and our articles of association require that resolutions regarding the following matters must be passed at a general meeting of our shareholders:
| ● | amendments to our articles of association; |
| ● | appointment, terms of service or termination of service of our auditors; |
| ● | appointment and dismissal of external directors, if and to the extent any are required to be appointed under the Companies Law; |
| ● | approval of acts and transactions requiring general meeting approval pursuant to the Companies Law; |
| ● | increases or reductions of our authorized share capital; |
| ● | a merger; and |
| ● | the exercise of our board of directors’ powers by a general meeting, if our board of directors is unable to exercise its powers and the exercise of any of its powers is required for our proper management. |
The Companies Law requires that a notice of any annual general meeting or special general meeting be provided to shareholders at least 21 days prior to the meeting and if the agenda of the meeting includes, among other things, the appointment or removal of directors, the approval of transactions with office holders or interested or related parties, or an approval of a merger, notice must be provided at least 35 days prior to the meeting. Under the Companies Law and our amended and restated articles of association, shareholders are not permitted to take action by way of written consent in lieu of a meeting.
Quorum
The quorum required for our general meetings of shareholders consists of at least two shareholders present in person or by proxy who hold or represent between them at least 331∕3% of the total outstanding voting rights, provided, however, that with respect to any general meeting that was convened pursuant to a resolution adopted by the board of directors and which at the time of such general meeting we qualify to use the forms and rules of a “foreign private issuer,” the requisite quorum shall consist of two or more shareholders present in person or by proxy who hold or represent between them at least 25% of the total outstanding voting rights. A meeting adjourned for lack of a quorum will be adjourned for one week, to the same day in the following week and at the same time and place, or to a later date if so specified in the notice of the meeting, or to another day or place determined by our board of directors in a notice to shareholders. At the reconvened meeting, if a quorum is not present within half an hour from the scheduled time, any number of our shareholders present in person or by proxy shall constitute a lawful quorum.
Vote Requirements
Our articles of association provide that all resolutions of our shareholders require a simple majority vote, unless otherwise required by the Companies Law or by our articles of association. Under the Companies Law, certain actions require the approval of a special majority, including: (i) an extraordinary transaction with a controlling shareholder or in which the controlling shareholder has a personal interest, (ii) the terms of employment or other engagement of a controlling shareholder of the company or a controlling shareholder’s relative (even if such terms are not extraordinary) and (iii) certain compensation-related matters described above under “Management—Compensation Committee—Compensation Policy under the Companies Law.” Under our articles of association, the alteration of the rights, privileges, preferences or obligations of any class of our shares (to the extent there are classes other than ordinary shares) requires the approval of a simple majority of the class so affected (or such other percentage of the relevant class that may be set forth in the governing documents relevant to such class), in addition to a majority of all classes of shares voting together as a single class at a shareholder meeting.
Dissolution
Generally under Israeli law, a resolution for the voluntary winding up of a company requires the approval of holders of 75% of the voting rights represented at the meeting, in person, by proxy or by written ballot and voting on the resolution.
In the event of our liquidation, after satisfaction of liabilities to creditors, our assets will be distributed to the holders of our ordinary shares (including holders of entitlements to shares, after deducting the nominal value (if any) of such shares and the price which would have been paid in order to exercise the right to such shares), in proportion to their shareholdings. This right, as well as the right to receive dividends, may be affected by the grant of preferential dividend or distribution rights to the holders of a class of shares with preferential rights that may be authorized in the future.
Access to Corporate Records
Under the Companies Law, all shareholders of a company generally have the right to review minutes of the company’s general meetings, its register of shareholders and material shareholders, articles of association, financial statements and any document it is required by law to file publicly with the Israeli Companies Registrar. Any of our shareholders may request to review any document in our possession that relates to any action or transaction with a related party, interested party, or office holder that requires shareholder approval under the Companies Law. We may deny a request to review a document if we determine that the request was not made in good faith, that the document contains a trade secret or patent, or that the document’s disclosure may otherwise prejudice our interests.
Acquisitions under Israeli Law
Full Tender Offer
A person wishing to acquire shares of a public Israeli company, and who would as a result hold over 90% of the target company’s issued and outstanding share capital, is required by the Companies Law to make a tender offer to all of the company’s shareholders for the purchase of all of the issued and outstanding shares of the company. A person wishing to acquire shares of a public Israeli company, and who would as a result hold over 90% of the issued and outstanding share capital of a certain class of shares, is required to make a tender offer to all of the shareholders who hold shares of that class for the purchase of all of the issued and outstanding shares of that class. If the shareholders who do not accept the offer hold less than 5% of the issued and outstanding share capital of the company or of the applicable class, all of the shares that the acquirer offered to purchase will be transferred to the acquirer by operation of law, provided that a majority of the offerees that do not have a personal interest in such tender offer, have accepted the tender offer. Alternatively, if shareholders who do not accept the tender offer represent less than 2% of the company’s issued and outstanding share capital (or less than 2% of the applicable class of shares), approval by a majority of the offerees that do not have a personal interest in such tender offer is not required to complete the tender offer. A shareholder whose shares are so transferred may petition the court regarding the fair value to be paid in consideration of such shares, within six months from the date of acceptance of the full tender offer; this right of petition applies to all offeree shareholders, unless the acquirer stipulated in the tender offer that a shareholder accepting the offer may not seek appraisal rights, and prior to the acceptance of the full tender offer, the acquirer and the company disclosed the information required by law in connection with a full tender offer. To the extent a court so
petitioned determines that the offered value was less than the fair value per share, the court may order the difference to be paid.
Special Tender Offer
The Companies Law provides that an acquisition of shares of an Israeli public company must be made by means of a “special tender offer” complying with the relevant provisions of the Companies Law if, as a result of the acquisition, the purchaser would become a holder of 25% or more of the voting rights in the company, if there did not previously exist a holder of 25% or more of the voting rights in the company, or if, as a result of the acquisition, the purchaser would become a holder of more than 45% of the voting rights in the company, if there did not previously exist a holder of more than 45% of the voting rights in the company. This requirement does not apply if the acquisition: (a) occurs in the context of a private placement by the company that received shareholder approval as a private placement giving the offeree 25% or 45% of the company’s voting rights (as the case may be); (b) is from a holder of 25% or more of the voting rights in the company and results in the acquirer becoming a holder of 25% or more of the voting rights in the company; or (c) is from a holder of more than 45% of the voting rights in the company and results in the acquirer becoming a holder of more than 45% of the voting rights in the company.
In the event that a special tender offer is made, the target company’s board of directors is required to express its opinion on the advisability of the offer, or may abstain from expressing any opinion if it is unable to do so, provided that it gives the reasons for its abstention.
A special tender offer must be directed to all offerees, and the offerees may give notice of their agreement or opposition to the special tender offer. The special tender offer will be consummated only if: (a) at least 5% of the voting rights attached to the company’s outstanding shares will be acquired by the offeror, and (b) among those shareholders who gave notice of their position (excluding any controlling shareholders of the offeror, holders of 25% or more of the voting rights in the target company, and any person having a personal interest in the acceptance of the tender offer, including relatives or corporations under the control of any of the above), the number of shares whose holders agreed to the offer exceeds the number of shares whose holders objected to the offer.
If a special tender offer is accepted by the procedure described above, then shareholders who did not respond to or who objected the offer may accept the offer within four days of the last day set for the acceptance of the offer.
An office holder in a company which is the target of a special tender offer who, in his or her capacity as an office holder, performs an act or omits to act for in order to cause the failure of an existing or foreseeable special tender offer, or to impair the likelihood of its acceptance, is liable to the offeror and offerees for damages, unless such office holder acted in good faith and had reasonable grounds to believe that such act or omission was beneficial to the company. As a safe harbor, office holders of the target company may negotiate with a potential purchaser in order to improve the terms of a special tender offer, or negotiate with third parties in order to obtain a competing offer.
In the event that a special tender offer is accepted, the purchaser, any person or entity controlling or controlled by the purchaser, or under common control with the purchaser, may not make a subsequent tender offer for the purchase of shares of the target company, and may not enter into a merger with the target company, for a period of one year from the date of the offer, unless the purchaser or such person or entity undertakes to effect such an offer or merger as a special tender offer in compliance with the Companies Law requirements.
Merger
The Companies Law permits merger transactions if approved by each party’s board of directors and, unless certain conditions described under the Companies Law are met, by each party’s shareholders by a majority vote as described below.
For purposes of the shareholder vote, unless a court rules otherwise, the merger will not be deemed approved if a majority of the shares voted at the shareholders meeting held by shareholders who are not the other party to the merger, or held by any person who holds 25% or more of the outstanding shares or the right to appoint 25% or more of the directors of the other party to the merger (including relatives or
entities in control of the above), vote against the merger. If the transaction would have been approved but for the separate approval of each class or the exclusion of the votes of certain shareholders as provided above, a court may still approve the merger upon the request of holders of at least 25% of the voting rights of a company, if the court holds that the merger is fair and reasonable, taking into account the relative value of the merger parties and the consideration offered to the shareholders. If the non-surviving entity of the merger has more than one class of shares, the merger must be approved by each class of shareholders. If a merger is with a company’s controlling shareholder, or if a controlling shareholder has a personal interest in the merger, then the merger will be subject to the special majority approval required for an extraordinary transaction with a controlling shareholder (see: Approval of Related Party Transactions under Israeli Law - Declaration of Personal Interest of Controlling Shareholders and Approval of Certain Transactions). In the context of mergers (as well as other related party transactions), a “controlling shareholder” under Israeli law is deemed to include any shareholder holding 25% or more of the voting rights in the company if no other shareholder owns more than 50% of the voting rights in the company, and two or more shareholders with a personal interest in the approval of the same transaction are deemed to be one shareholder for such purpose.
The Companies Law requires the board of directors of a merging company to discuss and determine whether, in its view, there exists a reasonable concern that as a result of the proposed merger, the surviving company will not be able to satisfy its obligations towards its creditors, and if not, the board of directors may not approve the merger. The Companies Law requires each merging company to inform its secured creditors of the proposed merger plan. Upon the request of a creditor of either party to the proposed merger, the court may delay or prevent the merger if it concludes that there exists a reasonable concern that, as a result of the merger, the surviving company will be unable to satisfy the obligations of any of the parties to the merger, and may further give instructions to secure the rights of creditors.
A merger may not be completed unless at least 50 days have passed from the date that a proposal for approval of the merger is filed with the Israeli Registrar of Companies, and 30 days have passed from the date the merger was approved by the shareholders of each merging company.
Antitakeover Measures
The Companies Law allows us to create and issue shares having rights different from those attached to our ordinary shares, including shares providing certain preferred rights, distributions or other matters, and shares having preemptive rights. As of the date of this Annual Report on Form 10-K, we do not have any authorized or issued classes of shares other than our ordinary shares. In the future, if we do create and issue a class of shares other than ordinary shares, such class of shares, depending on the specific rights that may be attached to them, may delay or prevent a takeover or otherwise prevent our shareholders from realizing a potential premium over the market value of their ordinary shares. The authorization of a new class of shares will require an amendment to our articles of association which requires the prior approval of the holders of a majority of our shares at a general meeting. Shareholders voting in such meeting will be subject to the restrictions provided in the Companies Law as described above.
DESCRIPTION OF AMERICAN DEPOSITARY SHARES
The Bank of New York Mellon (the “Depositary”), as depositary, has registered and delivered American Depositary Shares, also referred to as ADSs. Each ADS represents one (1) ordinary share (or a right to receive one (1) ordinary shares) deposited with The Bank of New York Mellon in Manchester, United Kingdom, as custodian for the Depositary. The Depositary’s corporate trust office at which the ADSs will be administered is located at 240 Greenwich Street, New York, New York 10286. The Bank of New York Mellon’s principal executive office is located at 240 Greenwich Street, New York, New York 10286.
ADSs may be held either (a) directly (1) by having an American Depositary Receipt, also referred to as an ADR, which is a certificate evidencing a specific number of ADSs or (2) by having uncertificated ADSs, or (b) indirectly by holding a security entitlement in ADSs through a broker or other financial institution that is a direct or indirect participant in The Depository Trust Company, also called DTC. If ADSs are held directly by the holder, then that holder is registered as such, and is referred to in our description here an ADS holder. An indirect holder of ADSs indirectly must rely on the procedures of
the holder’s broker or other financial institution to assert the rights of ADS holder described in this Exhibit.
Registered holders of uncertificated ADSs will receive statements from the depositary confirming their holdings.
We will not treat registered ADS holders as one of our shareholders, and they will not have shareholder rights. Israeli law governs shareholder rights. The depositary will be the holder of the ordinary shares underlying ADSs. A registered holder of ADSs will have ADS holder rights. A deposit agreement among us, the depositary, ADS holders and all other persons indirectly or beneficially holding ADSs sets out ADS holder rights as well as the rights and obligations of the depositary. New York law governs the deposit agreement and the ADSs.
The following is a summary of the material provisions of the deposit agreement. For more complete information, you should read the entire deposit agreement and the form of ADR.
Dividends and Other Distributions
How will you receive dividends and other distributions on the shares?
The depositary has agreed to pay or distribute to ADS holders the cash dividends or other distributions it or the custodian receives on ordinary shares or other deposited securities, upon payment or deduction of its fees and expenses. You will receive these distributions in proportion to the number of ordinary shares your ADSs represent.
Cash. The depositary will convert any cash dividend or other cash distribution we pay in non-U.S. currency on the ordinary shares into U.S. dollars, if it can do so on a reasonable basis and can transfer the U.S. dollars to the United States. If that is not possible or if any government approval is needed and cannot be obtained, the deposit agreement allows the depositary to distribute the foreign currency only to those ADS holders to whom it is possible to do so. It will hold the foreign currency it cannot convert for the account of the ADS holders who have not been paid. It will not invest the foreign currency, and it will not be liable for any interest.
Before making a distribution, the depositary will deduct any withholding taxes, or other required governmental charges. The depositary will distribute only whole U.S. dollars and cents and will round fractional cents to the nearest whole cent. If the exchange rates fluctuate during a time when the depositary cannot convert the foreign currency, you may lose some or all of the value of the distribution.
Shares. The depositary may distribute additional ADSs representing any ordinary shares we distribute as a dividend or free distribution. The depositary will only distribute whole ADSs. It will sell ordinary shares which would require it to deliver a fraction of an ADS (or ADSs representing those shares) and distribute the net proceeds in the same way as it does with cash. If the depositary does not distribute additional ADSs, the outstanding ADSs will also represent the new shares. The depositary may sell a portion of the distributed ordinary shares (or ADSs representing those shares) sufficient to pay its fees and expenses in connection with that distribution.
Rights to purchase additional shares. If we offer holders of our securities any rights to subscribe for additional ordinary shares or any other rights, the depositary may (1) exercise those rights on behalf of ADS holders, (2) distribute those rights to ADS holders or (3) sell those rights and distribute the net proceeds to ADS holders, in each case after deduction or upon payment of its fees and expenses. To the extent the depositary does not do any of those things, it will allow the rights to lapse. In that case, you will receive no value for them. The depositary will exercise or distribute rights only if we ask it to and provide satisfactory assurances to the depositary that it is legal to do so. If the depositary will exercise rights, it will purchase the securities to which the rights relate and distribute those securities or, in the case of ordinary shares, new ADSs representing the new ordinary shares, to subscribing ADS holders, but only if ADS holders have paid the exercise price to the depositary. U.S. securities laws may restrict the ability of the depositary to distribute rights or ADSs or other securities issued on exercise of rights to all or certain ADS holders, and the securities distributed may be subject to restrictions on transfer.
Other Distributions. The depositary will send to ADS holders anything else we distribute on deposited securities by any means it thinks is legal, fair and practical. If it cannot make the distribution in that way, the depositary has a choice. It may decide to sell what we distributed and distribute the net proceeds, in the same way as it does with non-U.S. currency. Alternatively, it may decide to hold what we distributed, in which case ADSs will also represent the newly distributed property. However, the depositary is not required to distribute any securities (other than ADSs) to ADS holders unless it receives satisfactory evidence from us that it is legal to make that distribution. The depositary may sell a portion of the distributed securities or property sufficient to pay its fees and expenses in connection with that distribution. U.S. securities laws may restrict the ability of the depositary to distribute securities to all or certain ADS holders, and the securities distributed may be subject to restrictions on transfer.
The depositary is not responsible if it decides that it is unlawful or impractical to make a distribution available to any ADS holders. We have no obligation to register ADSs, shares, rights or other securities under the Securities Act. We also have no obligation to take any other action to permit the distribution of ADSs, shares, rights or anything else to ADS holders. This means that you may not receive the distributions we make on our ordinary shares or any value for them if it is illegal or impractical for us to make them available to you.
Deposit, Withdrawal and Cancellation
How are ADSs issued?
The depositary will deliver ADSs upon deposits of ordinary shares or evidence of rights to receive ordinary shares with the custodian. Upon payment of its fees and expenses and of any taxes or charges, such as stamp taxes or stock transfer taxes or fees, the depositary will register the appropriate number of ADSs and will deliver the ADSs to or upon the order of the person or persons that made the deposit.
How can ADS holders withdraw the deposited securities?
ADS holders may surrender ADSs for the purpose of withdrawal at the Depositary’s account at DTCC (BNYM’s DTC participant #2504). Upon payment of its cancellation fees and expenses and of any taxes or charges, such as stamp taxes or stock transfer taxes or fees, the depositary will deliver the ordinary shares and any other deposited securities underlying the ADSs to the ADS holder or a person the ADS holder designates in accordance with the Cancellation Instruction provided to The Bank of New York Mellon.
How do ADS holders interchange between certificated ADSs and uncertificated ADSs?
ADS holders may surrender ADS to the depositary for the purpose of exchanging ADS for uncertificated ADSs. The depositary will cancel that ADS and will send to the ADS holder a statement confirming that the ADS holder is the registered holder of uncertificated ADSs. Upon receipt by the depositary of a proper instruction from a registered holder of uncertificated ADSs requesting the exchange of uncertificated ADSs for certificated ADSs, the depositary will execute and deliver to the ADS holder an ADS evidencing those ADSs.
Voting Rights
ADS holders may instruct the depositary how to vote the number of deposited ordinary shares their ADSs represent. If we request the depositary to solicit your voting instructions (and we are not required to do so), the depositary will notify you of a shareholders’ meeting and send or make voting materials available to you. Those materials will describe the matters to be voted on and explain how ADS holders may instruct the depositary how to vote. For instructions to be valid, they must reach the depositary by a date set by the depositary.
The depositary will try, as far as practical, subject to the laws of Israel and the provisions of our articles of association or similar documents, to vote or to have its agents vote the ordinary shares or other deposited securities as instructed by ADS holders. If we do not request the depositary to solicit your voting instructions, you can still send voting instructions, and, in that case, the depositary may try to vote as you instruct, but it is not required to do so.
Except by instructing the depositary as described above, ADS holders will not be able to exercise voting rights, unless they surrender your ADSs and withdraw the ordinary shares. However, ADS holders may not know about the meeting sufficiently in advance to withdraw the ordinary shares. In any event, the depositary will not exercise any discretion in voting deposited securities and it will only vote or attempt to vote as instructed.
We cannot assure that ADS holders will receive the voting materials in time to ensure that they can instruct the depositary to vote ordinary shares. In addition, the depositary and its agents are not responsible for failing to carry out voting instructions or for the manner of carrying out voting instructions. This means that ADS holders may not be able to exercise voting rights and there may be nothing they can do if your ordinary shares are not voted as requested.
In order to give ADS holders a reasonable opportunity to instruct the depositary as to the exercise of voting rights relating to deposited securities, if we request the Depositary to act, we agree to give the depositary notice of any such meeting and details concerning the matters to be voted upon at least thirty days in advance of the meeting date.
Fees and Expenses
Persons depositing or withdrawing shares or ADS holders must pay: |
| For: |
$5.00 (or less) per 5,000 ADSs (or portion of 5,000 ADSs) | | Issuance of ADSs, including issuances resulting from a distribution of ordinary shares or rights or other property Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates |
$0.05 (or less) per ADS | | Any cash distribution to ADS holders |
A fee equivalent to the fee that would be payable if securities distributed to you had been ordinary shares and the ordinary shares had been deposited for issuance of ADSs | | Distribution of securities distributed to holders of deposited securities (including rights) that are distributed by the depositary to ADS holders |
$0.05 (or less) per ADSs per calendar year | | Depositary services |
Registration or transfer fees | | Transfer and registration of ordinary shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw ordinary shares |
Expenses of the Depositary | | Cable, telex and facsimile transmissions (when expressly provided in the deposit agreement); converting foreign currency to U.S. dollars |
Taxes and other governmental charges the Depositary or the custodian have to pay on any ADS or share underlying an ADS, for example, stock transfer taxes, stamp duty or withholding taxes | | As necessary |
Any charges incurred by the Depositary or its agents for servicing the deposited securities | | As necessary |
The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing ordinary shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may collect any of its fees by deduction from any cash distribution payable (or by selling a portion of securities or other property distributable) to ADS holders that are obligated to pay those fees. The depositary may generally refuse to provide fee-attracting services until its fees for those services are paid.
From time to time, the depositary may make payments to us to reimburse us for costs and expenses generally arising out of establishment and maintenance of the ADS program, waive fees and expenses for services provided to us by the depositary or share revenue from the fees collected from ADS holders. In performing its duties under the deposit agreement, the depositary may use brokers, dealers, foreign currency dealers or other service providers that are owned by or affiliated with the depositary and that may earn or share fees, spreads or commissions.
The depositary may convert currency itself or through any of its affiliates and, in those cases, acts as principal for its own account and not as agent, advisor, broker or fiduciary on behalf of any other person and earns revenue, including, without limitation, transaction spreads, that it will retain for its own account. The revenue is based on, among other things, the difference between the exchange rate assigned to the currency conversion made under the deposit agreement and the rate that the depositary or its affiliate receives when buying or selling foreign currency for its own account. The depositary makes no representation that the exchange rate used or obtained in any currency conversion under the deposit agreement will be the most favorable rate that could be obtained at the time or that the method by which that rate will be determined will be the most favorable to ADS holders, subject to the depositary’s obligations under the deposit agreement. The methodology used to determine exchange rates used in currency conversions is available upon request.
Payment of Taxes
ADS holders are responsible for any taxes or other governmental charges payable on their ADSs or on the deposited securities represented by any of their ADSs. The depositary may refuse to register any transfer of ADSs or allow a withdrawal of the deposited securities represented by your ADSs, until such taxes or other charges are paid. It may apply payments owed to the ADS holder or sell deposited securities represented by the ADSs to pay any taxes owed and the ADS holder will remain liable for any deficiency. If the depositary sells deposited securities, it will, if appropriate, reduce the number of ADSs to reflect the sale and pay to ADS holders any proceeds, or send to ADS holders any property, remaining after it has paid the taxes.
Tender and Exchange Offers; Redemption, Replacement or Cancellation of Deposited Securities
The depositary will not tender deposited securities in any voluntary tender or exchange offer unless instructed to do by an ADS holder surrendering ADSs and subject to any conditions or procedures the depositary may establish.
If deposited securities are redeemed for cash in a transaction that is mandatory for the depositary as a holder of deposited securities, the depositary will call for surrender of a corresponding number of ADSs and distribute the net redemption money to the holders of called ADSs upon surrender of those ADSs.
If there is any change in the deposited securities such as a sub-division, combination or other reclassification, or any merger, consolidation, recapitalization or reorganization affecting the issuer of deposited securities in which the depositary receives new securities in exchange for or in lieu of the old deposited securities, the depositary will hold those replacement securities as deposited securities under the deposit agreement. However, if the depositary decides it would not be lawful and to hold the replacement securities because those securities could not be distributed to ADS holders or for any other reason, the depositary may instead sell the replacement securities and distribute the net proceeds upon surrender of the ADSs.
If there is a replacement of the deposited securities and the depositary will continue to hold the replacement securities, the depositary may distribute new ADSs representing the new deposited securities or ask you to surrender your outstanding ADRs in exchange for new ADSs identifying the new deposited securities.
If there are no deposited securities underlying ADSs, including if the deposited securities are cancelled, or if the deposited securities underlying ADSs have become apparently worthless, the depositary may call for surrender or of those ADSs or cancel those ADSs upon notice to the ADS holders.
Amendment and Termination
How may the deposit agreement be amended?
We may agree with the depositary to amend the deposit agreement and the ADSs without consent of the ADS holders for any reason. If an amendment adds or increases fees or charges, except for taxes and other governmental charges or expenses of the depositary for registration fees, facsimile costs, delivery charges or similar items, or prejudices a substantial right of ADS holders, it will not become effective for outstanding ADSs until 30 days after the depositary notifies ADS holders of the amendment. At the
time an amendment becomes effective, ADS holders are considered, by continuing to hold your ADSs, to agree to the amendment and to be bound by the ADRs and the deposit agreement as amended.
How may the deposit agreement be terminated?
The depositary will initiate termination of the deposit agreement if we instruct it to do so. The depositary may initiate termination of the deposit agreement if
| ● | 60 days have passed since the depositary told us it wants to resign but a successor depositary has not been appointed and accepted its appointment; |
| ● | we delist our ordinary shares from an exchange on which they were listed and do not list the ordinary shares on another exchange; |
| ● | we appear to be insolvent or enter insolvency proceedings all or substantially all the value of the deposited securities has been distributed either in cash or in the form of securities; |
| ● | there are no deposited securities underlying the ADSs or the underlying deposited securities have become apparently worthless; or |
| ● | there has been a replacement of deposited securities. |
If the deposit agreement will terminate, the depositary will notify ADS holders at least 90 days before the termination date. At any time after the termination date, the depositary may sell the deposited securities. After that, the depositary will hold the money it received on the sale, as well as any other cash it is holding under the deposit agreement, unsegregated and without liability for interest, for the pro rata benefit of the ADS holders that have not surrendered their ADSs. Normally, the depositary will sell as soon as practicable after the termination date.
After the termination date and before the depositary sells, ADS holders can still surrender their ADSs and receive delivery of deposited securities, except that the depositary may refuse to accept a surrender for the purpose of withdrawing deposited securities if it would interfere with the selling process. The depositary may refuse to accept a surrender for the purpose of withdrawing sale proceeds until all the deposited securities have been sold. The depositary will continue to collect distributions on deposited securities, but, after the termination date, the depositary is not required to register any transfer of ADSs or distribute any dividends or other distributions on deposited securities to the ADSs holder (until they surrender their ADSs) or give any notices or perform any other duties under the deposit agreement except as described in this paragraph.
Limitations on Obligations and Liability
Limits on our Obligations and the Obligations of the Depositary; Limits on Liability to Holders of ADSs
The deposit agreement expressly limits our obligations and the obligations of the depositary. It also limits our liability and the liability of the depositary. We and the depositary:
| ● | are only obligated to take the actions specifically set forth in the deposit agreement without negligence or bad faith; |
| ● | are not liable if we are or it is prevented or delayed by law or circumstances beyond our or its control from performing our or its obligations under the deposit agreement; |
| ● | are not liable if we or it exercises discretion permitted under the deposit agreement; |
| ● | are not liable for the inability of any holder of ADSs to benefit from any distribution on deposited securities that is not made available to holders of ADSs under the terms of the deposit |
agreement, or for any special, consequential or punitive damages for any breach of the terms of the deposit agreement;
| ● | have no obligation to become involved in a lawsuit or other proceeding related to the ADSs or the deposit agreement on your behalf or on behalf of any other person; |
| ● | are not liable for the acts or omissions of any securities depository, clearing agency or settlement system; and |
| ● | may rely upon any documents we believe or it believes in good faith to be genuine and to have been signed or presented by the proper person. |
In the deposit agreement, we and the depositary agree to indemnify each other under certain circumstances.
Requirements for Depositary Actions
Before the depositary will deliver or register a transfer of ADSs, make a distribution on ADSs, or permit withdrawal of shares, the depositary may require:
| ● | payment of stock transfer or other taxes or other governmental charges and transfer or registration fees charged by third parties for the transfer of any ordinary shares or other deposited securities; |
| ● | satisfactory proof of the identity and genuineness of any signature or other information it deems necessary; and |
| ● | compliance with regulations it may establish, from time to time, consistent with the deposit agreement, including presentation of transfer documents. |
The depositary may refuse to deliver ADSs or register transfers of ADSs when the transfer books of the depositary or our transfer books are closed or at any time if the depositary or we think it advisable to do so.
Right to Receive the Ordinary Shares Underlying ADSs
ADS holders have the right to cancel their ADSs and withdraw the underlying ordinary shares at any time except:
| ● | when temporary delays arise because: (1) the depositary has closed its transfer books or we have closed our transfer books; (2) the transfer of ordinary shares is blocked to permit voting at a shareholders meeting; or (3) we are paying a dividend on our shares; |
| ● | when you owe money to pay fees, taxes and similar charges; or |
| ● | when it is necessary to prohibit withdrawals in order to comply with any laws or governmental regulations that apply to ADSs or to the withdrawal of ordinary shares or other deposited securities. |
This right of withdrawal may not be limited by any other provision of the deposit agreement.
Pre-release of ADSs
The deposit agreement permits the depositary to deliver ADSs before deposit of the underlying shares. This is called a pre-release of the ADSs. The depositary may also deliver ordinary shares upon cancellation of pre-released ADSs (even if the ADSs are canceled before the pre-release transaction has been closed out). A pre-release is closed out as soon as the underlying ordinary shares are delivered to the depositary. The depositary may receive ADSs instead of ordinary shares to close out a pre-release. The depositary may pre-release ADSs only under the following conditions: (1) before or at the time of
the pre-release, the person to whom the pre-release is being made represents to the depositary in writing that it or its customer owns the ordinary shares or ADSs to be deposited; (2) the pre-release is fully collateralized with cash or other collateral that the depositary considers appropriate; and (3) the depositary must be able to close out the pre-release on not more than five business days’ notice. In addition, the depositary will limit the number of ADSs that may be outstanding at any time as a result of pre-release, although the depositary may disregard the limit from time to time if it thinks it is appropriate to do so.
Direct Registration System
In the deposit agreement, all parties to the deposit agreement acknowledge that the Direct Registration System, or DRS, and Profile Modification System, or Profile, will apply to the ADSs. DRS is a system administered by DTC that facilitates interchange between registered holdings of uncertificated ADSs and holdings of security entitlements in ADSs through DTC and a DTC participant. Profile is a feature of DRS that allows a DTC participant, claiming to act on behalf of a registered holder of ADSs, to direct the depositary to register a transfer of those ADSs to DTC or its nominee and to deliver those ADSs to the DTC account of that DTC participant without receipt by the depositary of prior authorization from the ADS holder to register that transfer.
In connection with and in accordance with the arrangements and procedures relating to DRS/Profile, the parties to the deposit agreement understand that the depositary will not determine whether the DTC participant that is claiming to be acting on behalf of an ADS holder in requesting registration of transfer and delivery as described in the paragraph above has the actual authority to act on behalf of the ADS holder (notwithstanding any requirements under the Uniform Commercial Code). In the deposit agreement, the parties agree that the depositary’s reliance on and compliance with instructions received by the depositary through the DRS/Profile system and in accordance with the deposit agreement will not constitute negligence or bad faith on the part of the depositary.
Shareholder communications; inspection of register of holders of ADSs
The depositary will make available for your inspection at its office all communications from us that we make generally available to holders of deposited securities. The depositary will send you copies of those communications or otherwise make those communications available to you upon our request. You have a right to inspect the register of holders of ADSs, but not for the purpose of contacting those holders about a matter unrelated to our business or the ADSs.
Exhibit 10.44
Non-Employee Directors’ Compensation Program, as amended
Each non-employee director of Quoin Pharmaceuticals Ltd. is entitled to receive as compensation for their service (i) an annual cash retainer of $82,500, (ii) an annual option grant having a value of no less than $20,000 and no more than $60,000, with such value being determined annually at the discretion of the Compensation Committee and the Board, (iii) each committee chairperson shall receive an additional $15,000 cash per year for service as the chairperson of a board committee; and (iv) each standing committee member shall receive $5,000 per year for service on each applicable committee. In addition, each of Quoin Pharmaceuticals Ltd.’s non-employee directors receive an inaugural award of options to purchase shares valued at $165,000.
Exhibit 10.45
Compensation Program for Dr. Michael Myers
At the 2024 annual meeting of shareholders, the Company’s shareholders approved a compensation program for the Company’s Chief Executive Officer and Chairman of the Board, Dr. Michael Myers. The program sets forth the following compensation limitations applicable to Dr. Myers which the Compensation Committee and the Board can utilize in setting Dr. Myers’ compensation, beginning with the compensation to be paid in fiscal 2024, without the need to obtain further shareholder approval:
(i)an annual increase of base salary of up to 15% of Dr. Myers’ then effective base salary;
(ii)an annual cash bonus of up to 50% of Dr. Myers’ annual base salary during the fiscal year for which the annual cash bonus is paid (for example, Dr. Myers’ bonus to be paid in fiscal 2025 for fiscal 2024 services would be based upon a percentage, up to 50%, of Dr. Myers’ annual base salary in fiscal 2024); and
(iii)an annual equity grant in any form permitted under the Company’s equity incentive plan in effect from time to time with an annual value (determined in accordance with the Black-Scholes formula or another widely accepted and suitable formula for calculating the value of equity awards) of up to 500% of the maximum total fixed component (base salary and benefits) to which Dr. Myers is entitled in the grant year.
(together the “CEO Compensation Program”).
Exhibit 10.46
Compensation Program for Denise Carter
At the 2024 annual meeting of shareholders, the Company’s shareholders approved a compensation program for the Company’s Chief Operating Officer and a Member of the Board, Denise Carter. The program sets forth the following compensation limitations applicable to Ms. Carter which the Compensation Committee and the Board can utilize in setting Ms. Carter’s compensation, beginning with the compensation to be paid in fiscal 2024, without the need to obtain further shareholder approval:
(i)an annual increase of base salary of up to 15% of Ms. Carter’s then effective base salary;
(ii)an annual cash bonus of up to 50% of Ms. Carter’s annual base salary during the fiscal year for which the annual cash bonus is paid (for example, Ms. Carter’s bonus to be paid in fiscal 2025 for fiscal 2024 services would be based upon a percentage, up to 50%, of Ms. Carter’s annual base salary in fiscal 2024); and
(iii)an annual equity grant in any form permitted under the Company’s equity incentive plan in effect from time to time with an annual value (determined in accordance with the Black-Scholes formula or another widely accepted and suitable formula for calculating the value of equity awards) of up to 500% of the maximum total fixed component (base salary and benefits) to which Ms. Carter is entitled in the grant year.
(together the “COO Compensation Program”).
Exhibit 19.1
Quoin Pharmaceuticals Ltd.
Insider Trading Policy
This Insider Trading Policy (“Policy”) provides the standards of Quoin Pharmaceuticals Ltd. (“Quoin” or the “Company”) with respect to transactions in securities of the Company and the handling of confidential information about Quoin and the companies with which Quoin does business. The federal securities laws prohibit insider trading. Insider trading occurs when a person uses material nonpublic information obtained through involvement with the Company to make decisions to engage in transactions in Company's securities or transmits such information to any other person who may trade on the information. lease note that this Policy supplements the restrictions set forth in the Quoin Code of Ethics and Business Conduct.
This Policy applies to all transactions in the Company's securities, including ordinary shares, American Depositary Shares evidencing ordinary shares, options and any other securities that the Company may issue, as well as to derivative securities relating to any of the Company's securities.
From time to time, the Company may engage in transactions in its own securities. It is the Company’s policy to comply with all insider trading laws, rules and regulations, and any applicable listing standards when engaging in transactions in its own securities.
Section 1 of this Policy applies to all members of the Company’s board of directors and all officers and employees of the Company and its subsidiaries. Section 1 of this Policy also applies to such persons’ family members, other members of such persons’ households and entities controlled by such persons, as described in more detail below. The Company may also determine that other persons should be subject to Section 1 of this Policy, such as contractors or consultants. Section 2 of this Policy applies to all directors and executive officers of the Company and the employees listed in Appendix A hereto. Section 2 of this Policy also applies to such persons’ family members, other members of such persons’ households and entities controlled by such persons, as described in more detail below. Section 3 of this Policy sets forth additional requirements applicable to directors and officers of the Company under Section 16 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”).
If this Policy applies to you, it also applies to family members who reside with you (including a spouse, a child, a child away at college, stepchildren, grandchildren, parents, stepparents, grandparents, siblings, in-laws and adoptive relationships) or are financially dependent on you, and also includes other family members whose transaction in securities are directed by you or are subject to your influence or control (collectively referred to as “Family Members”). This Policy also applies to any other person who lives in your household and to any legal entities (such as a corporation, limited liability company, partnership or trust) that are influenced or controlled by you (collectively referred to as “Controlled Entities”).
Transactions by your Family Members, household members and Controlled Entities should be treated for the purposes of this Policy as if they were for your own account. Accordingly, all references to you with regard to all trading restrictions and pre-clearance procedures in this Policy also apply to your Family Members, household members and Controlled Entities. You are personally responsible for the actions of your Family Members, household members and Controlled Entities.
The Company has appointed the Chief Operating Officer of the Company as the Compliance Officer for this Policy. All determinations and interpretations by the Compliance Officer shall be final and not subject to further review. The Compliance Officer’s approval of a transaction submitted for pre-clearance
1
does not constitute confirmation that you do not possess material nonpublic information and does not relieve you of any of your legal obligations.
Any violation of this Policy may result in immediate dismissal and may subject you to both civil and criminal penalties. This is an extremely important matter, and we urge you to read the following with care. If you have any questions about this Policy, including its application to any proposed transaction, you may obtain additional guidance from the Compliance Officer by email at dcarter@quoinpharma.com. Do not try to resolve uncertainties on your own, as the rules relating to insider trading are often complex, not always intuitive and carry severe consequences.
SECTION 1: TRADING RESTRICTIONS AND GUIDELINES
Section 1 of this Policy applies to all of the directors, officers and employees of the Company and its subsidiaries. Section 1 of this Policy also applies to such persons’ Family Members, household members and Controlled Entities. All references to you with regard to all trading restrictions in this Policy also apply to your Family Members, household members and Controlled Entities. The Company may also determine that other persons should be subject to Section 1 of this Policy, such as contractors or consultants.
A. | General Policy - Prohibition Against Trading on, or Tipping, Material Nonpublic Information |
While in the possession of material nonpublic information relating to Quoin, you may not directly or indirectly through Family Members, household members and Controlled Entities or other persons or entities:
| 1. | Engage in transactions in Company securities, except as otherwise specified in this Policy under the heading “Certain Exceptions to the Trading Restrictions in this Policy;” |
| 2. | Recommend the purchase or sale of any Company securities; or |
| 3. | Communicate material nonpublic information concerning Quoin to any other person (including relatives, friends or business associates), except to the extent necessary to perform authorized work for Quoin or as required or specifically permitted by law or legal process. Nor should such information be discussed with any person within Quoin under circumstances where it could be overheard. Written information should be appropriately safeguarded and should not be left where it may be seen by persons not entitled to the information. |
In addition, if in the course of employment with the Company or the performance of services on the Company’s behalf, you learn material nonpublic information about another company with which the Company proposes to, or does, business, including a vendor, customer or supplier of the Company, you may not (i) trade in that company’s securities until the information becomes public or is no longer material, or (ii) communicate that information or make any recommendation relating to the buying or selling of securities of such company to any other person, including family and friends, business associates, or in any consulting capacity.
There are no exceptions to this Policy, except as specifically noted herein. Transactions that may be necessary or justifiable for independent reasons (such as the need to raise money for an emergency expenditure), or small transactions, are not excepted from this Policy. The securities laws do not recognize any mitigating circumstances, and, in any event, even the appearance of an improper
2
transaction must be avoided to preserve the Company’s reputation for adhering to the highest standards of conduct. This means that you may have to forgo a proposed transaction in the Company’s or another company’s securities even if you planned to make the transaction before learning the material nonpublic information and even though you believe that waiting may cause you to suffer an economic loss or not realize anticipated profit.
This Policy continues to apply to transactions in Company securities even after termination of service to Quoin. If an individual is in possession of material nonpublic information when his or her service terminates, that individual may not trade in Company securities until that information has become public or is no longer material.
B.Blackout periods for any or all personnel.
The Compliance Officer may issue instructions from time to time advising some or all personnel that they may not engage in transactions in Company securities for certain periods, or that our securities may not be traded without prior approval (“Event-Specific Blackout Period”). Due to the confidential nature of the events that may trigger these blackout periods, the Compliance Officer may find it necessary to inform affected individuals of a blackout period without disclosing the reason. If you are a director or an executive officer, you may also be subject to event-specific blackouts pursuant to the SEC’s Regulation BTR (Blackout Trading Restriction), as applicable. This regulation prohibits certain sales and other transfers by insiders during specified pension plan blackout periods. If you are made aware of such a blackout period, do not disclose its existence to anyone. Exceptions will not be granted during an Event-Specific Blackout Period.
C.Definitions
Material Information. Material information is any information that a reasonable investor would consider important in determining whether to buy, sell or hold securities. Positive or negative information may be material to investors. A determination as to whether information is material depends on all of the related facts and circumstances. Material information is not limited to historical facts but may also include projections and forecasts. Materiality is based on an assessment of all the facts and circumstances and is often evaluated by courts and enforcement authorities with the benefit of hindsight.
Information that you should consider material includes, but is not limited to:
| ● | earnings information and quarterly results; |
| ● | financial forecasts, including earnings estimates; |
| ● | changes in previously released forecasts; |
| ● | significant merger, acquisition or divestiture proposals or agreements; |
| ● | product developments; |
| ● | changes in auditors; |
| ● | significant changes in the Company’s prospects; |
| ● | significant or unusual borrowing or liquidity issues; |
| ● | equity or debt offerings; |
| ● | purchases or redemptions of securities; |
| ● | change in management or the Company’s board of directors; |
| ● | significant related party transactions; |
| ● | development of a significant new product or service; |
| ● | pending or threatened significant litigation, or the resolution of such litigation; and |
| ● | significant cybersecurity incidents. |
3
Nonpublic Information. Information that has not been disclosed to the public is generally considered to be nonpublic information. Information is considered to be public when it has been released in a manner that is reasonably designed to provide broad, non-exclusionary distribution (e.g., by means of a press release or an SEC filing) and after enough time has elapsed to permit the investment market to absorb and evaluate the information. As a general rule, information can be considered fully absorbed by the market beginning on the third full trading day following the public release of the information. Note that the information disseminated must be some form of “official” announcement. In other words, the fact that rumors, speculation, or statements attributed to unidentified sources are public is insufficient to be considered broadly distributed even when the information is accurate.
Officer. Officer means the individuals classified by the Company as officers for purposes of SEC rules under Section 16 of the Exchange Act.
D.Certain Exceptions to the Trading Restrictions in this Policy
The trading restrictions in this Policy do not apply in the case of the following transactions, except as specifically noted:
Exercise of Stock Options. The trading restrictions in this Policy do not apply to the exercise of an employee stock option acquired pursuant to the Company’s equity plans, or to the exercise of a tax withholding right pursuant to which a person has elected to have the Company withhold shares subject to an option to satisfy tax withholding requirements, as applicable. The trading restrictions in this Policy do apply, however, to any sale of stock as part of a broker-assisted cashless exercise of an option, or any other market sale for the purpose of generating the cash needed to pay the exercise price of an option.
Vesting of Restricted Stock Awards and Restricted Stock Unit Awards. The trading restrictions in this Policy do not apply to the vesting of restricted stock or restricted stock units, if any, or the exercise of a tax withholding right pursuant to which you elect to have the Company withhold shares of stock to satisfy tax withholding requirements upon the vesting of any restricted stock or restricted stock unit. The trading restrictions in this Policy do apply, however, to any market sale of restricted stock and shares of stock received upon the vesting of restricted stock units.
Rule 10b5-1 Trading Plan. The trading restrictions in this Policy do not apply to purchases or sales of the Company’s securities pursuant to a pre-approved Rule 10b5-1 trading program (a “Rule 10b5-1 Plan”). Implementation of a Rule 10b5-1 Plan under the Exchange Act provides an affirmative defense (which must be proven) from insider trading liability under Rule 10b-5. A Rule 10b5-1 Plan must be entered into at a time when the person entering into the plan is not aware of material non-public information. Once the plan is adopted, the person must not exercise any influence over the amount of securities to be traded, the price at which they are to be traded or the date of the trade. The plan must either specify the amount, pricing and timing of transactions in advance or delegate discretion on these matters to an independent third party. Entry into a Rule 10b5-1Plan must comply with the requirements set forth in “Rule 10b5-1 Plans” below.
E.Rule 10b5-1 Plans
Entry into a Rule 10b5-1 Plan requires the prior written approval of the Compliance Officer (which approval may include an email confirmation). Any Rule 10b5-1 Plan must be submitted for approval five days prior to the entry into the Rule 10b5-1 Plan. No further pre-approval of transactions conducted pursuant to the Rule 10b5-1 Plan will be required. Directors, executive officers and Designated Employees, as well as their respective Family Members, household members and Controlled Entities, may only adopt Rule 10b5-1 plans during a quarterly window period, as described in Section 2
4
below. You may not adopt a Rule 10b5-1 Plan during any Event-Specific Blackout Period, or at a time when you are aware of material non-public information. The following requirements apply to all Rule 10b5-1 Plans:
| ● | directors and Officers may not commence sales under a Rule 10b5-1 plan until the later of (i) 90 days following the date of adoption or modification of such plan; or (ii) two business days following the disclosure of the Company’s financial results in a Form 10-K or Form 10-Q relating to the fiscal quarter in which the Rule 10b5-1 plan was adopted or modified (but not to exceed 120 days following plan adoption or modification); |
| ● | all persons other than directors and Officers, may not commence sales under a Rule 10b5-1 plan until 30 days following the date of adoption or modification of such plan; |
| ● | directors and Officers must provide a representation in the Rule 10b5-1 plan certifying that, on the date of adoption or modification of the plan, they (i) are not aware of material nonpublic information about the Company or its securities; and (ii) are adopting or modifying the plan in good faith and not as part of a plan or scheme to evade the prohibitions of Rule 10b-5; |
| ● | subject to the limited exceptions set forth in Rule 10b5-1, you may not maintain multiple, overlapping plans; |
| ● | subject to the limited exceptions set forth in Rule 10b5-1, you can utilize only one single-trade plan (i.e. a plan designed to effect only a single transaction) during any 12 month period; and |
| ● | you must act in good faith with respect to the Rule 10b5-1 plan, not just in connection with entering into the plan. |
The Company may impose additional restrictions on Rule 10b5-1 Plans, including without limitation:
| ● | requiring that all plans be managed by an administrator selected by the Company; |
| ● | restrictions on termination or modification of plans; |
| ● | prohibition on entry into new plans for extended periods following termination of an existing plan; and |
| ● | prescribed periods during which persons may enter into plans. |
Modification or termination of Rule 10b5-1 Plans are generally discouraged absent compelling circumstances. Any modification to any Rule 10b5-1 Plan is treated as the entry into a new plan and must comply with all of the above requirements.
F.Violations of Insider Trading Laws
Penalties for trading on or communicating material nonpublic information can be severe, both for individuals involved in such unlawful conduct and their employers and supervisors, and may include jail terms, criminal fines, civil penalties and civil enforcement injunctions. Given the severity of the potential penalties, compliance with this Policy is absolutely mandatory. Individuals also may be prohibited from serving as directors or officers of the Company or any other public company. Keep in mind that there are no limits on the size of a transaction that will trigger insider trading liability; relatively small trades have in the past occasioned SEC investigations and lawsuits.
Legal Penalties. A person who violates insider trading laws by engaging in transactions in a company's securities when he or she has material nonpublic information can be sentenced to a substantial jail term and required to pay a penalty of several times the amount of profits gained or losses avoided. In addition, a person who tips others may also be liable for transactions by the tippees to whom he or she has disclosed material nonpublic information. Tippers can be subject to the same penalties and sanctions
5
as the tippees, and the SEC has imposed large penalties even when the tipper did not profit from the transaction.
The SEC can also seek substantial penalties from any person who, at the time of an insider trading violation, “directly or indirectly controlled the person who committed such violation,” which would apply to the Company and/or management and supervisory personnel. These control persons may be held liable for up to the greater of $1 million or three times the amount of the profits gained or losses avoided. Even for violations that result in a small or no profit, the SEC can seek a minimum of $1 million from a company and/or management and supervisory personnel as control persons.
Company-Imposed Penalties. An individual who violates this Policy may be subject to disciplinary action by the Company, including dismissal or removal for cause.
G.Additional Guidelines
The Company considers it improper and inappropriate for those employed by or associated with the Company to engage in short-term or speculative transactions in the Company’s securities or in other transactions in the Company’s securities that may lead to inadvertent violations of the insider trading laws. It therefore is the Company’s policy that any persons covered by this Policy may not engage in any of the following transactions (even if they do not possess material nonpublic information):
Short Sales. You may not engage in short sales of the Company’s securities (sales of securities that are not then owned), including a “sale against the box” (a sale with delayed delivery). Short sales of Company securities may evidence an expectation on the part of the seller that the securities will decline in value, and therefore have the potential to signal to the market that the seller lacks confidence in the Company’s prospects. In addition, short sales may reduce a seller’s incentive to seek to improve the Company’s performance. For these reasons, short sales of Company securities are prohibited.
Publicly Traded Options. You may not engage in transactions in publicly traded options related to the Company’s securities, such as puts, calls and other derivative securities, on an exchange or in any other organized market. Given the relatively short term of publicly traded options, transactions in options related to the Company’s securities may create the appearance that you are trading based on material nonpublic information and focus your attention on short-term performance at the expense of the Company’s long-term objectives. Accordingly, transactions in publicly traded options related to the Company’s securities on an exchange or in any other organized market are prohibited by this Policy.
Hedging Transactions. Hedging or monetization transactions can be accomplished through a number of possible mechanisms, including through the use of financial instruments such as prepaid variable forwards, equity swaps, collars and exchange funds. Such hedging transactions may permit you to continue to own Company securities obtained through employee benefit plans or otherwise, but without the full risks and rewards of ownership. When that occurs, you may no longer have the same objectives as the Company’s other shareholders. Therefore, you are prohibited from engaging in any such transactions.
Margin Accounts and Pledges. Securities held in a margin account or pledged as collateral for a loan may be sold without your consent by the broker if you fail to meet a margin call or by the lender in foreclosure if you default on the loan. A margin or foreclosure sale that occurs when you are aware of material nonpublic information may, under some circumstances, result in unlawful insider trading. Therefore, you are prohibited from holding Company securities in margin accounts or pledging Company securities as collateral for a loan.
6
SECTION 2: ADDITIONAL RESTRICTIONS ON TRADING APPLICABLE TO DIRECTORS, EXECUTIVE OFFICERS, AND DESIGNATED EMPLOYEES
Section 2 of this Policy imposes additional trading restrictions and applies to all directors and executive officers of the Company and the employees listed in Appendix A hereto (“Designated Employees”). This section of the Policy also applies to such persons’ Family Members, household members and Controlled Entities.
A.Trading Window Periods
Quarterly Trading Windows. Directors, executive officers and Designated Employees, as well as their respective Family Members, household members and Controlled Entities, can only engage in transactions in Company securities during an “open” “window period,” and only so long as such person does not have any material nonpublic information about Quoin. Quoin has established four “trading windows” of time during the fiscal year. The “trading windows” open on the third full trading day following Quoin’s public release of its quarterly or annual earnings for the prior fiscal quarter or fiscal year, as the case may be, continuing through the 15th day of the final month of each fiscal quarter (i.e. March 15th, June 15th, September 15th, and December 15th). Because directors, executive officers, and Designated Employees are especially likely to receive regular nonpublic information regarding Quoin’s operations, limiting trading to this “window period” helps prevent trading that is based on material information that is not available to the public. Before trading in Company securities during the “window period,” directors, executive officers, and Designated Employees, as well as their respective Family Members, household members and Controlled Entities, must also comply with the pre-clearance procedures discussed below.
Under certain very limited circumstances, a person subject to this restriction may be permitted to trade when the quarterly trading window is closed, but only if the Compliance Officer concludes that the person does not in fact possess material nonpublic information. Persons wishing to trade when the quarterly trading window is closed must contact the Compliance Officer for approval at least two business days in advance of any proposed transaction involving Company securities.
Event-Specific Blackout Periods. As described in Section 1(B) above, from time to time, an event may occur that is material to the Company (such as negotiation of mergers, acquisitions or dispositions) and is known by only a few directors, officers and/or employees. So long as the event remains material and nonpublic, persons designated by the Compliance Officer may not engage in transactions in Company securities. The existence of an Event-Specific Blackout Period will not be announced to the Company as a whole, and should not be communicated to any other person. Even if the Compliance Officer has not designated you as a person who should not trade due to an event-specific restriction, you should not trade while aware of material nonpublic information. Exceptions will not be granted during an Event-Specific Blackout Period.
B.Pre-Clearance
Quoin requires that all directors, executive officers and Designated Employees, as well as their respective Family Members, household members and Controlled Entities, obtain prior written approval from the Compliance Officer (which approval may include an email confirmation) before engaging in any transaction in Company securities. A request for pre-clearance should be submitted to the Compliance Officer at least two business days in advance of the proposed transaction. The Compliance Officer is under no obligation to approve a transaction submitted for pre-clearance, and may determine not to permit the transaction. If a person seeks pre-clearance and permission to engage in the transaction is denied, then he or she should refrain from initiating any transaction in Company securities, and should not inform any other person of the restriction. If approved, the transaction must be completed
7
within five business days, but in no event after the expiration of the applicable window period. If the transaction does not occur during the five business day period, pre-clearance of the transaction must be re-requested. A form of “Request for Approval” is attached as Appendix B hereto.
The Compliance Officer’s approval of a transaction submitted for pre-clearance does not constitute legal advice, does not constitute confirmation that you do not possess material nonpublic information and does not relieve you of any of your legal obligations.
The Compliance Officer may not engage in a transaction in Company securities unless the Company’s Chief Executive Officer has pre-cleared such transaction.
C.Exceptions
The quarterly trading window and event-specific trading restrictions do not apply to transactions to which the trading restrictions in this Policy do not apply as described in Section 1(D) above.
SECTION 3:ADDITIONAL REQUIREMENTS APPLICABLE TO DIRECTORS AND OFFICERS PURSUANT TO SECTION 16 OF THE EXCHANGE ACT.
Directors and Officers of the Company (the “Section 16 Insiders”) are also subject to the reporting and short-swing profit rules under Section 16 of the Exchange Act.
A. | Reporting Requirements |
Section 16(a) requires Section 16 Insiders to file reports with the SEC that identify their beneficial ownership of the Company's equity securities and any transactions they make in those securities. A Form 3 must be filed no later than the tenth (10th) calendar day after an individual becomes a director or Officer of the Company, and any subsequent change in beneficial ownership by a Section 16 Insider must, unless exempt from reporting or eligible for deferred reporting, be reported on a Form 4 filed within two business days. These reports must be filed with the SEC via EDGAR and are therefore immediately publicly available upon filing. Section 16(a) imposes the obligation to file ownership reports with the SEC on the individual insiders, not on the Company. However, the Company must disclose any delinquent Section 16 filers in its annual proxy statement and identify the trading information that was not properly filed. While it is not the Company’s obligation to do so, it is the Company’s practice to assist each of its Section 16 Insiders in filing their Section 16(a) reports.
In order to facilitate timely compliance, a Section 16 Insider (or his or her broker) must immediately report (no later than the same day such Section 16 Insider engages in the transaction) detailed trade information, in writing, to the Compliance Officer for all transactions made in Company securities by such insider, any Family Members, household members and Controlled Entities. Although it is the individual responsibility and legal obligation of each Section 16 Insider to comply with the reporting requirements described herein, the Compliance Officer will, upon being advised of a transaction, endeavor to prepare and, pursuant to a Section 16 Insider’s written authorization (including via email), timely file Section 16(a) reports on behalf of each Section 16 Insider. A Section 16 Insider may also sign a power of attorney that authorizes Company personnel to prepare, complete and file Section 16(a) reports, and otherwise act on a Section 16 Insider’s behalf regarding Section 16(a) reports. A copy of such power of attorney can be obtained from the Compliance Officer.
In addition to the disclosure requirements imposed by Section 16 of the Exchange Act, Section 16 Insiders are required to file a Form 144 before making an open market sale of Quoin securities. Form 144
8
notifies the SEC of your intent to sell Quoin securities. This form is generally prepared and filed by your broker.
B. | Short-Swing Profit Rules |
Section 16(b) provides for the recovery of “short-swing” profits from a Section 16 Insider resulting from certain transactions in Company securities “beneficially owned” by them. Specifically, a Section 16 Insider is required by law to turn over to the Company any “profit” realized upon a purchase followed by a sale, or a sale followed by a purchase, of any equity security of the Company that is beneficially owned by a Section 16 Insider and made within a period of less than six months. A profit may result even if the purchase and sale involve different types of equity securities. Moreover, any sale or purchase may be matched with any purchase or sale within the period such that there may be recoverable “profit” even if there has been no economic benefit to the individual in question. The good faith of a Section 16 Insider is irrelevant to whether recovery is required under Section 16.
Transactions in the Company’s securities by persons related to a Section 16 Insider (e.g., spouse, children, grandchildren and in-laws), or by entities in which he or she may have an indirect interest (e.g., a corporation, limited liability company, partnership or trust) may be attributed to the Section 16 Insider. Accordingly, such related persons or entities should be advised not to engage in trades within six months of trades engaged in by the Section 16 Insider, or engaged in by each other, without considering the implications of the short-swing profit rules.
Adopted: March 7, 2025
9
Appendix A
Designated Employees
Our current Designated Employees for purposes of our Insider Trading Policy are:
Dr. Michael Myers
Denise Carter
Gordon Dunn
The Compliance Officer may alter this list of Designated Employees at any time, in which case the Compliance Officer will provide written notice to any individuals to be added or removed from this list.
A-1
Appendix B
REQUEST FOR APPROVAL TO TRADE COMPANY SECURITIES
Number of Securities (e. g., ordinary shares or ADSs): | |
Type of Security [check all applicable boxes]:
☐ Ordinary Shares or ADSs | |
☐ Stock Option | |
☐ Restricted stock | |
☐ Debt Securities | |
Type of Transaction [check all applicable boxes]
☐ Stock option exercise (must complete applicable exercise form) | |
☐ Purchase | |
☐ Sale | |
☐ Gift (Name of Donnee) | |
☐ Rule l0b5-1 Plan (attach a copy of the 10b5-1 Plan to this request form) | |
☐ Sale under benefit plans | |
Broker Contact Information
Company Name | | |
Contact Name | | |
Telephone | | |
Fax | | |
Account Number | | |
Social Security or other Tax Identification Number | | |
Status (check all applicable boxes and complete blanks)
☐ Employee
☐ Board Member
I am not currently in possession of any material nonpublic information relating to Quoin Pharmaceuticals Ltd. I hereby certify that the statements made on this form are true and correct. I have also discussed any questions I had with respect to Quoin's securities trading policy and its applicability to the transactions contemplated hereby with Quoin’s Compliance Officer.
Signature | | Print Name | |
Date: | | Telephone Number | |
| |||
(office use only)
☐ Request Approved (transaction must be completed within 5 business days after approval) | |
☐ Request Denied | |
☐ Request Approved with the following modification | |
Signature of Quoin’s Compliance Officer & Date | |
B-1
C-1
Exhibit 23.1
Independent Registered Public Accounting Firm’s Consent
We consent to the incorporation by reference in the Registration Statement of Quoin Pharmaceuticals Ltd. on Form S-8 (File No. 333-270555) and Form S-1 (File No. 333-283734) of our report dated March 13, 2025, with respect to our audits of the consolidated financial statements of Quoin Pharmaceuticals Ltd. as of and for the years ended December 31, 2024 and 2023, which report is included in this Annual Report on Form 10-K of Quoin Pharmaceuticals Ltd. for the year ended December 31, 2024.
/s/ Marcum llp
Marcum llp
Morristown, New Jersey
March 13, 2025
Exhibit 31.1
CERTIFICATION PURSUANT TO
RULES 13a-14(a) AND 15d-14(a) UNDER THE SECURITIES EXCHANGE ACT OF 1934,
AS ADOPTED PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002
I, Dr. Michael Myers, certify that:
1. | I have reviewed this Annual Report on Form 10-K of Quoin Pharmaceuticals Ltd. (the “registrant”); |
2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report; |
4. | The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have: |
a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within these entities, particularly during the period in which this report is being prepared; |
b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
d) | Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
5. | The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions): |
a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and |
b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting. |
/s/ Dr. Michael Myers | |
Name: Dr. Michael Myers | |
Title: Chief Executive Officer | |
Date: March 13, 2025 | |
| |
Exhibit 31.2
CERTIFICATION PURSUANT TO
RULES 13a-14(a) AND 15d-14(a) UNDER THE SECURITIES EXCHANGE ACT OF 1934,
AS ADOPTED PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002
I, Gordon Dunn, certify that:
1. | I have reviewed this Annual Report on Form 10-K of Quoin Pharmaceuticals Ltd. (the “registrant”); |
2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report; |
4. | The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have: |
a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us, by others within those entities, particularly during the period in which this report is being prepared; |
b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
d) | Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
5. | The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions): |
a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and |
b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting. |
/s/ Gordon Dunn | |
Name: Gordon Dunn | |
Title: Chief Financial Officer | |
Date: March 13, 2025 | |
Exhibit 32.1
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350, AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
In connection with the Annual Report on Form 10-K of Quoin Pharmaceuticals Ltd. (the “Company”) for the year ended December 31, 2024, as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I certify, pursuant to 18 U.S.C. § 1350, as adopted pursuant to § 906 of the Sarbanes-Oxley Act of 2002, that, to the best of my knowledge:
| (1) | The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and |
| (2) | The information contained in the Report fairly presents, in all material respects, the financial condition and result of operations of the Company. |
/s/ Dr. Michael Myers | |
Name: Dr. Michael Myers | |
Title: Chief Executive Officer | |
Date: March 13, 2025 | |
The foregoing certification is being furnished solely to accompany the Report pursuant to 18 U.S.C. § 1350, and is not being filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, and is not to be incorporated by reference into any filing of the Company, whether made before or after the date hereof, regardless of any general incorporation language in such filing.
Exhibit 32.2
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350, AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
In connection with the Annual Report on Form 10-K of Quoin Pharmaceuticals Ltd. (the “Company”) on Form 10-K for the year ended December 31, 2024, as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I certify, pursuant to 18 U.S.C. § 1350, as adopted pursuant to § 906 of the Sarbanes-Oxley Act of 2002, that, to the best of my knowledge:
(1) | The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and |
(2) | The information contained in the Report fairly presents, in all material respects, the financial condition and result of operations of the Company. |
/s/ Gordon Dunn | |
Name: Gordon Dunn | |
Title: Chief Financial Officer | |
Date: March 13, 2025 | |
The foregoing certification is being furnished solely to accompany the Report pursuant to 18 U.S.C. § 1350, and is not being filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, and is not to be incorporated by reference into any filing of the Company, whether made before or after the date hereof, regardless of any general incorporation language in such filing.
